Clear Sky Science · en
Behavioral screening defines the molecular Parkinsonism-related subgroups in Drosophila
Why tiny flies matter for a big brain disease
Parkinson’s disease is best known for its shaking and stiffness, but beneath those symptoms lies a tangle of different biological causes. This study uses fruit flies to tackle a crucial question: do the many genetic forms of Parkinsonism actually boil down to just a few core problem types inside cells? If so, doctors might one day match patients to treatments based on the specific cellular trouble their neurons face, rather than relying on a one-size-fits-all approach.
Many genes, one shared movement problem
The researchers started by building a collection of 24 fruit fly strains, each carrying a defect in a different gene known to cause or increase the risk of Parkinsonism in people. These flies were carefully engineered so that only the chosen gene differed between them, allowing for clean comparisons. As the flies aged, most developed movement problems in a standard climbing test, echoing the gradual loss of motor control in human patients. These problems were tied to damage in dopamine-producing nerve cells in the fly brain, and in many cases could be eased by feeding the flies L-Dopa, the same drug used to treat human Parkinson’s symptoms.
Beyond movement: sleep, activity, and hidden patterns
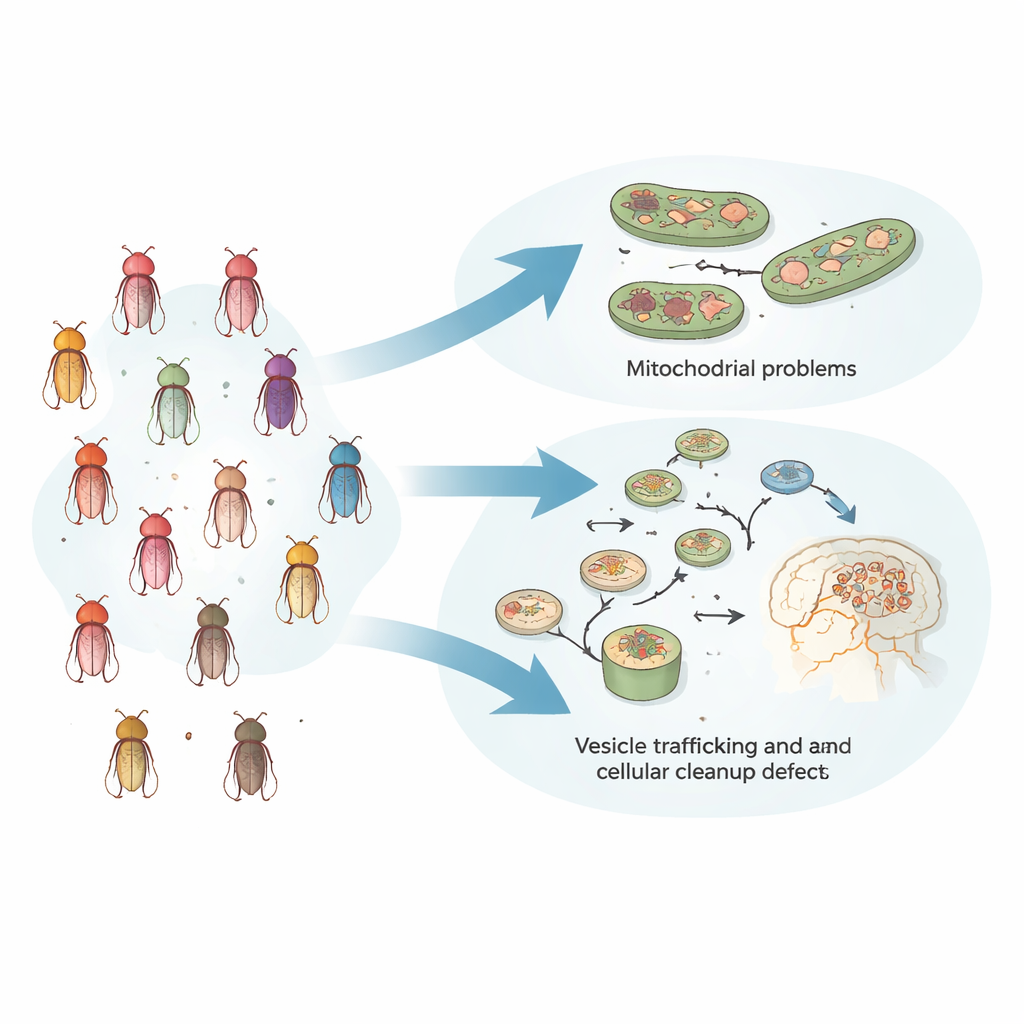
Parkinsonism also affects sleep, alertness, and other daily rhythms long before severe movement problems appear. To capture this early stage, the team placed young flies—before strong motor decline—into automated monitoring devices that tracked their activity and sleep over several days. Using machine learning to sift through features like how long flies slept, when they fell asleep, and how often they woke, the researchers uncovered clear behavioral fingerprints for each genetic model. When these patterns were clustered, the 24 fly lines consistently fell into two broad groups, each with its own characteristic sleep and activity profile, suggesting that very different genes can drive similar whole-animal behavior by disturbing related internal processes.
