Clear Sky Science · en
Bioengineered ROS-tolerant probiotic reshapes gut microbiota-host axis to ameliorate type 2 diabetes in male mice
Reimagining Friendly Bacteria for Blood Sugar Health
Type 2 diabetes is usually blamed on sugar, fat, and lack of exercise, but deep inside the gut another player matters just as much: trillions of microbes that help digest food and talk to our metabolism. This study explores whether we can deliberately re‑engineer one familiar probiotic strain so it not only survives in the harsh, inflamed intestines of diabetes, but actively helps restore a healthy microbiome and improve blood sugar control—at least in mice.
A Tougher Gut Ally for a Hostile Environment
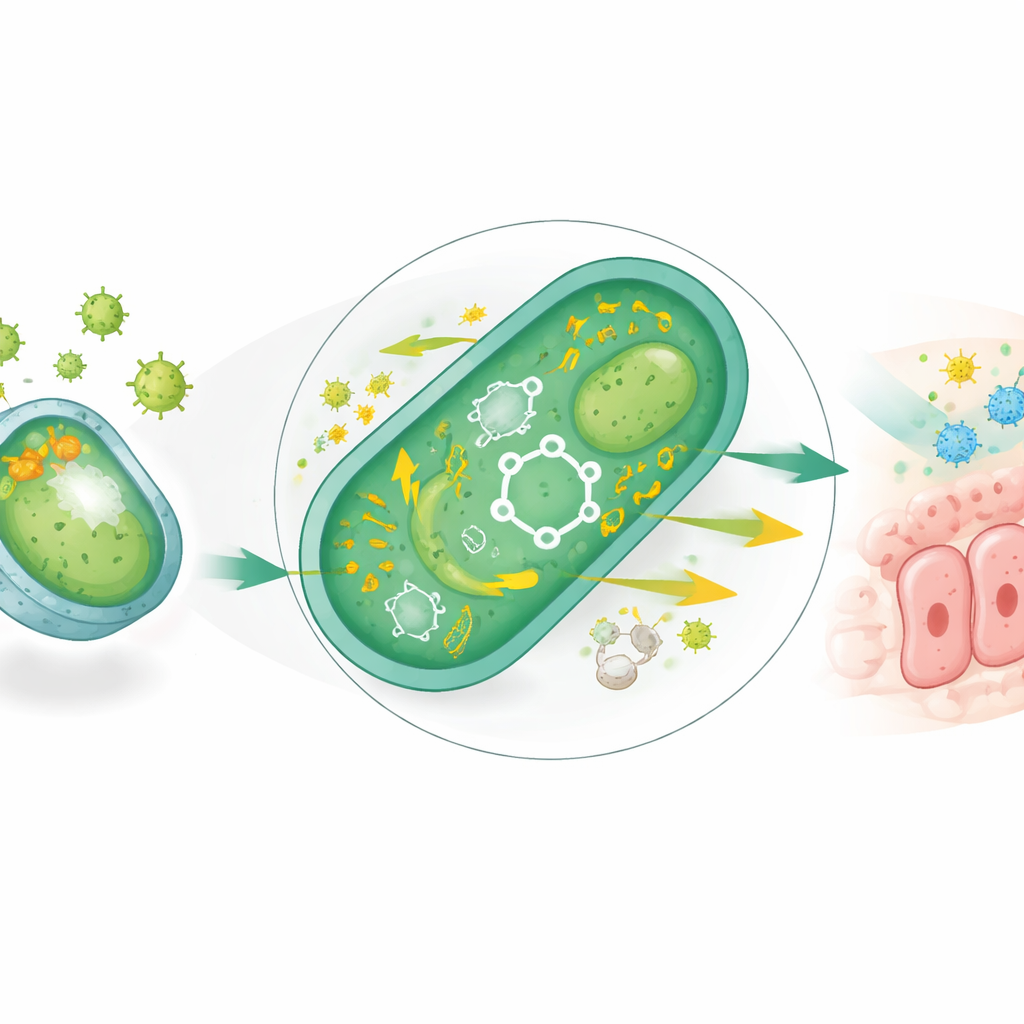
The researchers started with Escherichia coli Nissle 1917, a well‑studied probiotic that can calm gut inflammation but struggles in the highly oxidative, chemically stressful intestines of obesity and type 2 diabetes. To toughen it up, they repeatedly exposed the bacteria to increasing doses of hydrogen peroxide, a reactive oxygen molecule that mimics the chronic oxidative stress found in a diabetic gut. Over ten rounds of selection, they obtained a new strain, dubbed REcN, that could tolerate much higher levels of oxidative stress. Genetic and biochemical tests showed that REcN had switched on whole networks of antioxidant defenses, including enzymes that detoxify reactive molecules and a boosted system for handling cellular energy and redox balance.
