Clear Sky Science · en
A cyanide-free route towards the electrosynthesis of nitriles
Cleaning Up a Hidden Corner of Chemistry
Nitriles are quiet workhorses of modern life, found in medicines, plastics, dyes, and farm chemicals. Yet making them at industrial scale often spews out carbon dioxide, nitrogen oxides, and even toxic cyanide. This study introduces a way to make nitriles using electricity and a modest catalyst instead of harsh heat or poisonous reagents, pointing toward cleaner factories that could even reuse their own waste gases.

Why Today’s Routes Cause Trouble
Industry usually makes nitriles by heating simple chemicals with ammonia and oxygen to several hundred degrees Celsius. Those fiery conditions are hard to control, so part of the feedstock burns all the way to carbon dioxide and nitrogen oxides. Alternative routes swap heat for chemistry that uses large amounts of cyanide salts, which are highly toxic and difficult to handle safely at scale. With millions of tons of cyanide used every year, even small inefficiencies leave behind dangerous waste. A more sustainable approach would run at room temperature, plug into renewable electricity, and avoid cyanide entirely while still producing the same valuable products.
Turning Electricity into a Chemical Tool
The authors designed an electrochemical system that does just that: it uses electrical energy to drive the reaction that turns an aldehyde (here, benzaldehyde) and ammonia into the nitrile benzonitrile. The heart of the system is a carefully structured catalyst grown on copper foam. Tiny clusters of cobalt oxide sit on the tips of copper oxide nanorods, creating a large, active surface. When a modest positive voltage is applied in a liquid mixture of benzaldehyde, ammonia, water, and acetonitrile, the electrode converts nearly every electron into the desired nitrile, with a Faradaic efficiency close to 99% and almost perfect selectivity. The catalyst keeps working for at least 100 hours with little loss in performance, suggesting it could be robust enough for industrial use.
Following the Atoms During Reaction
To understand how this clean conversion works, the team watched the reaction in real time using several spectroscopic techniques. They found that benzaldehyde and ammonia first combine to form a short-lived intermediate that resembles an imine—essentially, the carbon-oxygen double bond is replaced by a carbon-nitrogen double bond. This species attaches to copper sites on the catalyst surface, where hydrogen atoms are gradually stripped away under the applied voltage, leaving behind the carbon–nitrogen triple bond characteristic of nitriles. Computer simulations support this picture, showing that copper in a higher oxidation state is especially good at pulling electrons from the intermediate, while nearby cobalt oxide clusters increase the number of active sites and subtly tune the copper’s electronic character to favor this dehydrogenation step.
