Clear Sky Science · en
Calcium-mediated calreticulin-IRE1α interaction drives dynamic fluctuation of IRE1α activity under chronic endoplasmic reticulum stress
How Cells Surf the Waves of Internal Stress
Inside every cell, a sprawling network of folded membranes called the endoplasmic reticulum acts like a factory floor for building and processing proteins. When this factory is overloaded, the cell faces a dilemma: repair and adapt, or shut down and die. This study reveals that one key stress sensor inside this factory, a protein called IRE1α, does not simply switch on or off. Instead, its activity rises, falls, and rises again in waves—carefully tuned by calcium ions and a helper protein named calreticulin. Understanding this subtle control could shed light on diseases where long‑term cellular stress plays a central role, such as diabetes, cancer, and neurodegeneration.
When Protein Factories Get Overloaded
In healthy cells, fresh proteins are fed into the endoplasmic reticulum, where they are folded into precise shapes. If too many unfolded proteins pile up, the cell experiences what scientists call “ER stress.” To cope, cells launch a protective program known as the unfolded protein response. Three sentinels on the ER membrane detect trouble and relay signals to the rest of the cell. One of these sentinels, IRE1α, is especially important because it helps boost the cell’s ability to fold and process proteins. But if stress is too strong or too long‑lasting, the very same system can flip and help push the cell toward suicide. How cells manage this balance under ongoing, non‑lethal stress has been a major open question.
A Stress Signal That Pulses Instead of Staying Flat
The researchers exposed human cells to chemicals that cause different levels of ER stress—mild, intermediate, and severe—and tracked what happened over two days. They measured how strongly IRE1α was turned on by looking at its chemical modification and at the processing of a partner message molecule that it controls. Under moderate and strong stress, they found a striking “up‑down‑up” pattern: IRE1α became strongly active within several hours, quieted down later, and then surged again many hours afterward. This was not a one‑time event or a quirk of a single cell type; similar waves appeared in several kinds of human and mouse cells. In contrast, another stress branch that favors cell death kicked back in only under the harshest, lethal conditions. These observations suggest that the cell uses a dynamic, pulsing strategy to stay alive under chronic strain.
The Tug‑of‑War Between Two Helpers
To understand what controls these waves, the team examined which proteins physically latch onto IRE1α during long‑term stress. They discovered a see‑saw interaction between two ER helpers. One, BIP, is well known as a brake that keeps IRE1α in check. The other, calreticulin, usually helps fold certain proteins but here turned out to be an unexpected activator of IRE1α. Under sustained stress, calreticulin’s grip on IRE1α rose and fell in step with IRE1α activity, while BIP’s binding showed the opposite pattern. Experiments with purified proteins and engineered cells confirmed that calreticulin and BIP compete directly for the same sensor: when one binds more strongly, the other is pushed away. Cells lacking calreticulin failed to mount the late “second wave” of IRE1α activity and were more prone to die, highlighting calreticulin’s role as a pro‑survival partner.
