Clear Sky Science · en
Spatiotemporal interplay between epithelial and mesenchymal cells drives human dentinogenesis
Why this matters for teeth
Most of a tooth is made of dentin, the hard, living tissue under the enamel that gives teeth their strength and sensitivity. When deep cavities, cracks, or genetic diseases destroy too much dentin, today’s treatments can only patch the damage with artificial materials, not rebuild natural tissue. This study uncovers how human teeth normally build dentin during development and shows how those same rules can be reused to coax adult stem cells to regrow real dentin, pointing toward future treatments that repair teeth from the inside out.
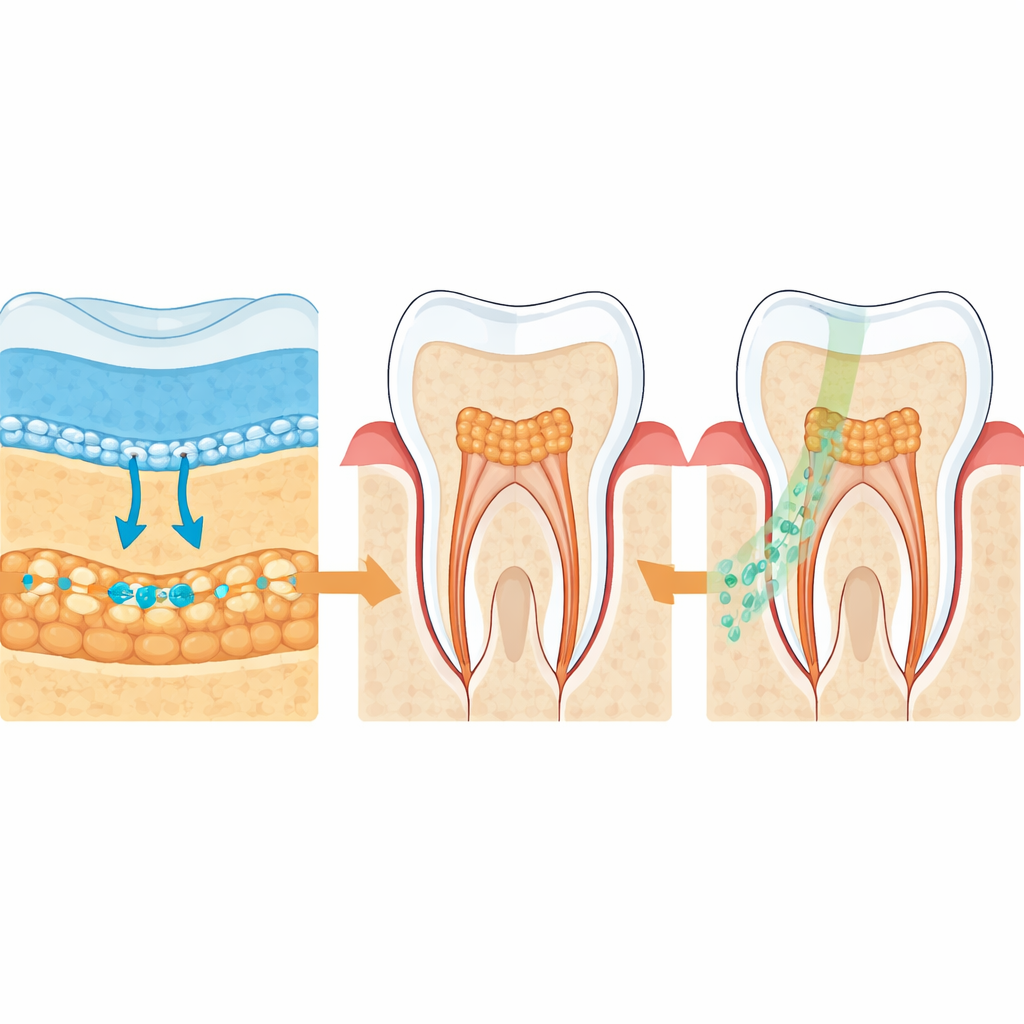
How a tooth takes shape
Human teeth arise from a close partnership between two cell layers: an outer sheet of epithelial cells and an inner mass of mesenchymal cells that later form the dental pulp and dentin. The authors created a detailed “cell atlas” of human tooth development, from early embryonic buds through erupted teeth, by combining single-cell RNA sequencing with spatial transcriptomics, which reads out which genes are active and where. This allowed them to track which cell types appear when, how they are arranged in the forming tooth, and which chemical signals they exchange as the tooth matures.
