Clear Sky Science · en
Low-penetrance TP53 variants are mainly hypomorphic: an underestimated issue with high clinical significance
Why some inherited cancer risks fly under the radar
The TP53 gene, often called the guardian of our DNA, is famous for its powerful role in stopping cells from turning cancerous. Doctors have long known that certain rare, "all-or-nothing" TP53 mutations can almost guarantee cancer early in life. This paper explores a quieter, much less understood side of TP53: inherited changes that weaken, but do not completely destroy, the gene’s protective power. These subtle variants are far more common than previously thought and could reshape how we judge cancer risk and plan screening for many families.
A key bodyguard gene with many shades of strength
TP53 tells cells when to pause, repair damage, or self-destruct if their DNA is too injured. Classic high-risk TP53 mutations, seen in Li-Fraumeni syndrome, switch this bodyguard off almost entirely, leaving people with a very high chance of developing multiple cancers at young ages. But many TP53 changes are missense variants, which swap just one DNA letter and may only partly weaken the protein. Until now, most attention has gone to the most dangerous forms, while the milder, low-penetrance variants — those that raise cancer risk only modestly and not in every carrier — have remained in a grey zone.
Finding the quiet troublemakers in massive mutation databases
The authors analyzed the 2025 release of the UMD_TP53 database, the largest collection of TP53 mutations from tumors and inherited cases worldwide, and cross-checked it with other cancer and population datasets. They focused on how often each TP53 variant appeared in tumors (somatic) versus being inherited (germline), calculating a “germline-versus-somatic ratio” (GVSr). Most cancer-driving TP53 mutations show low ratios, appearing frequently as acquired changes in tumors. In contrast, the team discovered a distinct set of variants with unusually high GVSr: they cropped up disproportionately often as inherited changes but were only modestly represented as stand‑alone drivers in tumors. Many of these had already been reported in families with atypical or milder cancer patterns, hinting that they might represent a special low-penetrance class.
Part-time guardians: what the function tests reveal
