Clear Sky Science · en
Prediction of strong Cu(I)–He interaction at open metal sites enables isotope-selective helium adsorption
A New Way to Tame the Most Untouchable Gas
Helium is famous for being almost impossible to make react with anything, yet it is vital for MRI scanners, advanced electronics, and cutting‑edge physics. One rare form, helium‑3, is so scarce and valuable that the world is actively searching for better ways to separate it from ordinary helium‑4. This paper reveals that certain copper‑based materials can grab helium much more strongly than anyone expected, opening a path toward more practical helium‑3 recovery at temperatures that current technology struggles to reach.
Why Helium‑3 Matters So Much
Helium‑3 makes up only a few parts per million of natural helium, but its unusual quantum behavior under extreme cold gives it unique uses. It helps cool powerful magnets in medical scanners, enables sensitive leak detection in industry, and serves as a workhorse in low‑temperature physics experiments. It is also a candidate fuel for advanced fusion reactors, which could offer cleaner energy by producing charged particles rather than dangerous neutron radiation. Today, most helium‑3 still comes indirectly from the decay of tritium in nuclear stockpiles, and separating it from helium‑4 usually requires temperatures of just a few degrees above absolute zero. That combination of scarcity, demand, and costly processing makes any new separation method highly attractive.
Discovering an Unexpected Bond
Conventional wisdom says helium hardly sticks to anything because its electrons are tightly bound and difficult to distort. The authors challenge this view by examining simple clusters in which a copper ion in the +1 oxidation state, Cu(I), is surrounded by different companion atoms or molecules and then approached by a helium atom. Using high‑level quantum chemistry, they find that when copper is paired with suitable negatively charged partners such as fluoride or hydroxide, a helium atom can bind surprisingly strongly—up to about 19 kilojoules per mole, far more than the feeble attractions between typical noble‑gas pairs. Analysis of the electron density shows that helium becomes polarized and even donates a small fraction of an electron to copper, creating a bond that is partly electrostatic and faintly covalent in character. 
From Simple Clusters to Real Materials
Armed with this insight, the team searches for realistic materials that host undercoordinated Cu(I) sites—copper atoms that are bonded to only two or three neighbors and thus remain relatively exposed. They study molecular “crown ether” rings, fragments of zeolites (porous aluminosilicate minerals), and metal–organic frameworks (MOFs), which are crystalline networks of metal nodes and organic linkers. By combining an efficient but accurate quantum method with a tailored way to treat the quantum motion of helium nuclei, they estimate how strongly helium binds in each environment and how that binding differs between helium‑3 and helium‑4. In many cases, helium still sticks much more than expected, especially when the copper center is two‑fold coordinated and arranged in a bent geometry that leaves room for helium to approach closely.
Exploiting Subtle Quantum Differences
The key to isotope separation lies not only in how tightly helium binds, but in how quantum zero‑point motion differs between the lighter helium‑3 and the heavier helium‑4. Even at low temperatures, each atom vibrates in its binding pocket. Because helium‑3 is lighter, it has a higher zero‑point energy and explores a slightly larger region of space, which effectively weakens its binding compared with helium‑4. The authors compute these quantum effects carefully by mapping the energy landscape along the copper–helium bond and solving the corresponding Schrödinger equation numerically. They show that strong Cu–He interactions amplify the difference in zero‑point energies enough to create sizable preferences for one isotope over the other, quantified as separation factors well above those of today’s technologies at the same temperature.
