Clear Sky Science · en
Targeting NAT10 alleviates colonic senescence and elderly-onset colitis by disrupting N4-acetylation of DYRK1A
Why Aging Guts Matter
As people live longer, more of us face digestive troubles that used to be rare in old age. One such problem is ulcerative colitis that first appears in seniors, bringing pain, bleeding, and an increased risk of colon cancer. This study asks a simple but powerful question: can we slow or even reverse age-related damage in the lining of the colon by targeting a single molecular "switch," and in doing so make elderly-onset colitis less severe?
Aging Cells at the Colon’s Front Line
The inner surface of the colon is covered by a single layer of epithelial cells that renew themselves constantly, forming a barrier between our body and the teeming world of gut microbes. With age, this barrier weakens: the mucus layer thins, stem cells lose vigor, and many cells enter a state called senescence, where they stop dividing but refuse to die. These senescent cells leak inflammatory substances that make the intestine more fragile and more prone to diseases like ulcerative colitis, especially in older adults whose treatment options are limited by other health issues.
A Chemical Mark on RNA as a Hidden Driver
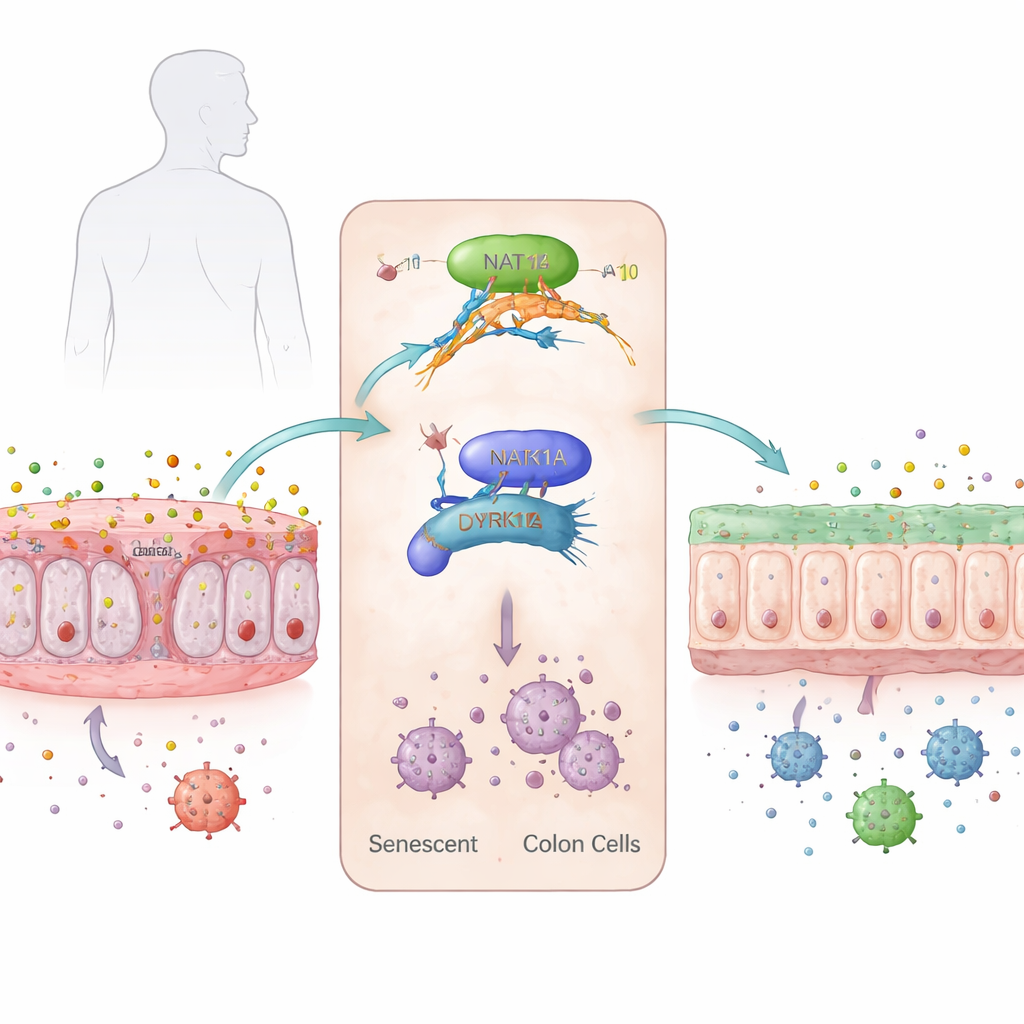
The researchers focused on a subtle chemical decoration on RNA called N4-acetylcytidine, or ac4C, which can make certain messages inside cells more stable and easier to translate into protein. They discovered that this mark, and its main "writer" enzyme NAT10, are increased in aged and artificially aged human colon cells. When cells were pushed into senescence in the lab, levels of ac4C rose, NAT10 became more abundant, and classic aging markers and inflammatory genes were switched on. In mouse colons, NAT10 levels climbed steadily from youth to old age, matching an increase in inflammatory signals and signs of DNA damage.
