Clear Sky Science · en
Complement inhibition by a unique cluster of immunomodulatory outer surface proteins of Borrelia recurrentis
Why a blood-borne fever germ is hard to kill
Louse-borne relapsing fever is a severe yet neglected disease that still affects people living in poverty and crowded conditions. It is caused by the bacterium Borrelia recurrentis, which can survive for long periods in human blood despite a powerful immune surveillance system. This study uncovers a new family of five surface proteins, called ChiA to ChiE, that help the microbe dodge our natural defenses. Understanding how these proteins work could open doors to better diagnostics, treatments, or vaccines against this deadly infection. 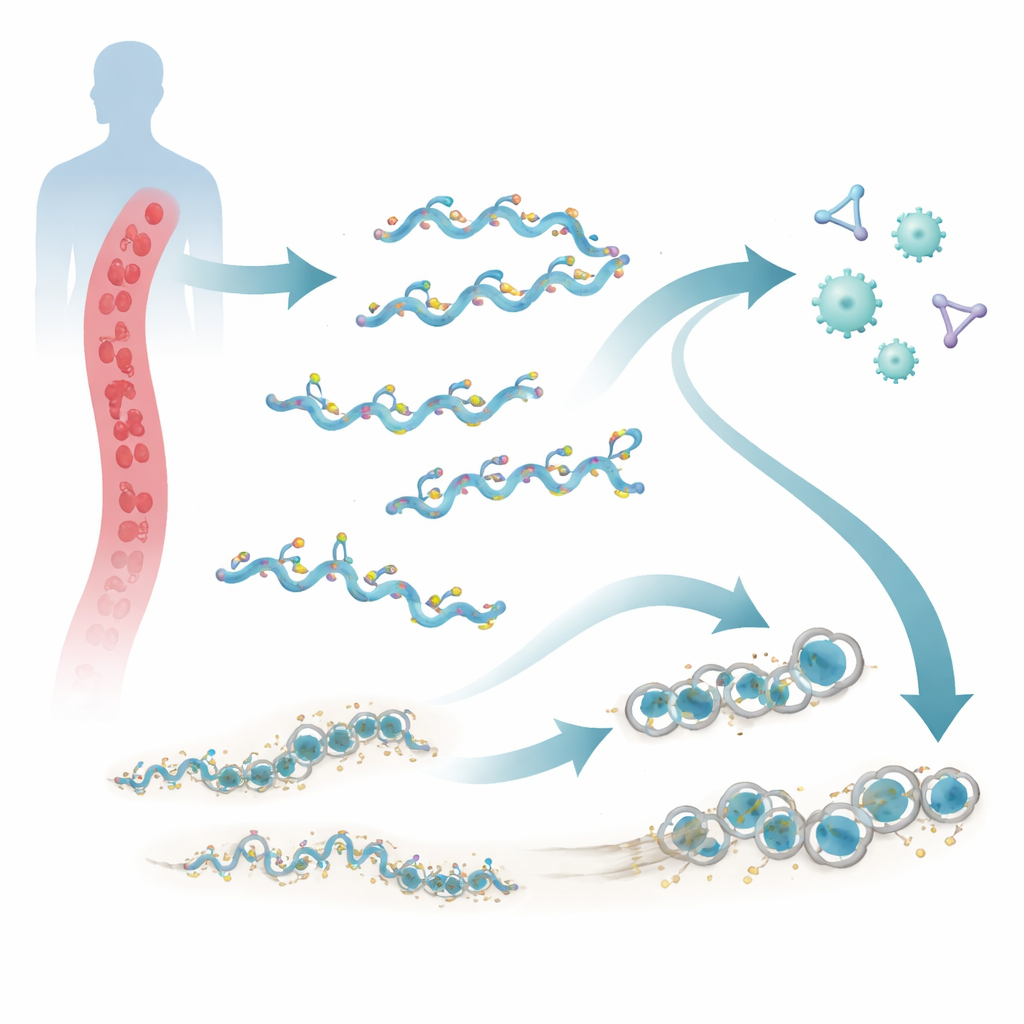
The body’s shield and how this germ slips past it
Our blood carries a network of proteins known as the complement system, which patrols for intruders. When activated, complement marks microbes for destruction and can punch holes in their membranes with a ring-shaped weapon called the membrane attack complex. Borrelia recurrentis lives directly in the bloodstream and therefore faces this attack from the moment it enters the body. Earlier work showed that the bacterium can borrow human protective factors to tone down complement. The new research reveals that B. recurrentis also brings its own tools: a cluster of five related outer surface proteins, ChiA through ChiE, located together on a large DNA segment called a megaplasmid.
