Clear Sky Science · en
Smallest acyclic tricationic molecule containing a Bis(phosphine)-stabilized low-valent triantimony-based Unit
Why this tiny charged puzzle matters
Chemists are fascinated by molecules that carry several units of positive charge, because crowding so much charge into a small space usually makes them fly apart. Understanding how to tame such unstable species helps reveal how chemical bonds really work and can open doors to new types of materials and catalysts. This study reports a record‑small, chain‑shaped molecule built from three antimony atoms that together carry three positive charges, along with an equally unusual negatively charged partner that helps hold the system together.
Building a fragile three-atom chain
Most known highly charged molecules based on main‑group elements form rings or larger clusters, which spread out their charge and avoid intense internal repulsion. The authors set out to create something more extreme: the smallest possible open chain of three bonded heavy atoms that still carries more than a single positive charge. They chose antimony, a heavier cousin of arsenic and phosphorus, because its large atoms can better accommodate extra charge. By carefully combining a pre‑made antimony fragment with a strongly pulling antimony salt at very low temperature, and wrapping the ends of the chain with a bulky “bis(phosphine)” grip, they succeeded in isolating a bright red compound containing a straight‑but‑bent three‑antimony unit with a total charge of plus three.
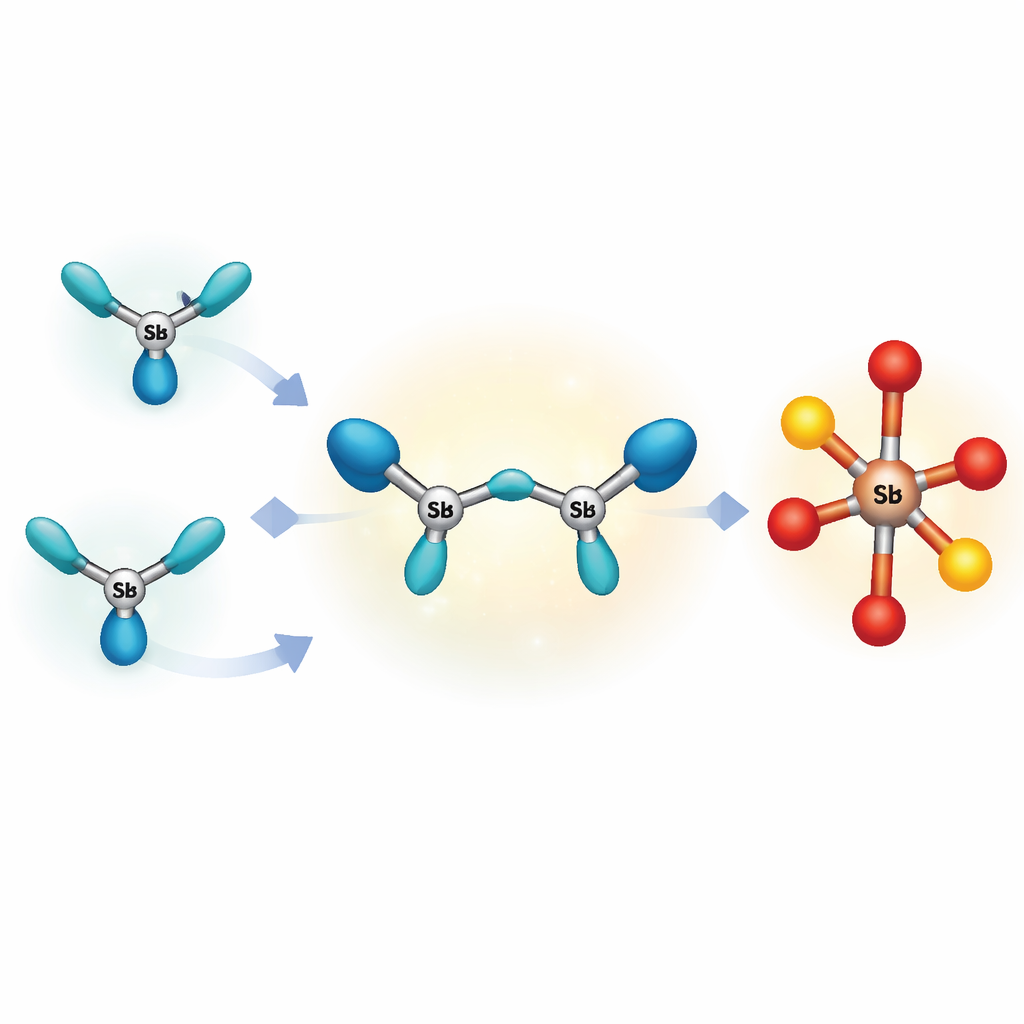
A strange partner on the negative side
To balance three positive charges, the crystal also contains a rarely seen anion, or negatively charged unit, based on antimony bonded to oxygen and four triflate groups. X‑ray measurements show an O–Sb–O spine with four additional oxygen‑rich arms, creating very polar antimony–oxygen links that are highly reactive. Because neither the positively charged chain nor this unusual negative partner is well protected by bulky surroundings, the entire salt is thermally fragile: even gentle warming causes it to decompose, metal antimony to precipitate, and a more stable product to appear. This behavior means the original triantimony chain exists only as a fleeting snapshot along the path to other compounds.
Looking inside the bonding
The team used quantum‑chemical calculations to peer into the electronic structure of the triantimony chain. They found that the three antimony atoms form a bent "W" arrangement supported by a weak three‑center, four‑electron bond along the chain. The central antimony atom carries two unused pairs of electrons, while each outer atom carries one. Charge analysis reveals a striking polarity: the ends of the chain and the phosphorus atoms of the supporting grips are relatively positive, whereas the central antimony atom is somewhat negative. This uneven distribution, together with the large size of antimony and the weak metal–metal bond, helps explain why the chain is so easy to break and so eager to react.
