Clear Sky Science · en
Thermal catalytic synthesis of ammonia using uranium/graphdiyne composite at mild conditions
Why a new path to ammonia matters
Ammonia is a quiet giant of modern life. It underpins fertilizer production for global agriculture and is emerging as a potential energy carrier. Yet the way we currently make it—the century‑old Haber–Bosch process—chews through huge amounts of fossil fuel, runs at searing temperatures and crushing pressures, and emits vast quantities of carbon dioxide. This study explores a radically different approach: a catalyst built from uranium atoms anchored on an ultra‑thin carbon sheet called graphdiyne, able to turn nitrogen and hydrogen into ammonia under much milder conditions, hinting at a cleaner future for this essential chemical.

Building a better stage for the reaction
The researchers begin with graphdiyne, a two‑dimensional carbon material made of benzene rings linked by short carbon chains. Its structure creates triangular pores, a large surface area, and a built‑in electronic gap that can be tuned by changing how many layers are stacked. Using supercritical carbon dioxide, they grow extremely thin graphdiyne sheets from single‑layer up to many layers, then carefully measure their thickness and electronic properties. They find that as more layers are added, the material’s bandgap shrinks in a predictable way and that a five‑layer version offers a particularly favorable electronic landscape for interacting with metals and activating nitrogen.
Pairing uranium with designer carbon
To turn this carbon sheet into a working catalyst, the team introduces uranium using a reactive uranium iodide solution. Thin graphdiyne layers are destroyed by this treatment, so they start from an eight‑layer film and partially etch it down, ending with a stable five‑layer graphdiyne scaffold decorated with tiny uranium clusters. Advanced electron microscopy shows that these clusters are made of only a few uranium atoms, regularly spaced on the carbon lattice at distances well suited to “grab” a nitrogen molecule between two neighboring metal sites. Spectroscopic measurements confirm that uranium sits mainly in intermediate oxidation states and that electrons flow from uranium into the graphdiyne, subtly reshaping the metal’s 5f orbitals, which are known to be powerful but hard‑to‑control participants in chemical bonding.
Making ammonia gently—and proving where it comes from