How the Engineered Microbe Handles Stress and Food
Digging deeper, the team found that REcN had rewired its metabolism. It more efficiently burned fats and carbohydrates through core energy pathways and showed stronger activity of key enzymes that manage electrons and reduce the formation of damaging by‑products. The strain also produced more hydrogen sulfide in a controlled way, which in this context acted as an additional chemical shield against oxidative damage. In cell‑culture experiments, fluid from REcN cultures reduced fat droplets in fat cells and dampened the activity of digestive enzymes that rapidly break down starches and sugars, hinting that the microbe could slow glucose surges after meals and encourage the body to burn fat rather than store it.
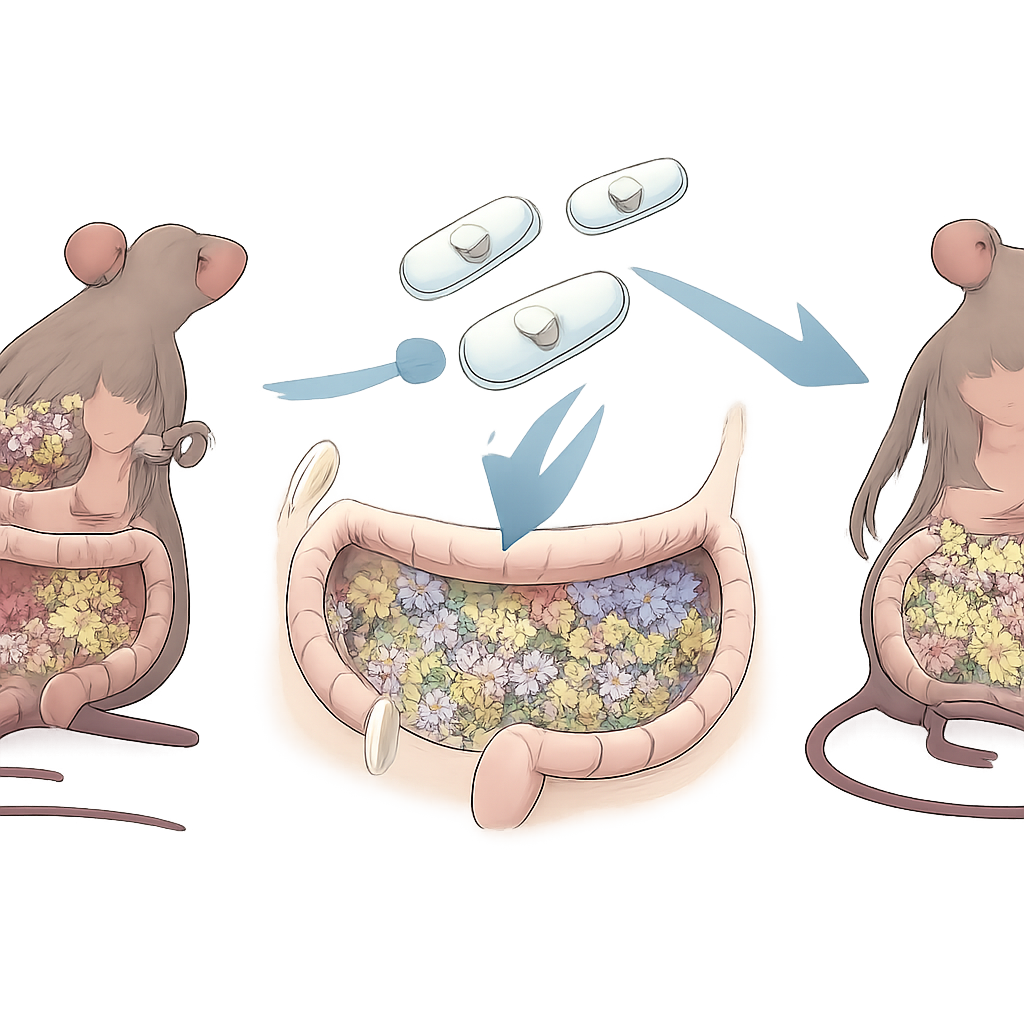
Armoring and Feeding the Probiotic for the Journey
To make sure this upgraded bacterium survived the trip through the stomach and small intestine, the scientists coated REcN with a layer of plant‑derived sugar chains called fructooligosaccharides and then mineralized it with a thin shell of calcium carbonate, creating REcN‑F/Ca. Electron microscopy revealed a smooth, protective shell around each cell. In simulated digestive fluids, this coating helped far more bacteria stay alive and keep their ability to neutralize highly reactive radicals. The mineral shell dissolved quickly in acidic stomach‑like fluid but more slowly in conditions mimicking the intestine and inflamed colon, releasing the bacteria where they are most needed. When given to mice, the coated strain colonized the gut better than its unmodified counterpart and nudged immune cells toward a less inflammatory state.
Turning Around Obesity, Blood Sugar, and Gut Ecology in Mice
The crucial test came in male mice fed a high‑fat diet that drives obesity and type 2 diabetes‑like symptoms. Compared with untreated animals, mice receiving REcN‑F/Ca gained far less weight, carried less abdominal fat, and showed improved fasting blood sugar and insulin sensitivity. Their bodies responded more normally to a sudden glucose challenge, unlike high‑fat‑fed mice whose insulin secretion faltered. Under the microscope, their fat tissues contained fewer inflammatory immune cells and fewer fat droplets, and their colons showed