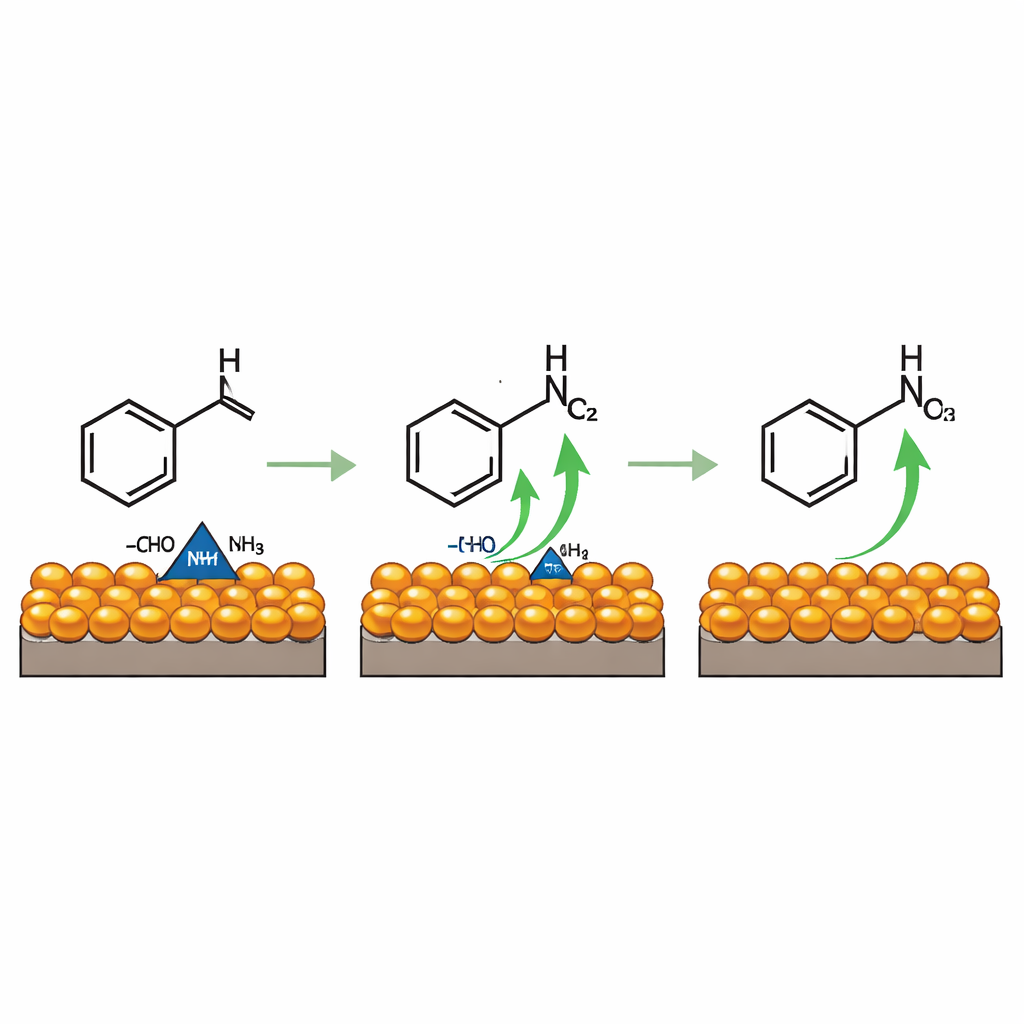
From Simple Aromatics to Real-World Products
Once established for benzaldehyde, the same approach proved versatile. A range of aromatic and aliphatic aldehydes, including ones bearing chlorine, bromine, or cyano groups, were smoothly converted into their nitriles with efficiencies between about 78% and 97%. The system can even be plugged into multi-step routes that start from alcohols or simple hydrocarbons like toluene, first oxidizing them to aldehydes and then pushing on to the nitrile. Notably, some of these starting materials can themselves be made from captured carbon dioxide, hinting at a fully electrified chain from waste gas to high-value chemicals.
Recycling Polluted Air into Useful Products
An especially appealing twist is that the ammonia needed for the reaction does not have to come from the traditional, energy-intensive Haber–Bosch process. The authors show that the anodic production of nitriles can be paired with a cathodic process that converts nitrogen oxides—major air pollutants—into ammonia in the same overall setup. At current densities up to 200 milliamps per square centimeter, the amount of ammonia formed at the cathode closely matches the amount consumed at the anode. In principle, this means a plant could turn its own nitrogen oxide emissions into the nitrogen source for cleaner nitrile manufacture.
A Safer Pathway for Everyday Molecules
In plain terms, this work replaces hot, dirty, and sometimes poisonous chemistry with a cooler, electrically powered method that makes the same useful nitriles with far fewer unwanted side products. By combining a smartly engineered catalyst with careful control of reaction conditions, the authors achieve high yields and long lifetimes while opening the door to using waste gases as ingredients. If scaled up, such electrochemical routes could help decarbonize part of the chemical industry, shrinking the environmental footprint of many everyday products without changing what those products are.
Citation: Xian, J., Wang, L., Mi, Z. et al. A cyanide-free route towards the electrosynthesis of nitriles. Nat Commun 17, 4095 (2026). https://doi.org/10.1038/s41467-026-70732-5
Keywords: electrosynthesis, green chemistry, nitriles, ammonia, waste gas utilization