Clear Sky Science · en
Cryo-EM structure of Chlamydomonas reinhardtii Photosystem I complexed with cytochrome c6
How Green Algae Keep Sunlight Flowing
Every green leaf and algal cell depends on an invisible relay race of electrons to turn sunlight into usable energy. This study looks at one tiny handoff in that race: how a small carrier protein called cytochrome c6 delivers electrons to a giant light-harvesting machine known as Photosystem I in green algae. By freezing these molecules in action and imaging them at near-atomic detail, the authors reveal how this crucial encounter works and how it connects to the evolutionary history of photosynthesis.
A Careful Chain of Energy Steps
In oxygen-producing photosynthesis, energy flows through a chain of large protein complexes buried in internal membranes. Photosystem II uses light to split water and pass electrons to a middle station called the cytochrome b6f complex. From there, small soluble carriers—either copper-based plastocyanin or iron-based cytochrome c6—shuttle electrons through the watery interior to Photosystem I. When Photosystem I receives these electrons, it helps generate the electrical and chemical gradients that ultimately power the cell’s production of ATP, the universal energy currency.
An Ancient Partner Meets a Modern Machine
From an evolutionary perspective, the earliest versions of Photosystem I are thought to have worked mainly with cytochrome c6. Over time, many organisms, especially plants, switched to using plastocyanin instead, partly because it uses copper instead of iron, which can be scarce. Green algae such as Chlamydomonas reinhardtii sit in the middle of this story: they can use both carriers, changing preference depending on metal availability. Understanding exactly how cytochrome c6 attaches to Photosystem I in these algae offers a window into how ancient and modern strategies of electron delivery coexist and how they may have evolved.
Freezing the Moment of Contact
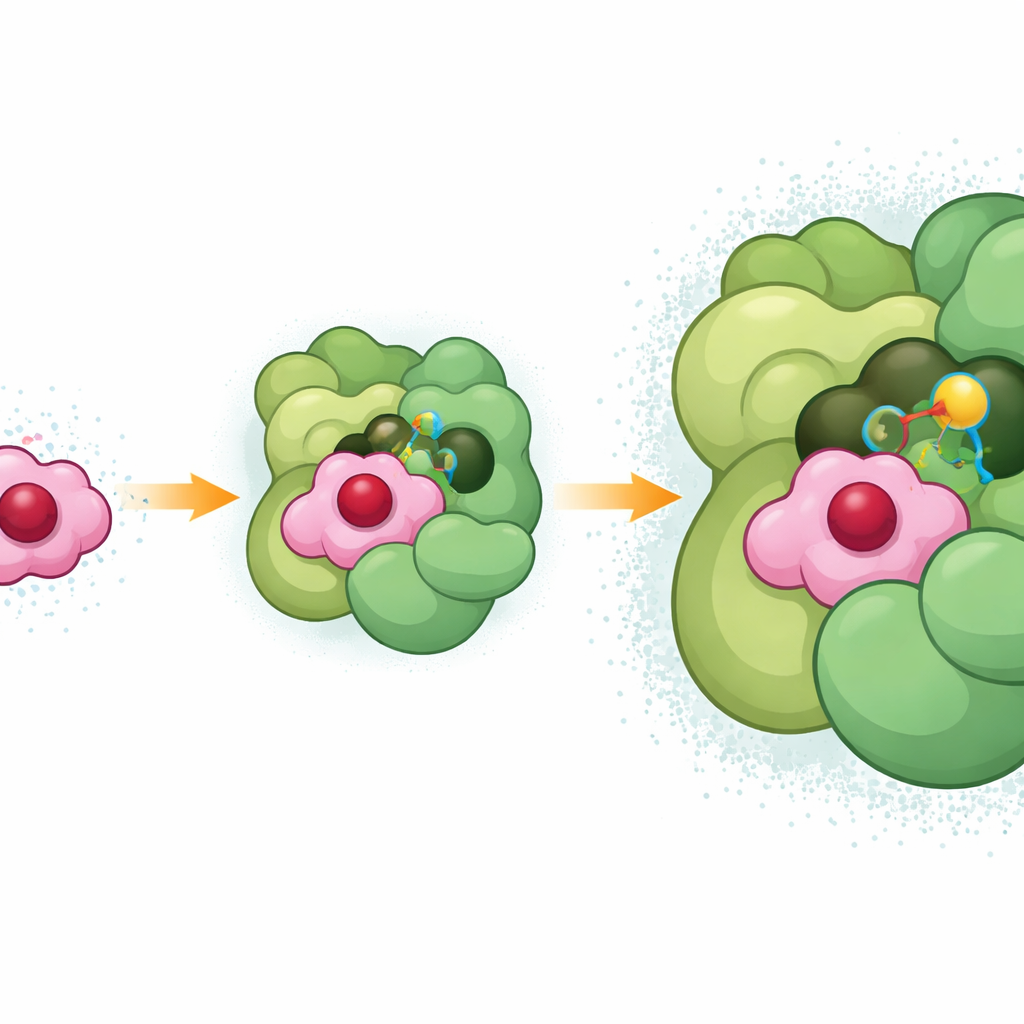
The researchers created stable complexes between cytochrome c6 and Photosystem I by using a short-range chemical crosslink that bonds the two partners only when they are naturally close. They then used high-resolution cryo–electron microscopy to reconstruct the three-dimensional structure of this frozen encounter at about two-angstrom detail, fine enough to place individual amino acid side chains, pigment molecules, and even many water molecules. The structure shows cytochrome c6 nestled into a shallow pocket formed where two core subunits of Photosystem I meet, with an extra surface helix from another subunit (PsaF) reaching over like a small arm to help hold it in place.