To understand what these high-GVSr variants actually do in cells, the researchers combined data from several large-scale experiments that had systematically tested thousands of TP53 variants in yeast and mammalian cells. Across these independent assays, the same pattern emerged: the high-GVSr variants rarely behaved like completely broken TP53, yet they were not fully normal either. Instead, they showed intermediate activity — strong enough to activate many protective genes but clearly weaker than the healthy version. Tumor samples and cancer cell lines carrying these variants also showed only a partial dampening of TP53-controlled gene programs, unlike the dramatic shutdown seen with classic hotspot or nonsense mutations. Together, this places most of these variants in a “hypomorphic” category: weakened but not wiped out.
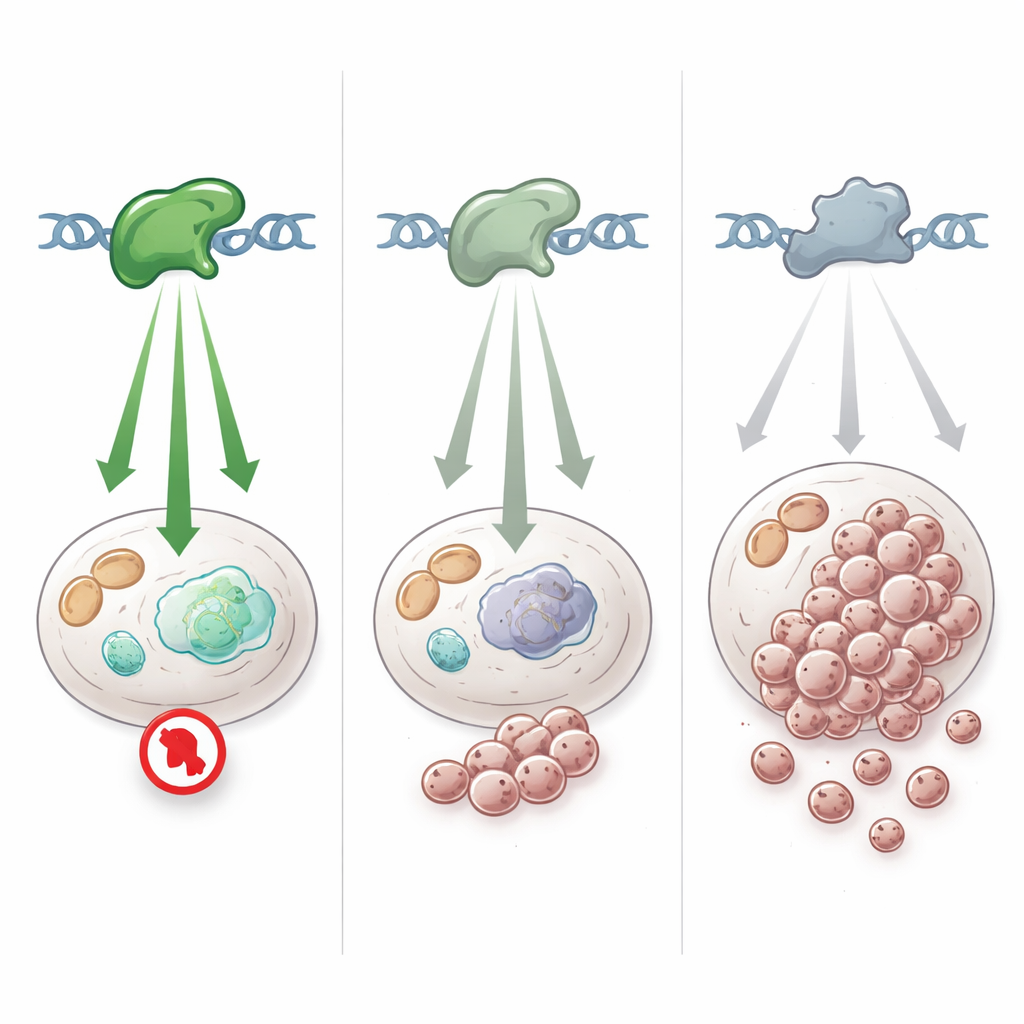
How subtle variants shape cancer risk in real life
Genetic and clinical data suggest that people inheriting these hypomorphic TP53 variants face a spectrum of cancer risk that is lower and more variable than in classic Li-Fraumeni syndrome. Some variants, such as changes at positions 181 or 337 of the protein, cluster in particular populations and are linked to increased rates of certain cancers, like breast tumors or childhood adrenal cancers, but with incomplete penetrance — many carriers never develop cancer at all, or do so later in life. Tumors that do arise in carriers often pick up a second, more disruptive TP53 mutation, implying that the weaker variant alone is not powerful enough to launch cancer but can still contribute to risk when combined with other hits or modifying genes.
What this means for patients and their families
This work shows that many inherited TP53 variants are not simply safe or dangerous; they sit along a continuum of strength. The newly defined high-GVSr, hypomorphic variants act as part-time guardians: they reduce, but do not abolish, the cell’s natural defenses against cancer. For patients, this means that some TP53 findings reported as uncertain or conflicting may in fact confer a real, though moderate, rise in risk. Recognizing this group and separating them from harmless background variation will allow more nuanced genetic counseling, better-tailored screening schedules, and, in time, treatment strategies that exploit the residual TP53 function. The authors estimate that such low-penetrance, hypomorphic variants could account for more than a third of all non-hotspot TP53 mutations, underscoring their importance for precision oncology.
Citation: Rodriguez, L., Leroy, B., Toledo, F. et al. Low-penetrance TP53 variants are mainly hypomorphic: an underestimated issue with high clinical significance. npj Genom. Med. 11, 22 (2026). https://doi.org/10.1038/s41525-026-00568-x
Keywords: TP53 variants, hypomorphic mutations, low-penetrance cancer risk, genetic counseling, precision oncology