Clear Sky Science · en
Compartmentalized cytoplasmic tradewinds direct soluble proteins
How Cells Send Supplies to Their Front Line
Whenever a cell crawls across a surface – to heal a wound, wire up the nervous system, or chase down an infection – it must rapidly deliver the right proteins to its front edge. This paper uncovers a hidden "tradewind" inside the cytoplasm: a gentle but organized internal fluid flow that sweeps many different soluble proteins toward the cell’s leading edge, helping the cell reshape itself and move with surprising speed and precision.
A Hidden Shipping Problem Inside Cells
Inside a cell, countless proteins drift through a crowded liquid interior. Some ride in membrane-bound packages along molecular tracks, but many important proteins float freely in solution. Classic textbook pictures suggest these free molecules simply diffuse – wandering randomly like perfume in air. That picture raised a puzzle: diffusion is slow and directionless, yet moving cells somehow keep a steady stream of building blocks, such as actin monomers, ready at their front edge where new structures are constantly assembled.
Finding a Forward Tradewind
The authors tackled this puzzle by inventing ways to watch individual soluble proteins move inside living cells with high precision. They used light to temporarily darken or activate fluorescently tagged proteins, then followed how the newly marked molecules spread. In several cell types, they saw something diffusion alone could not explain: after bleaching actin at the rear of the flat leading region, a sharp dark line appeared near the front within seconds, showing that non-fluorescent actin had been carried forward far faster than random motion would allow. When they inhibited myosin II – a motor protein that can generate contractile forces – this fast forward movement slowed dramatically, indicating that active contraction helps drive a forward flow of cytoplasm that boosts transport.
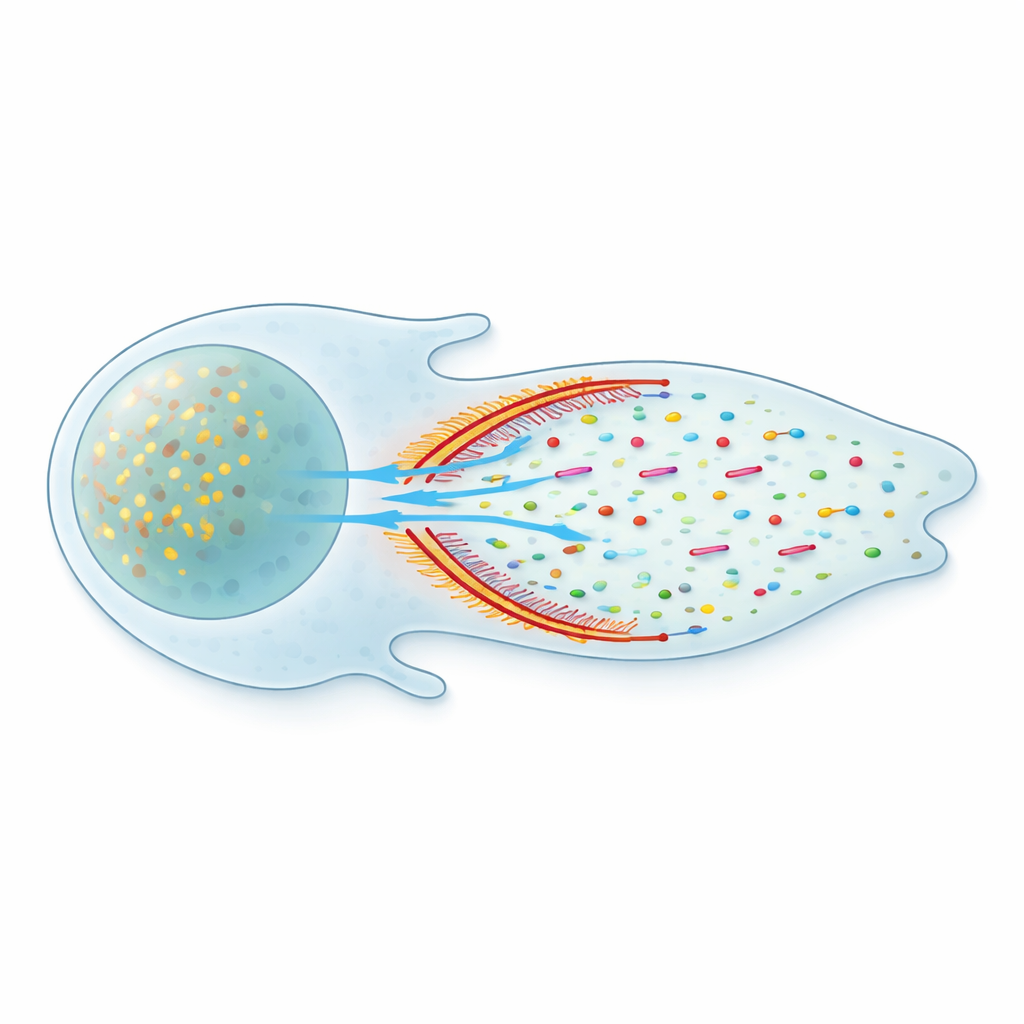
A Separate Front Compartment With a Soft Wall
Closer inspection revealed that this flow is not uniform throughout the cell. Instead, the front of the cell forms a distinct fluid compartment, separated from the rest of the cytoplasm by a curved band of densely packed actin and myosin. Using super-resolution microscopy, the team showed that these actin–myosin "arcs" form a vertical barrier that spans the thickness of the cell. Fluorescent tracers activated on one side of this barrier tended to stay in that region; crossing into the other side was measurably delayed. Yet the barrier is not completely sealed – proteins can leak through – so the authors describe it as a leaky condensate that shapes, but does not halt, molecular traffic.