less oxidative damage. At the gene level, the treated mice ramped up activity of pathways governed by a master regulator called PPARα, which promotes fat burning and calms inflammation. When this pathway was chemically blocked, the benefits of the engineered probiotic largely disappeared, suggesting that PPARα is a key link between the microbe and host metabolism.
Rebuilding a Healthier Microbial Neighborhood
The engineered probiotic also reshaped the wider gut community. High‑fat feeding had stripped the mice’s intestines of microbial diversity and depleted bacteria that produce short‑chain fatty acids—small molecules that nourish the gut lining and help regulate immunity and glucose control. REcN‑F/Ca partially restored diversity and strongly enriched families such as Lachnospiraceae and the genus Blautia, both known for generating the beneficial fatty acid butyrate and for being linked in human studies to better metabolic health. Measurements of fecal chemicals confirmed that levels of key short‑chain fatty acids, which had plunged under the high‑fat diet, rebounded to near‑normal in the treated mice.
What This Could Mean for Future Diabetes Care
Taken together, the work shows that it is possible to “train” a probiotic to thrive in the stressful gut environment of obesity and diabetes and to design a simple coating that delivers it alive to the colon. In mice, this bioengineered microbe corrected several hallmarks of metabolic disease at once: it neutralized excess reactive molecules, shifted the gut ecosystem toward beneficial species, activated host pathways that favor fat burning, and eased inflammation and insulin resistance. While many steps remain before such an approach could be tested in people—including long‑term safety, dosing, and studies in both sexes—this study outlines a path toward next‑generation probiotics that do more than survive: they actively partner with our bodies to rebalance metabolism from the inside out.
Citation: Mao, C., Jin, W., Dou, L. et al. Bioengineered ROS-tolerant probiotic reshapes gut microbiota-host axis to ameliorate type 2 diabetes in male mice. Nat Commun 17, 3339 (2026). https://doi.org/10.1038/s41467-026-70138-3
Keywords: engineered probiotics, gut microbiome, type 2 diabetes, oxidative stress, short-chain fatty acids