With this uranium/graphdiyne composite in hand, the researchers test ammonia production from nitrogen and hydrogen at temperatures around 150 °C and pressures near 15 bar—far below typical Haber–Bosch conditions. Ion chromatography shows that the catalyst produces far more ammonia than blank controls and conventional comparison catalysts under the same conditions, achieving one of the highest reported rates for such mild operation. The catalyst also works repeatedly over multiple cycles without significant loss of activity. To verify that the nitrogen in the product truly originates from the gas feed, the team performs labeling experiments with nitrogen‑15 and detects the corresponding isotopic signature in the resulting ammonium, ruling out contamination or decomposition of background nitrogen sources.
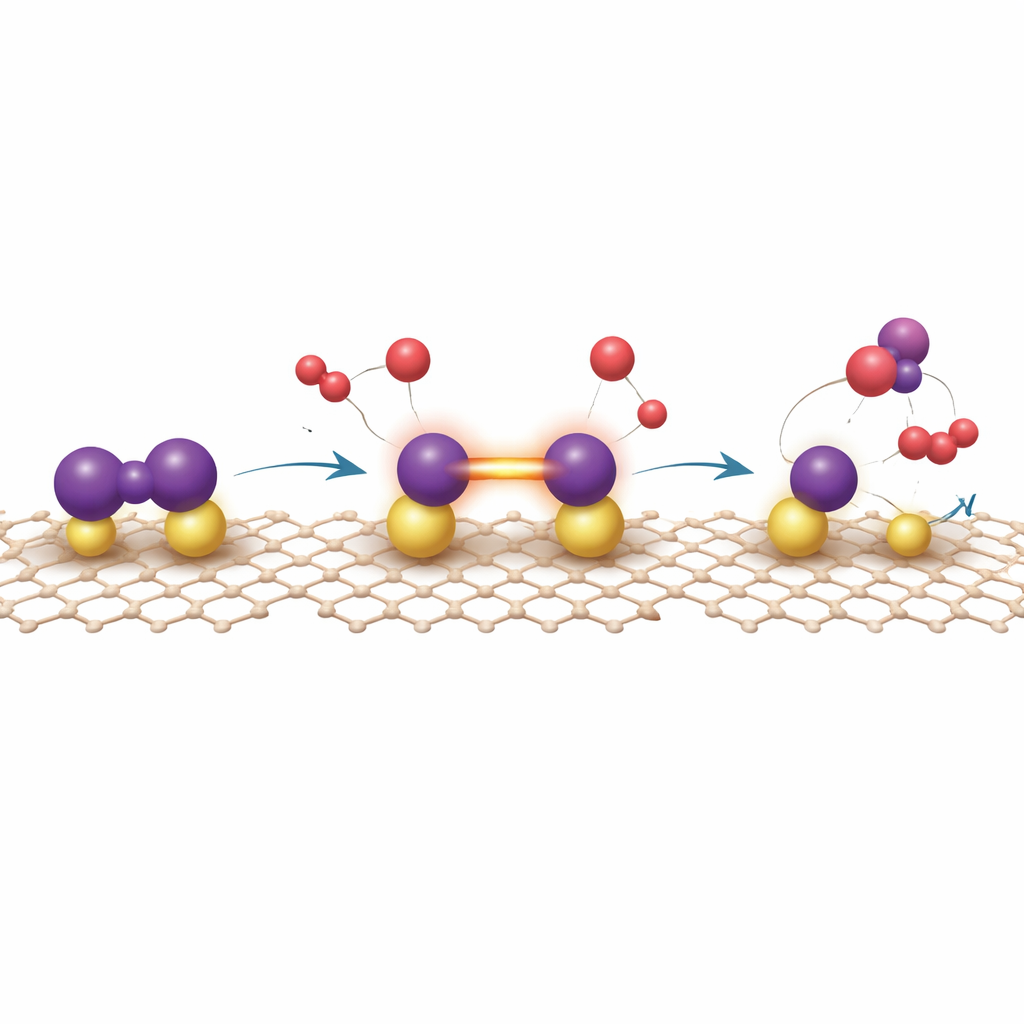
Peering into how the catalyst actually works
Experiments alone cannot fully reveal how this unusual surface breaks the stubborn nitrogen–nitrogen bond. The authors therefore combine X‑ray spectroscopies with quantum‑mechanical calculations. Theory shows that nitrogen prefers to bind in a “bridging” fashion between two uranium atoms, causing its bond to stretch and its antibonding orbitals to fill with back‑donated electrons from uranium’s 5f states, which are themselves tuned by their interaction with graphdiyne. From this activated state, hydrogen atoms add stepwise to one end of the nitrogen molecule—a so‑called distal pathway—eventually yielding ammonia that desorbs relatively easily, resetting the uranium centers. Competing routes in which nitrogen splits completely into separate atoms turn out to trap nitrogen too strongly, making further reaction sluggish. Calculations also indicate that hydrogen binds only moderately to the active site, explaining why the catalyst resists hydrogen “poisoning,” a common problem for conventional metal catalysts.
What this means for cleaner chemistry
Taken together, the results show that a carefully engineered partnership between uranium and graphdiyne can drive ammonia synthesis efficiently at far milder conditions than those used in today’s plants. The graphdiyne scaffold stabilizes uranium in just the right form and spacing, while its extended electron system reshapes the metal’s 5f orbitals to activate nitrogen and guide the reaction along a productive, easily reversible route. Although this specific catalyst is not yet ready to replace industrial Haber–Bosch units, it demonstrates a powerful design idea: actinide atoms anchored on tailored carbon frameworks can carry out demanding chemical transformations in ways that might one day be both more energy‑efficient and more environmentally friendly.
Citation: Xiong, S., Wang, W., Wang, F. et al. Thermal catalytic synthesis of ammonia using uranium/graphdiyne composite at mild conditions. Nat Commun 17, 2894 (2026). https://doi.org/10.1038/s41467-026-69691-8
Keywords: ammonia synthesis, nitrogen fixation, uranium catalyst, graphdiyne, green chemistry