A hidden toolkit of surface proteins
Using genetic comparisons across different patient isolates, the authors found that this Chi gene cluster is highly conserved in B. recurrentis, implying that it is important for survival. The Chi proteins are lipoproteins anchored in the outer surface of the bacterium, where they can directly meet incoming immune molecules. Structural work with X-ray crystallography and advanced computer modeling showed that all five share a compact, eight-helix core and a protruding region the authors term the S-domain. At the heart of each protein lies a deep oily pocket; in one protein, ChiB, this pocket holds a phospholipid, suggesting that these molecules may bind fats from their surroundings. Two members, ChiC and ChiE, contain pairs of sulfur-bearing amino acids positioned so they can form reversible bonds, hinting that these proteins may switch their behavior depending on the local chemical environment in the host. 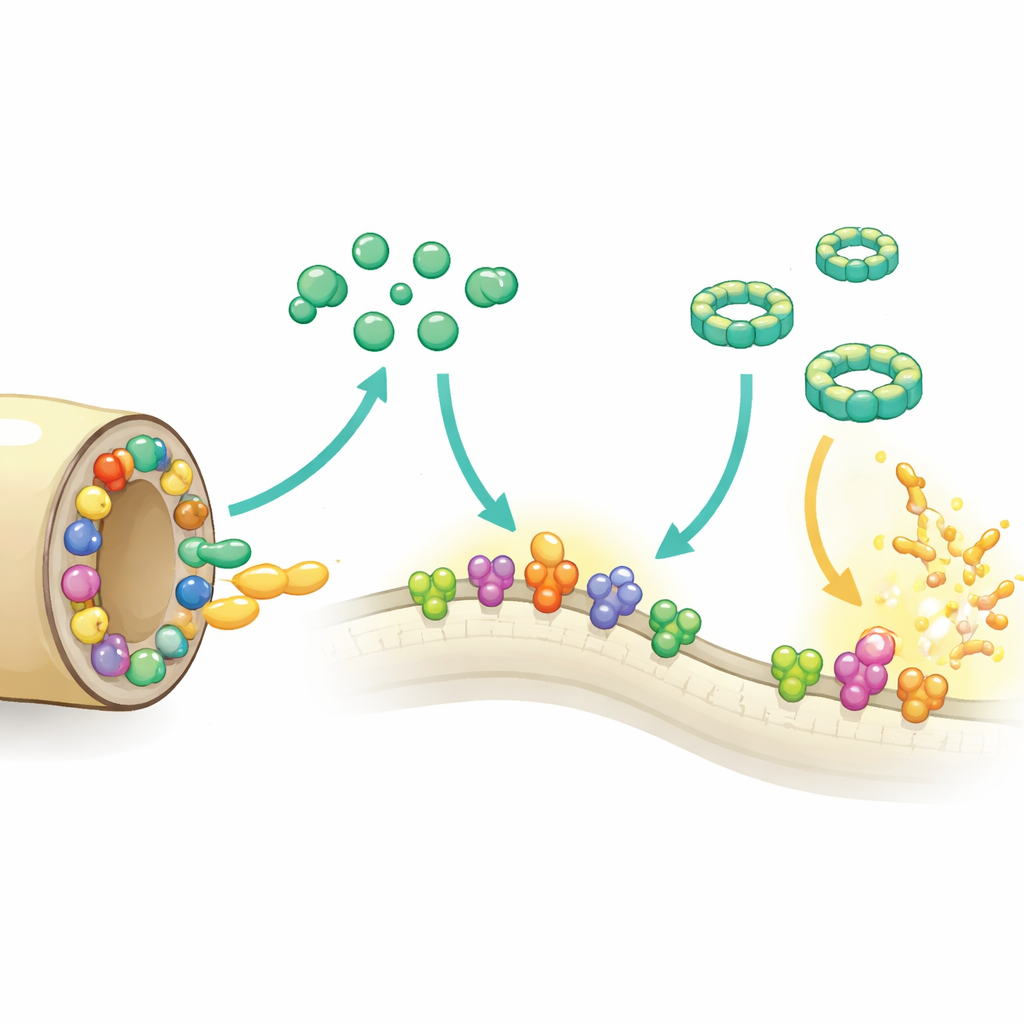
Shutting down immune attack at multiple steps
The team tested how each Chi protein affects the complement system in test-tube experiments. All five could tone down the so-called alternative pathway, one of the main routes by which complement is triggered, although ChiA was weaker. ChiE went further and also dampened the classical and lectin pathways, which are activated by antibodies or sugar patterns on microbes. Several Chi proteins, especially ChiB, ChiC, ChiD, and ChiE, interfered with the final stage in which complement proteins assemble into damaging rings on cell surfaces, preventing full ring formation and protecting red blood cells in a hemolysis assay. The proteins bound key complement components, such as C3b and parts of the classical pathway, and in doing so blocked the cascade without needing to cut these molecules themselves.
Borrowing a human enzyme to clear a path
Beyond blocking complement directly, the Chi proteins also harness another human system: plasminogen, the inactive form of the tissue-dissolving enzyme plasmin. Most Chi proteins bound plasminogen tightly, and once a human activator was added, the captured plasmin could cut complement component C3b. This means B. recurrentis can decorate its surface with host-derived enzymes that help trim away complement tags and possibly other barriers, such as tissue structures, aiding spread through the body. When the researchers added purified Chi proteins to a serum-sensitive Borrelia strain, or engineered that strain to produce particular Chi proteins, the bacteria survived much better in human serum, confirming that these molecules make complement attack less lethal in practice.
What this means for patients and prevention
Together, the findings reveal a specialized family of surface proteins that give Borrelia recurrentis a layered defense against the human immune system. The Chi proteins block complement at several stages and recruit human enzymes to further weaken immune tagging, allowing the bacterium to persist in blood and cause repeated fever episodes. For non-specialists, the key message is that this microbe survives not by hiding, but by actively disarming our molecular defenses. By mapping the shapes and functions of these Chi proteins, the study provides a blueprint for future work to design drugs or vaccines that neutralize them, potentially restoring the body’s ability to clear this neglected but dangerous infection.
Citation: Röttgerding, F., Reyer, F., Gerlach, E. et al. Complement inhibition by a unique cluster of immunomodulatory outer surface proteins of Borrelia recurrentis. Nat Commun 17, 3900 (2026). https://doi.org/10.1038/s41467-026-72359-y
Keywords: louse-borne relapsing fever, Borrelia recurrentis, complement evasion, bacterial surface proteins, plasminogen binding