Two main problem types inside cells
To see whether these behavior-based groups reflected deeper biology, the team turned to two independent approaches. First, they measured how pairs of gene mutations interacted in the fly visual system using a sensitive electrical readout from the eye. Gene pairs that strongly worsened or unexpectedly improved each other’s defects tended to come from the same behavior-defined subgroup, indicating that they operate in shared or tightly linked pathways. Second, the known functions of the genes in each group told a clear story: one group centered on mitochondria, the cell’s energy factories, while the other revolved around vesicle trafficking, recycling of cellular components, and protein cleanup systems. Importantly, where and how strongly the genes were expressed in the body did not explain the grouping—what mattered was how they functionally interacted.
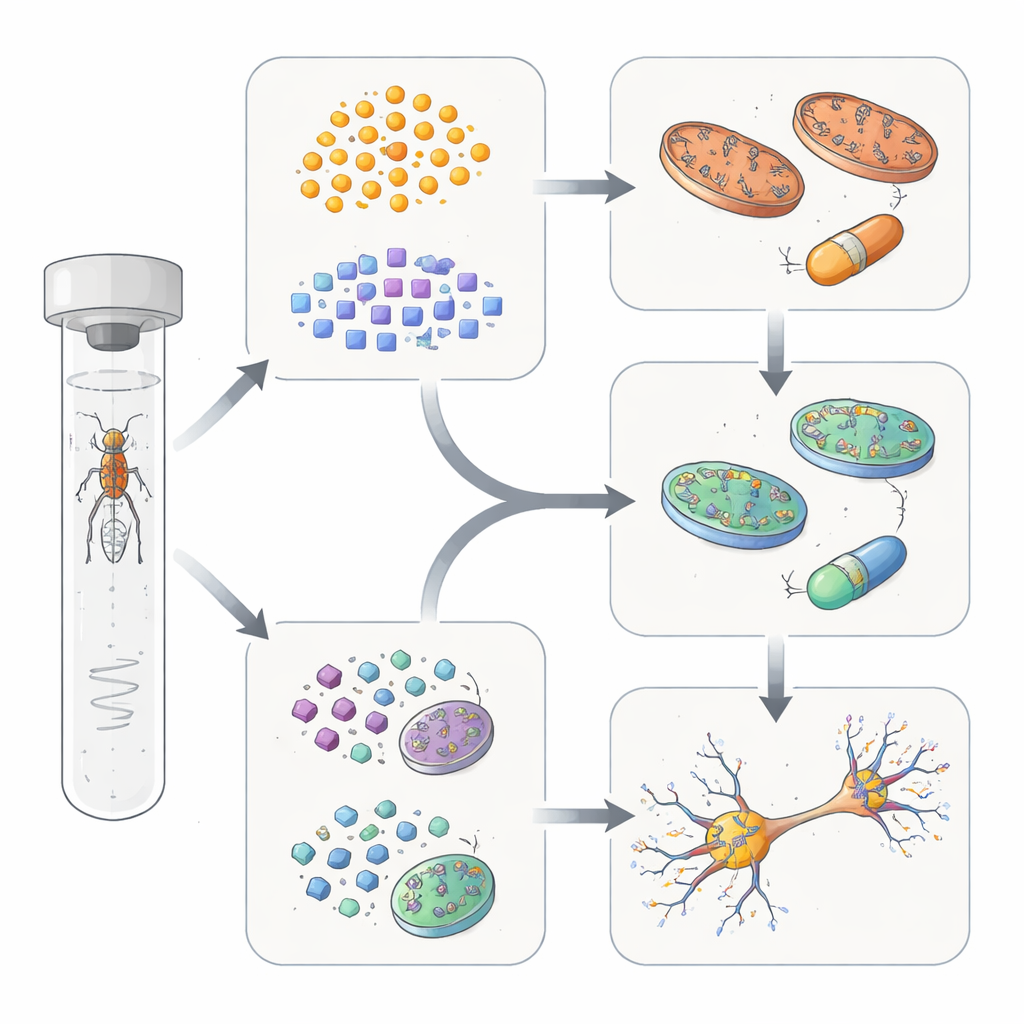
Testing targeted fixes in the fly brain
The authors then asked whether these subgroup differences could guide treatment. In flies whose genes fell into the mitochondrial group, feeding Coenzyme Q10—a molecule that supports energy production in mitochondria—improved both the health of dopamine connections and motor performance. In contrast, these same measures did not improve in flies from the trafficking and cleanup group. Instead, flies from that second group responded better to a compound known to stabilize a protein complex involved in cellular recycling, which restored dopamine nerve endings and movement without benefiting the mitochondrial group. Some genes in a third, smaller subgroup showed mixed responses, hinting that certain disease forms sit at the crossroads of these pathways.
What this means for people with Parkinsonism
Taken together, the work suggests that many inherited forms of Parkinsonism can be sorted into two major cellular problem types: one dominated by faulty energy production in mitochondria, and another by disturbed transport and disposal of cellular cargo. Although this study was done in fruit flies, the genes and pathways involved closely mirror those implicated in human disease. The findings support a future in which patients might be classified not only by symptoms, but also by the underlying pathway driving their disease—and in which drugs like Coenzyme Q10 or recycling-boosting compounds could be tested specifically in the patients most likely to benefit, rather than in broad, mixed populations where their true value may be hidden.
Citation: Kaempf, N., Valadas, J.S., Robberechts, P. et al. Behavioral screening defines the molecular Parkinsonism-related subgroups in Drosophila. Nat Commun 17, 3761 (2026). https://doi.org/10.1038/s41467-026-70303-8
Keywords: Parkinson’s disease, Drosophila models, mitochondrial dysfunction, vesicle trafficking, personalized neurology