A Precise Fit for Fast Electron Flow
Within this pocket, the heme group of cytochrome c6—the part that actually carries the electron—sits only about eleven angstroms away from the special chlorophyll pair in Photosystem I that receives the electron. The interface is supported by a dense web of interactions: negatively charged spots on cytochrome c6 attract positively charged regions on PsaF, while neutral and aromatic side chains make snug, oil-like contacts deeper in the groove. A particular arginine on cytochrome c6 (R66), long known to be vital in bacteria, stacks with neighboring rings on both cytochrome c6 and Photosystem I, forming a three-layered interaction that appears to help stabilize the docking position. This tight packing also pushes water away from the direct path between heme and chlorophyll, likely lowering resistance to electron flow and explaining why electron transfer occurs in just a few millionths of a second.
Testing the Critical Contact Points
To see which parts of cytochrome c6 matter most, the team introduced targeted changes to acidic and basic residues predicted to contact Photosystem I. When they neutralized certain negative charges near the PsaF “arm” or replaced the key arginine, the rate at which Photosystem I was re-reduced slowed markedly, and the crosslinking between the two proteins weakened. Some combinations of changes produced unexpected behavior, hinting that cytochrome c6 and Photosystem I may sometimes assemble in alternative, less efficient arrangements. Together, these tests confirm that both the PsaF-based electrostatic grip and the arginine-centered stacking are crucial for rapid and reliable electron delivery.
What This Means for Life’s Solar Machines
The work provides a detailed structural blueprint for how cytochrome c6 hands off electrons to Photosystem I in green algae, blending features seen in bacteria with those of more advanced plants. It shows how a small carrier protein and a large membrane complex have coevolved a finely tuned interface that balances strong binding, fast electron transfer, and quick release for the next cycle. By clarifying this ancient step in the photosynthetic relay, the study helps explain how organisms have optimized light-driven energy conversion over evolutionary time and offers design hints for future efforts to engineer or mimic nature’s solar energy systems.
Citation: Ogawa, Y., Mahapatra, G.P., Milrad, Y. et al. Cryo-EM structure of Chlamydomonas reinhardtii Photosystem I complexed with cytochrome c6. Nat Commun 17, 3031 (2026). https://doi.org/10.1038/s41467-026-70944-9
Keywords: photosynthesis, Photosystem I, cytochrome c6, electron transfer, cryo-EM