Clear Sky Science · en
Human milk oligosaccharide mediates mutualism between Escherichia coli and Bifidobacterium bifidum
Why baby milk sugars matter
Parents often hear that “breastfeeding is good for the microbiome,” but what that actually means can feel mysterious. This study peeks behind the curtain to show how specific sugars in human milk help two common gut bacteria, Escherichia coli and Bifidobacterium bifidum, cooperate instead of compete. By following babies and their mothers through the first year of life, and then recreating key interactions in the lab, the authors reveal a surprising story: milk sugars not only feed friendly microbes, they can also choreograph a delicate partnership that keeps potentially troublesome E. coli in check.
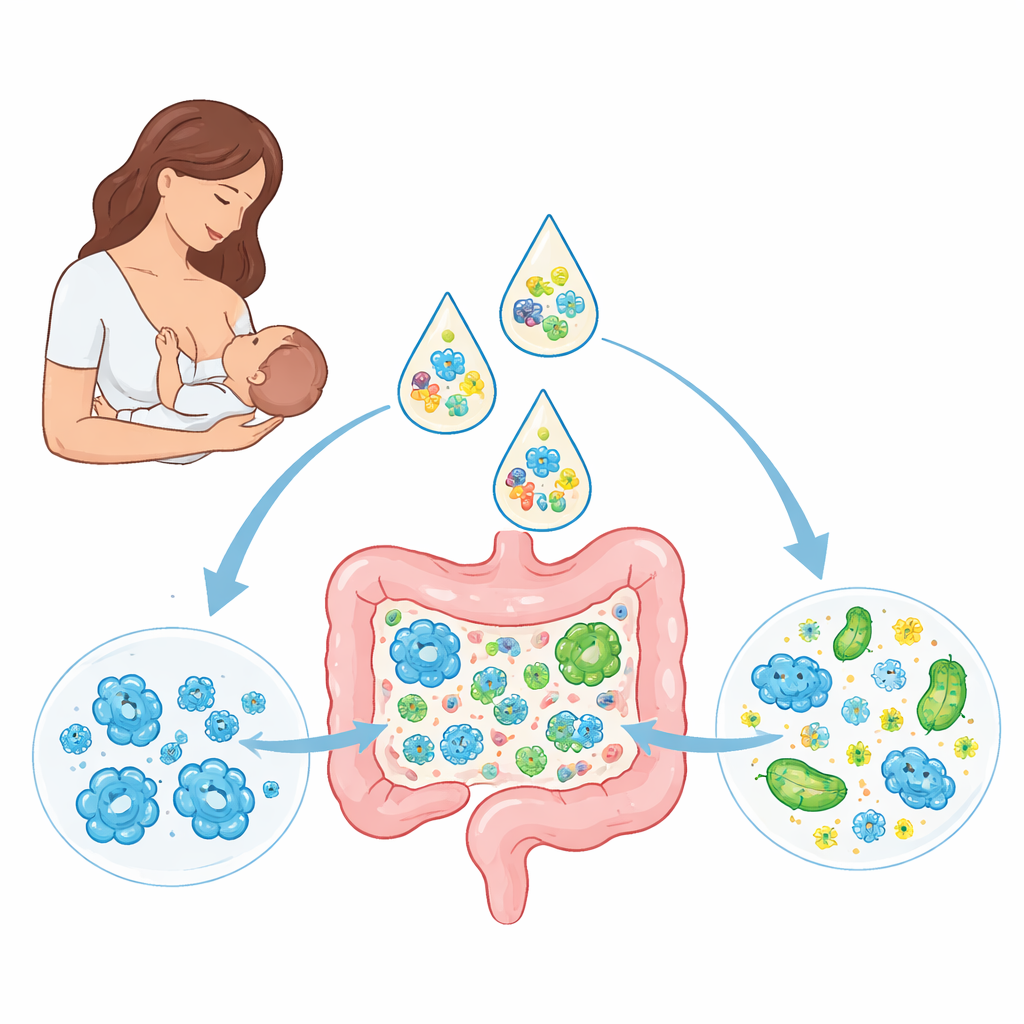
Early guests in the baby gut
The researchers followed 41 healthy, full‑term Dutch infants who were all exclusively breastfed at first, plus 30 of their mothers. Using deep DNA sequencing of stool samples at several ages, they mapped which microbes were present and how communities changed over time. As expected, species of Bifidobacterium were dominant in many breastfed babies and were often shared between mothers and infants, pointing to direct transfer within families. E. coli, a member of the Enterobacteriaceae family that can cause disease if it overgrows, appeared in most infants but at low levels. Interestingly, E. coli strains in babies did not match those of their mothers, suggesting that these bacteria usually arrive from other environmental sources yet can persist for months once established.
Patterns of co‑existence, not warfare
To test how individual species shape the broader community, the team built a new computational pipeline called MAJIC. Instead of looking only at who is present, MAJIC compares whole communities when a given species is there versus when it is absent, asking whether that species tends to narrow or broaden the possible community states. In very young infants, Bifidobacterium species were abundant but behaved like “background players”: losing one did not drastically change who else could live there, implying that other microbes could step in and perform similar roles. Across the first year, the authors found no signal that common Bifidobacterium species were suppressing E. coli. Rather than a direct battle, their frequent co‑occurrence looked more like peaceful sharing of a habitat shaped by the same food supply—milk sugars.