From fleeting chain to new antimony chemistry
Because the red triantimony species falls apart quickly, the researchers generated it in place for each test and then probed how it reacts. Small changes in conditions led to a range of new antimony compounds: swapping the oxygen in the counter‑anion for sulfur gave a sulfur‑bridged diantimony species, while adding simple chalcogen–chalcogen molecules (based on sulfur or selenium) produced antimony–chalcogen fragments by breaking those chalcogen–chalcogen bonds. Iodine‑containing reagents led to an iodide complex, and reacting the triantimony system with a cobalt carbonyl source yielded a striking cluster in which antimony atoms sit at the core of a triangle of cobalt centers bound to carbon monoxide ligands. Comparing these outcomes with reactions of a simpler antimony fragment showed that the highly charged chain and its reactive anion follow distinct chemical pathways.
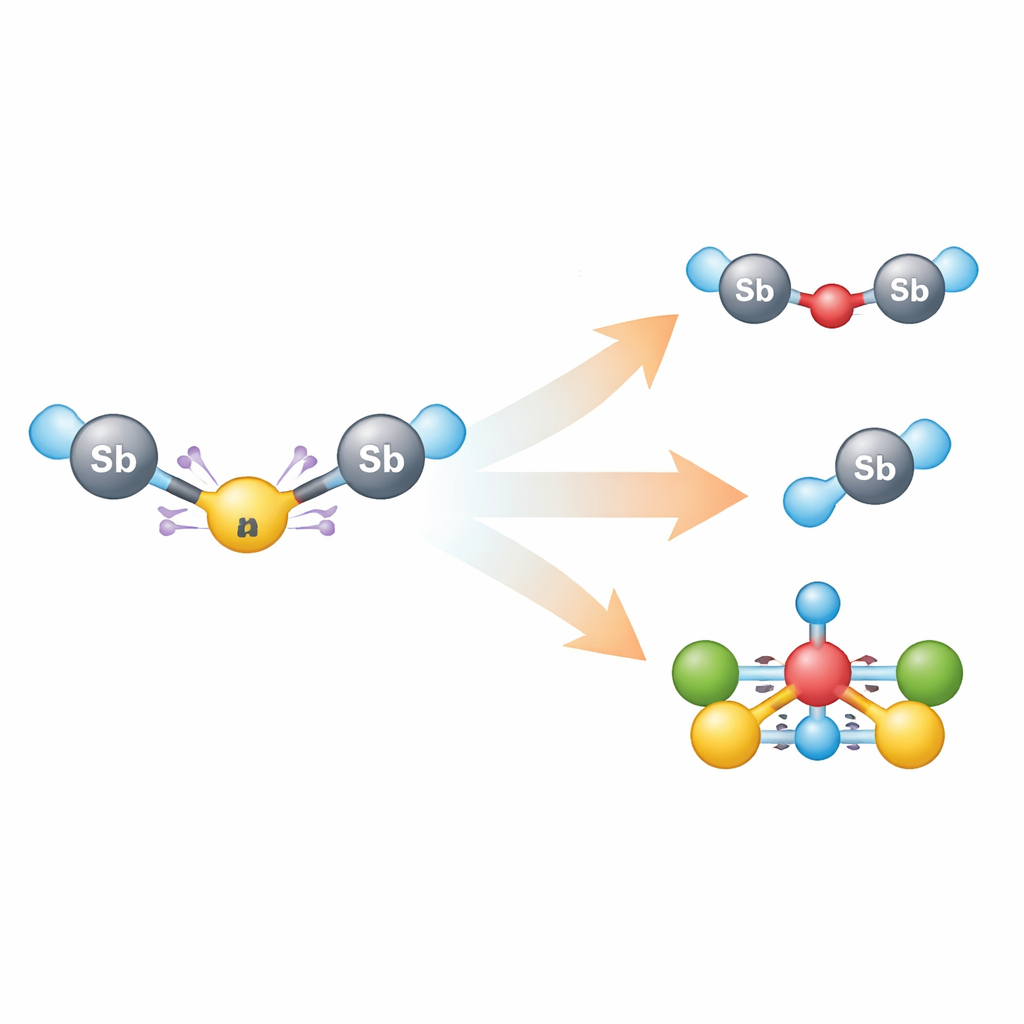
What the study shows in simple terms
In essence, the authors have managed to catch and characterize an exceptionally small and highly charged molecular chain that would normally be too unstable to study. By pairing a delicate triantimony cation with a reactive oxygen‑rich anion and stabilizing it just enough with phosphine grips, they reveal how heavy atoms can share electrons in unusual ways, how charge and bond weakness go hand in hand, and how such species can seed a family of new compounds, including mixed antimony–cobalt clusters. For non‑specialists, this work highlights how pushing molecules to their limits—here, packing three positive charges into a three‑atom line—uncovers new bonding patterns that may ultimately inspire fresh strategies in main‑group chemistry and materials design.
Citation: Mukherjee, N., Peerless, B., Nadurata, V.L. et al. Smallest acyclic tricationic molecule containing a Bis(phosphine)-stabilized low-valent triantimony-based Unit. Nat Commun 17, 2697 (2026). https://doi.org/10.1038/s41467-026-70910-5
Keywords: antimony chemistry, multicationic molecules, metal–metal bonding, main-group clusters, reactive intermediates