Clear Sky Science · en
Ascites protects against ferroptosis and enables the peritoneal growth of ovarian cancer
Why fluid in the belly matters
Many women with advanced ovarian cancer develop a build-up of fluid in the abdomen called ascites. Doctors have long known that this fluid signals serious disease, but not exactly how it helps tumors survive and spread. This study uncovers how components within ascites shield ovarian cancer cells from a particular kind of cell death and, in doing so, support the growth of cancer on the lining of the abdomen.
A dangerous partnership between fluid and cancer
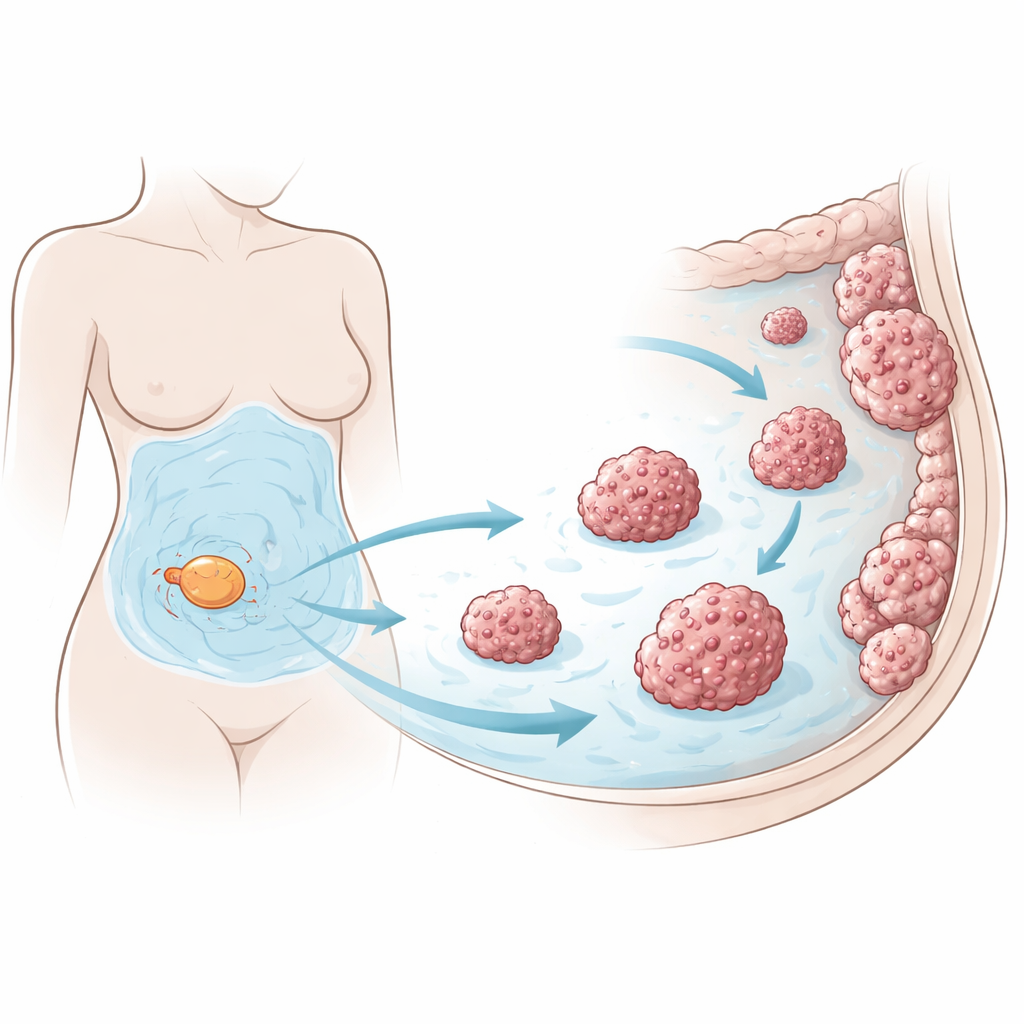
Ovarian cancer often spreads across the inner surface of the abdomen, a region called the peritoneum. As the disease advances, leaky blood vessels cause liters of ascitic fluid to collect in this cavity. The researchers focused on a vulnerability of ovarian cancer cells: they are highly prone to ferroptosis, a form of cell death driven by iron and damage to fats in cell membranes. Because detached cancer cells drifting in the abdomen should be especially exposed to this stress, the team asked how they manage to stay alive long enough to seed new tumors.
Ascites shields cells from a lethal stress
Using human ovarian cancer cell lines, fresh tumor cells from patients, and tiny three-dimensional organoids grown from patient tissue, the team exposed cells to drugs that normally trigger ferroptosis. They found that even small amounts of ascites from women with ovarian cancer strongly protected all of these models from ferroptotic death, while not shielding them from other types of toxic drugs. In mice, injecting ovarian cancer cells together with human ascites led to larger tumor burdens in the peritoneal cavity, fewer signs of fat damage in the tumors, and shorter survival, indicating that the fluid’s protective effect operates inside living animals, not just in dishes.