Promising Copper Frameworks for Cooler but Practical Operation
Among the many materials tested, certain zeolite models and especially a MOF based on a copper–chloride building block (known from the crystal WOLRIZ) stand out. In this MOF, two‑fold coordinated Cu(I) sites with a bent arrangement bind helium with around 4 to 6 kilojoules per mole—strong for helium yet not so strong that the gas cannot be released again. At the boiling point of liquid hydrogen, 20 kelvin, the calculated separation factor for helium‑4 over helium‑3 approaches three, clearly surpassing current approaches that require even lower temperatures to achieve similar or smaller effects. The study suggests that further tuning of the copper environment, or exploring other metals with similar behavior, could yield even better candidates. 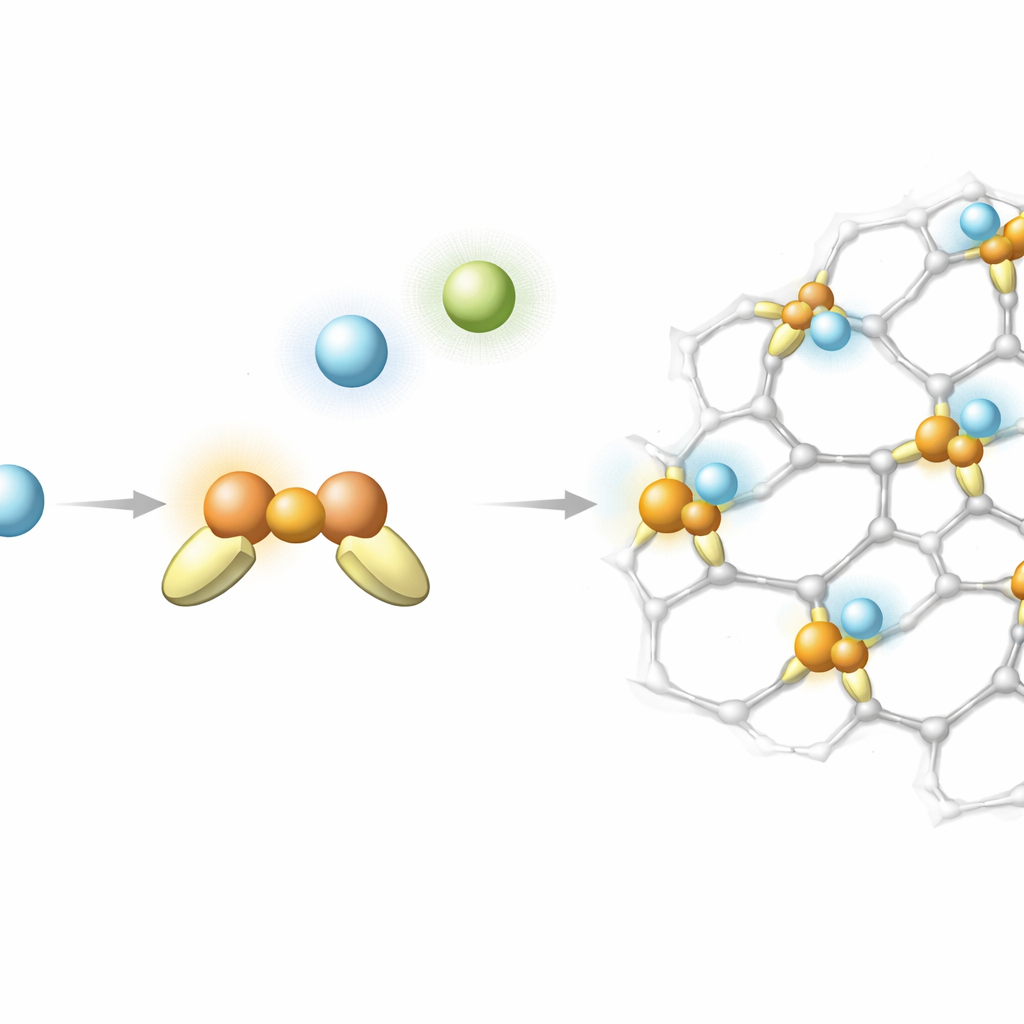
What This Means for Helium and Beyond
In everyday terms, the authors show that the “noblest” gas is not as aloof as it seems: when it encounters the right kind of exposed copper site, helium can form a surprisingly intimate partnership. This stronger‑than‑expected attraction naturally favors one isotope over the other because of their subtle quantum differences, potentially enabling more efficient helium‑3 harvesting at temperatures that are cold but technologically manageable. Beyond its practical implications, the work overturns long‑held assumptions about helium’s passivity and invites chemists to think creatively about doing “chemistry” with a gas previously regarded as almost completely untouchable.
Citation: Dongmo, E.G., Das, S., Moncada, F. et al. Prediction of strong Cu(I)–He interaction at open metal sites enables isotope-selective helium adsorption. Nat Commun 17, 2952 (2026). https://doi.org/10.1038/s41467-026-70901-6
Keywords: helium isotope separation, copper adsorption sites, metal-organic frameworks, nuclear quantum effects, porous materials