Non-Specific Flow That Moves Many Cargoes
Within the front compartment, the cytoplasm behaves like a slow, directed stream. The researchers saw that not only actin monomers but also actin-binding proteins and adhesion components – including Arp3, vinculin, and paxillin – drifted preferentially toward the edge. Even inert fluorescent probes with no special binding partners were carried forward the same way. Measurements of molecular motion showed that simple diffusion was similar in the front and body of the cell, but an added advective component – fluid flow – was much stronger at the front. This means the flow is largely non-specific: it boosts the delivery of any soluble protein small enough to move through the actin mesh, ensuring that a broad toolkit of molecules reaches the edge faster than diffusion alone could manage.
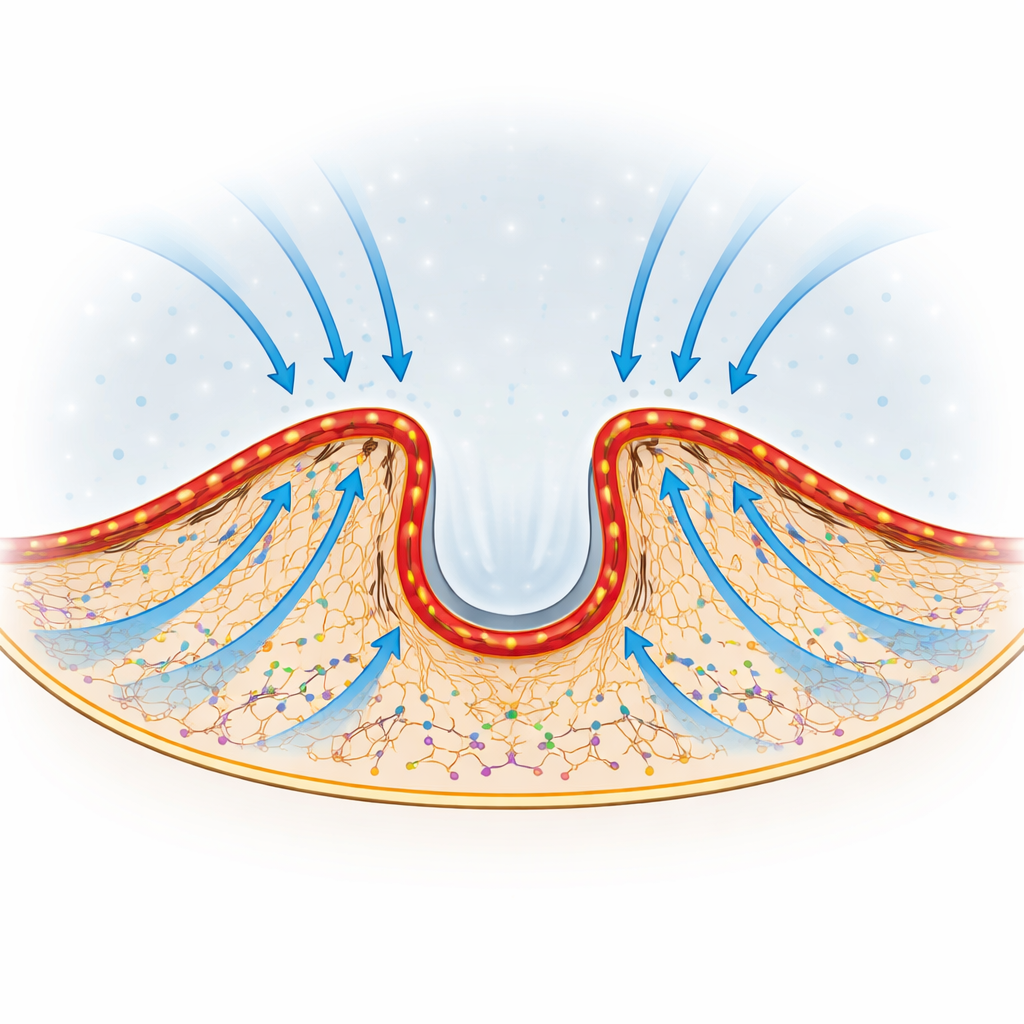
Steering Flow to Where the Edge Advances
Perhaps most striking, the direction of this internal tradewind can be tuned. As cells extend or retract different parts of their edge, the curvature and position of the actin–myosin arcs shift. The authors observed that newly activated actin preferentially streams toward the regions of the edge that are actively protruding. When they altered or flattened the arcs by inhibiting myosin or by cutting an individual arc with a focused laser, local edge advancement faltered only in the area in front of the disrupted structure. This suggests that the shape of the barrier acts like a set of adjustable baffles, redirecting flow and protein delivery toward whichever region of the edge needs to push forward.
Why This Matters for Cell Shape and Movement
By revealing a specialized front compartment and a myosin-driven internal tradewind, this work reshapes how we think about the moving cell. Rather than relying solely on slow, aimless diffusion, cells create a pseudo-organelle at their leading edge: a flexible zone bounded by a protein condensate that both concentrates key molecules and channels fluid flow. This arrangement lets cells quickly redistribute soluble proteins where they are most needed, tightly linking local protein supply to changes in cell shape, adhesion, and migration. In essence, the cell’s front is supplied by a constantly adjusting internal current, allowing rapid, energy-efficient responses to the outside world.
Citation: Galbraith, C.G., English, B.P., Boehm, U. et al. Compartmentalized cytoplasmic tradewinds direct soluble proteins. Nat Commun 17, 2589 (2026). https://doi.org/10.1038/s41467-026-70688-6
Keywords: cell migration, actin cytoskeleton, intracellular transport, protein localization, cytoplasmic flow