Zooming in on strains and genes
By reconstructing hundreds of bacterial genomes from the samples, the researchers could distinguish not just species but individual strains and their genetic variation. Bifidobacterium longum subspecies longum, for example, was often shared between mothers and infants and persisted over time in the same child, suggesting strong adaptation to the infant gut. B. bifidum showed particularly tight genetic constraints in key sugar‑handling genes, as if finely tuned to its niche. E. coli, in contrast, displayed high microdiversity, with multiple strains coexisting within an infant and less evidence of strict host‑specific tuning. Despite these differences, both Bifidobacterium and E. coli carried suites of genes linked to enzymes that break down complex carbohydrates, hinting at shared pressure to capture and use milk‑derived sugars efficiently.
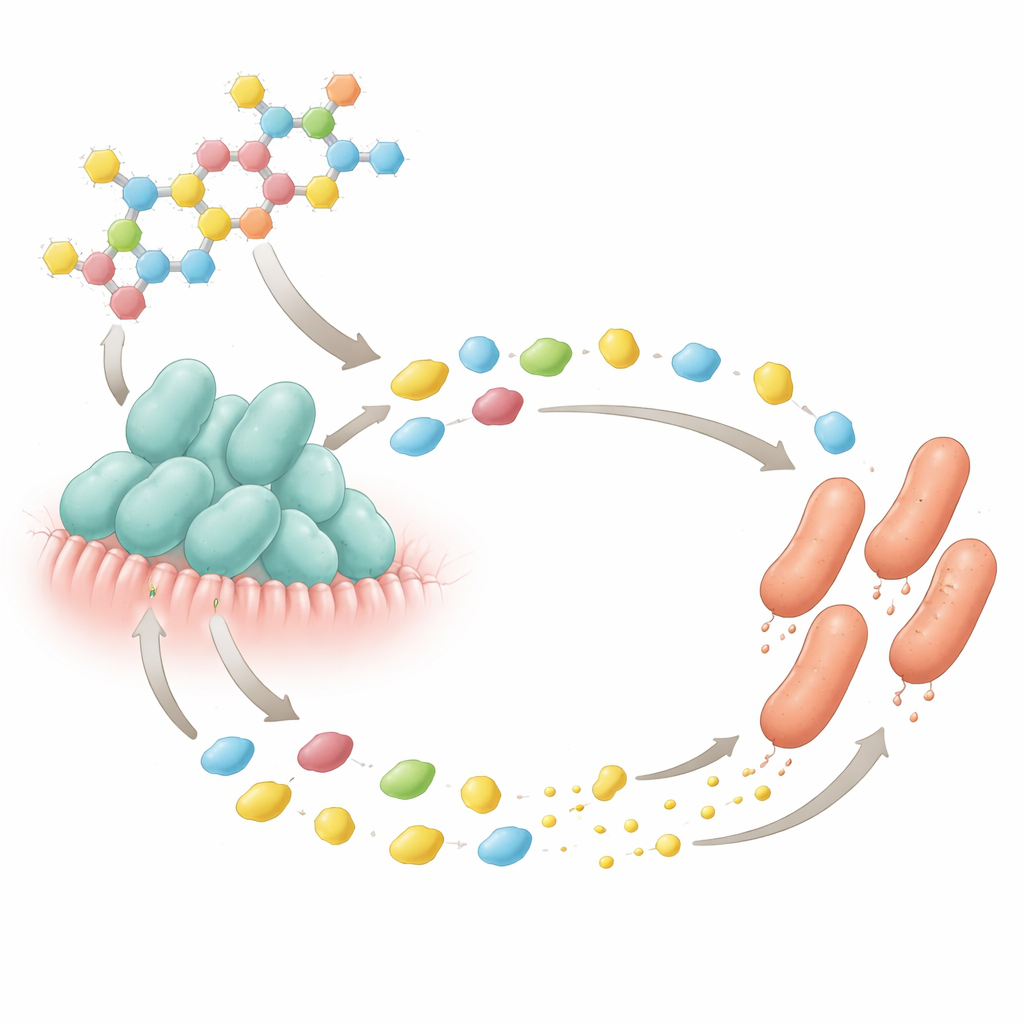
A microscopic sugar‑for‑nutrient deal
The most striking results came from lab experiments that recreated a key milk‑sugar interaction. The authors focused on 2′‑fucosyllactose, one of the most abundant complex sugars in human milk. B. bifidum isolates from infant stool could chop this sugar into simple building blocks but needed the amino acid cysteine to grow. E. coli could not digest the intact sugar but flourished on the released simple sugars. When the two microbes were grown together in a medium containing 2′‑fucosyllactose but no added cysteine, both grew well—yet neither could thrive alone under those conditions. Further tests with a mutant E. coli unable to make cysteine showed that normal E. coli was supplying this missing nutrient to B. bifidum, while B. bifidum provided the simple sugars that fueled E. coli. This revealed a tight two‑way exchange: a mutualism mediated by a human‑milk sugar.
What this means for infant health
For non‑specialists, the key message is that breastmilk does more than “feed the baby” or even “feed good bacteria.” Its complex sugars can set up cooperative relationships between different microbes that share resources. In this case, B. bifidum and E. coli form a sugar‑for‑nutrient trade that may help keep E. coli present but not dominant, while supporting a stable, efficient community that turns milk into useful metabolites for the infant. The work suggests that early‑life gut ecosystems are shaped by both competition for simple host‑derived sugars like lactose and by cross‑feeding on milk oligosaccharides. Understanding these invisible bargains may guide better formulas, probiotics, or diet tweaks aimed at steering the infant microbiome toward a stable, health‑promoting state.
Citation: Seki, D., Pollak, S., Kujawska, M. et al. Human milk oligosaccharide mediates mutualism between Escherichia coli and Bifidobacterium bifidum. Nat Commun 17, 3489 (2026). https://doi.org/10.1038/s41467-026-71764-7
Keywords: infant gut microbiome, human milk oligosaccharides, Bifidobacterium, Escherichia coli, cross-feeding