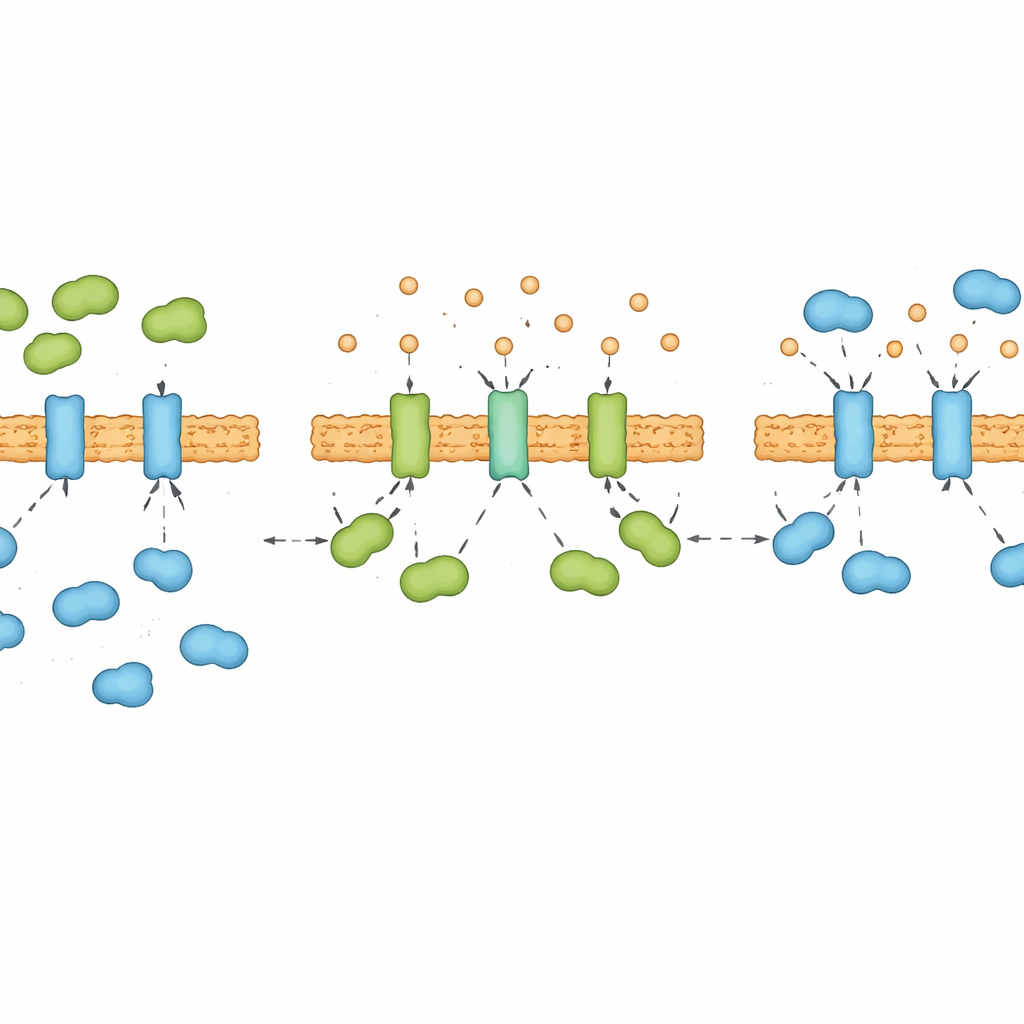
Calcium as a Hidden Dial for Stress Signaling
Calreticulin is also a major storage and binding partner for calcium inside the ER, and the researchers found that this connection is crucial. When calcium was stripped from calreticulin in test‑tube experiments, the protein changed shape and bound IRE1α much more tightly. Briefly lowering calcium levels inside the ER of living cells rapidly strengthened calreticulin’s contact with IRE1α, even before unfolded proteins had time to accumulate. Over many hours of chronic stress, ER calcium levels themselves followed a “down‑up‑down” rhythm that mirrored the alternating dominance of calreticulin and BIP on IRE1α. In essence, calcium acts as a hidden dial that reshapes calreticulin and decides whether it, or BIP, wins the tug‑of‑war for control of the stress sensor.
Daily Rhythms and the Fine Line Between Survival and Death
Strikingly, the same dance between IRE1α, calreticulin, BIP, and calcium appears in normal mouse liver, even without artificial stress. There, IRE1α activity and its partnership with calreticulin wax and wane over the day, suggesting that this mechanism helps healthy tissues adjust to natural cycles in workload, such as daily swings in metabolism. Taken together, the work paints IRE1α not as a simple on/off switch but as a finely tuned oscillator, guided by calcium‑shaped calreticulin and its rivalry with BIP. For a layperson, the key message is that cells facing long‑term strain do not just choose life or death once; they revisit that decision repeatedly, using internal chemical waves to buy time, adapt, and, when necessary, surrender.
Citation: Cao, J., Zhao, X., Xu, Y. et al. Calcium-mediated calreticulin-IRE1α interaction drives dynamic fluctuation of IRE1α activity under chronic endoplasmic reticulum stress. Nat Commun 17, 4043 (2026). https://doi.org/10.1038/s41467-026-70679-7
Keywords: endoplasmic reticulum stress, unfolded protein response, calcium signaling, calreticulin, cell survival