A Single Pathway Linking Marked RNA to Old Cells
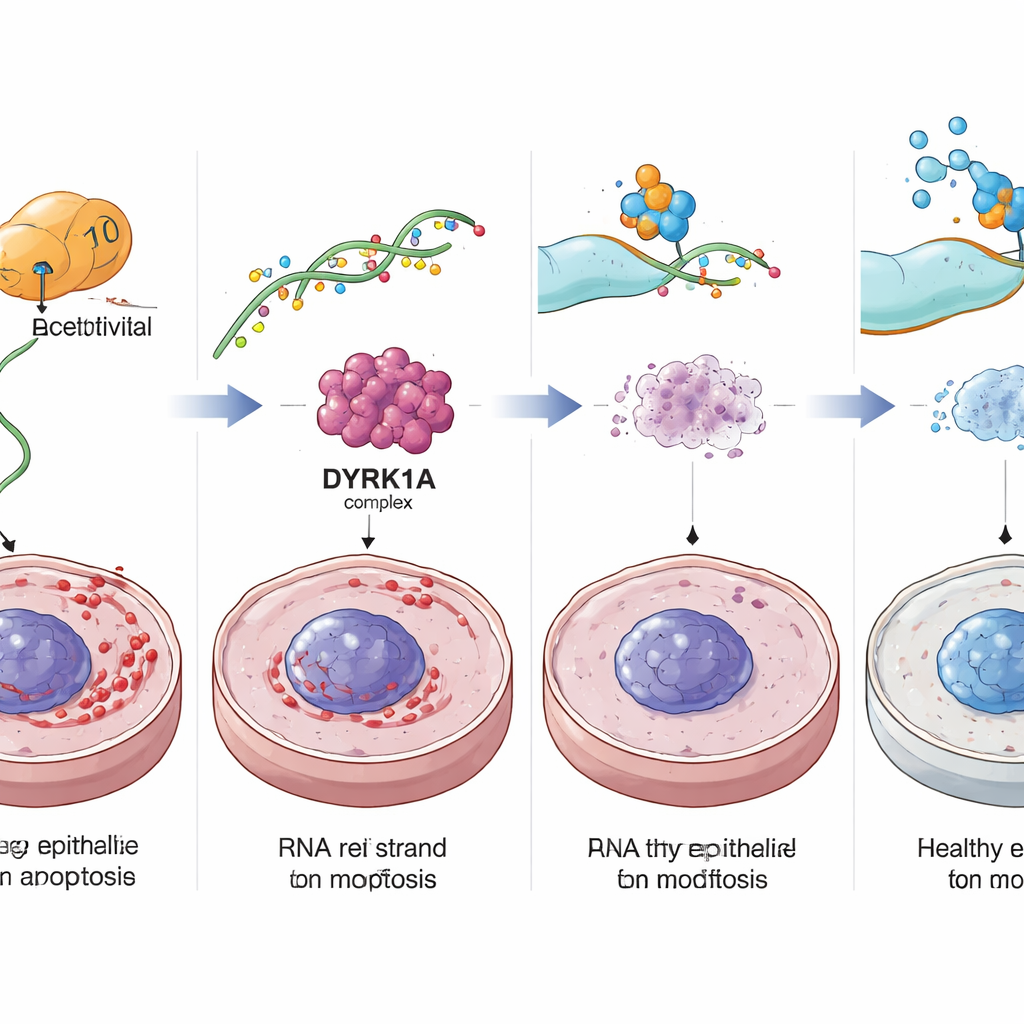
To work out how this RNA mark drives aging, the team scanned the entire set of RNA molecules in colon cells and asked which ones gained ac4C as cells became senescent and lost it when NAT10 was reduced. One clear standout was DYRK1A, a protein kinase that helps control the cell cycle and DNA damage responses. In senescent colon cells, DYRK1A’s RNA was heavily decorated with ac4C along its coding region, making the message more stable and more efficiently translated into protein. NAT10 physically bound to DYRK1A RNA, and blocking NAT10 caused the DYRK1A message to decay faster and be used less, lowering DYRK1A protein levels. Restoring DYRK1A in NAT10-depleted cells brought back the senescent, inflammatory state, marking it as a key downstream driver.
Rejuvenating the Aging Colon in Mice
The authors then tested whether dialing down this NAT10–DYRK1A pathway could actually rejuvenate the colon in living animals. Mice that carried only one working copy of the Nat10 gene, or aged mice treated with a NAT10-blocking drug called Remodelin, showed fewer senescent cells, more dividing stem-like cells, thicker mucosal layers, and improved barrier proteins in the colon. Tiny colon organoids grown from these mice formed more budding structures and resisted stress better, suggesting stronger regenerative capacity. When aged mice were given a chemical that induces colitis, reducing Nat10 or inhibiting DYRK1A with the drug Harmine led to less weight loss, milder colon inflammation, better healing of ulcers, and lower DNA damage markers.
Signals from Human Patients
To see if this mechanism matters in people, the team examined colon biopsy samples from healthy young adults, healthy elderly individuals, and patients with ulcerative colitis. Both NAT10 and DYRK1A were more abundant in the colons of older individuals, and their levels were even higher in elderly patients with ulcerative colitis. Moreover, the two proteins tended to rise together, and patients with more severe disease activity had the highest levels. These observations suggest that the NAT10–DYRK1A axis is not just a quirk of mouse models but is also active in human aging and disease.
What This Could Mean for Future Treatments
Taken together, the study paints a picture in which an RNA-modifying enzyme, NAT10, helps push colon cells into a harmful, long-lived senescent state by stabilizing DYRK1A RNA. In aging colons and in elderly-onset ulcerative colitis, this pathway appears to be overactive, weakening the barrier and amplifying inflammation. By turning down NAT10 or DYRK1A, the researchers were able to rejuvenate colon tissue and soften colitis in aged mice. For lay readers, the key message is that a reversible chemical mark on RNA may be a new handle for therapies that protect the aging gut and improve outcomes for older patients with inflammatory bowel disease.
Citation: Chen, J., Xue, M., Mi, S. et al. Targeting NAT10 alleviates colonic senescence and elderly-onset colitis by disrupting N4-acetylation of DYRK1A. Nat Commun 17, 3311 (2026). https://doi.org/10.1038/s41467-026-70220-w
Keywords: intestinal aging, ulcerative colitis, RNA modification, NAT10, cellular senescence