Fat-rich cargo in the fluid drives protection
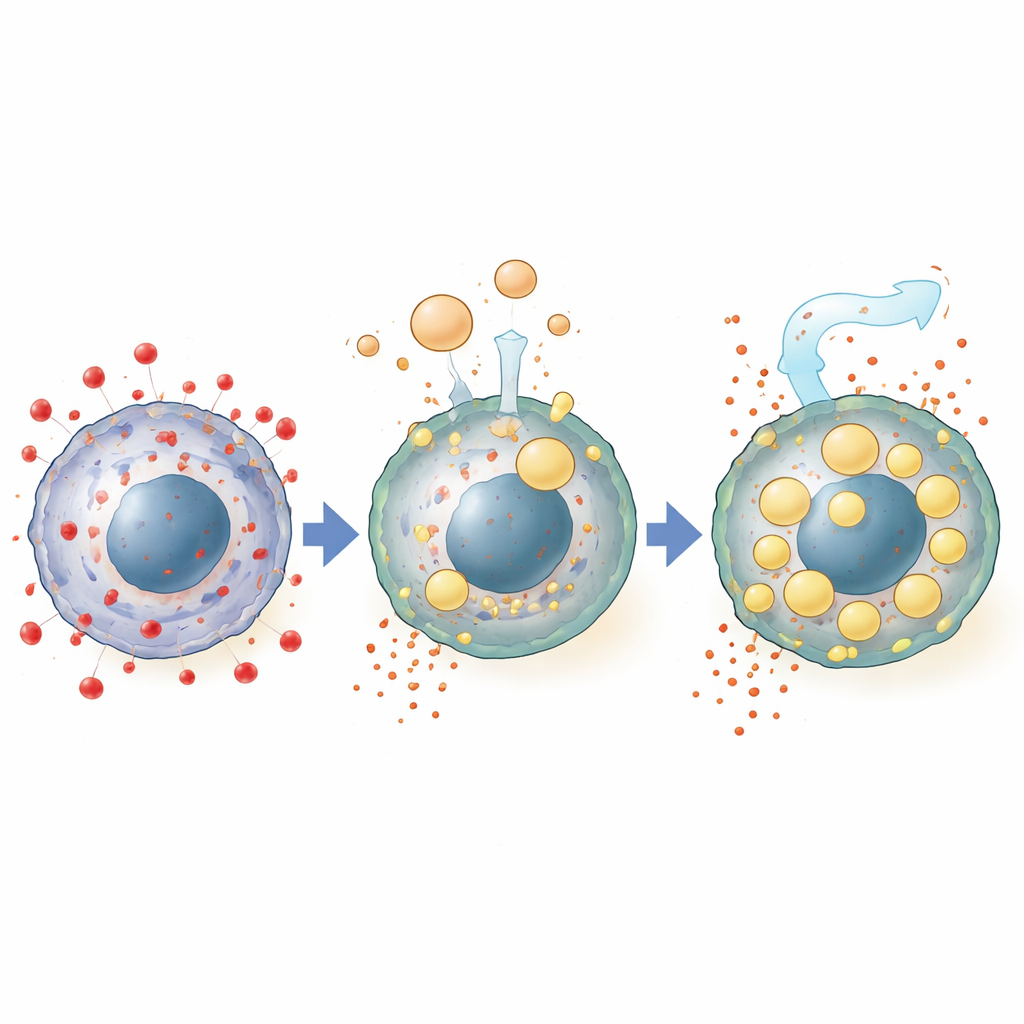
To pinpoint what in ascites carries this power, the scientists removed different components. Stripping out lipids, but not proteins or small molecules, erased the protection. Detailed chemical profiling showed that cancer cells exposed to ascites accumulated large amounts of neutral fats, especially triglycerides and cholesteryl esters, many of which matched species present in the fluid itself. These fats packed into numerous lipid droplets inside the cells, structures thought to act as safe storage depots for vulnerable fatty acids. Blocking the formation of these droplets, or interfering with key fat-uptake pathways from high-density lipoprotein particles in the fluid, reduced the protective effect, tying ferroptosis resistance directly to fat import and storage.
Rewiring cell metabolism and iron intake
Ascites did more than simply donate fats. Gene activity measurements revealed that the fluid suppressed a mitochondrial enzyme called HMGCS2, which normally supports the burning of fatty acids. Loss of this enzyme shifted cells away from fat breakdown and toward storing fats in droplets, further buffering them against damage. At the same time, ascites dampened levels of the transferrin receptor TFRC on the cell surface, reducing the influx of iron that helps fuel ferroptosis. The fatty acid oleic acid, abundant in ovarian cancer ascites, could mimic these effects, lowering TFRC activity and iron inside cells while enhancing survival under ferroptotic stress.
Turning protection into a treatment opportunity
Because this shield depends on altered fat handling, the researchers tested drugs called fibrates, commonly used to lower blood lipids by activating a protein switch named PPARα. Bezafibrate and related compounds partially reversed the lipid build-up caused by ascites, shrank lipid droplets and restored sensitivity to ferroptosis-inducing drugs in cells, organoids and mouse models. When cancer cells were pretreated with both a ferroptosis-triggering agent and bezafibrate before being placed into mice, peritoneal tumor growth slowed and the animals lived longer, suggesting that existing lipid-targeting medicines might help undermine the advantages ascites gives to metastatic cancer cells.
What this means for patients
This work shows that ascites is not just a passive by-product of advanced ovarian cancer, but an active partner that protects tumor cells from a specific kind of fat-based cell death. By donating lipids, promoting their safe storage and dialing down iron uptake, the fluid helps free-floating cancer cells survive the journey across the abdomen and form new implants. At the same time, the study highlights that this dependence on fat handling and iron control may be a weakness. Carefully designed therapies that upset this balance, possibly including repurposed lipid-lowering drugs, could one day help strip ovarian cancer cells of their protection and make them more vulnerable to treatment.
Citation: Setayeshpour, Y., Chen, SY., Dayanidhi, D.L. et al. Ascites protects against ferroptosis and enables the peritoneal growth of ovarian cancer. Nat Commun 17, 4190 (2026). https://doi.org/10.1038/s41467-026-72116-1
Keywords: ovarian cancer, ascites, ferroptosis, lipid droplets, fibrates