The relay of growth signals
The team focused on two major families of cell‑to‑cell signals. One, called WNT, is well known for driving cell growth and early patterning; the other, NOTCH, often helps cells choose their final identities. They found that epithelial cells overlying the developing tooth secrete WNT signals early on, while neighboring mesenchymal cells carry the matching receptors. As development advances, the pattern shifts: WNT signals fade, NOTCH signals from the epithelium increase, and the mesenchymal cells switch on NOTCH receptors and natural WNT blockers. This “relay” from a WNT‑dominated phase to a NOTCH‑dominated phase appears finely tuned in space and time, steering which pulp cells multiply and which become dentin‑forming odontoblasts.
The special pulp cells that hear the call
Among the mesenchymal cells, the researchers identified a previously underappreciated subgroup marked by a molecule called DLX6-AS1. These cells sit right beneath the epithelium, in prime position to sense incoming signals. Their gene activity shows that they respond strongly to both WNT and NOTCH and are closely linked to genes involved in dentin formation. Developmental analyses suggest that these DLX6-AS1–positive cells are progenitors that can either differentiate into odontoblasts or remain as a reserve pool just under the dentin layer, ready to respond to injury later in life.
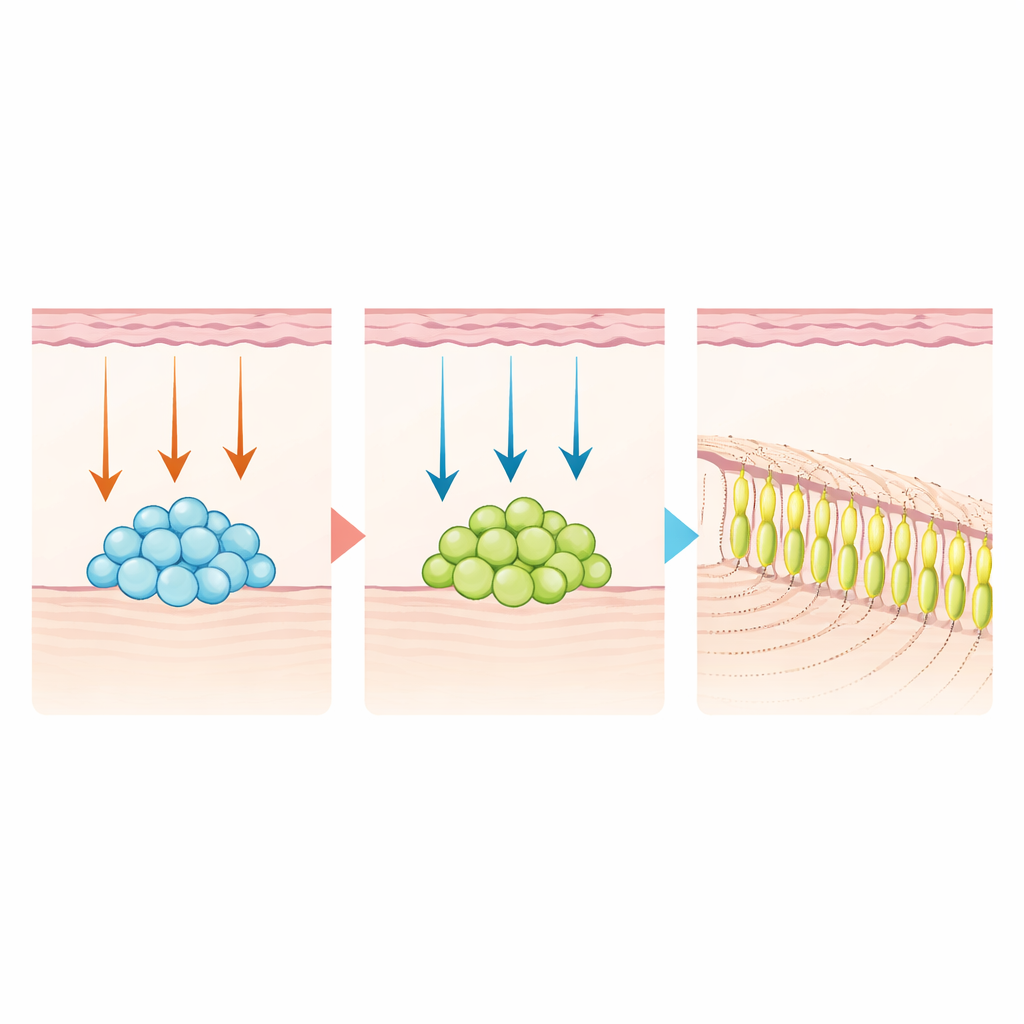
Replaying development to grow new dentin
To test whether these insights could be turned into therapy, the team isolated DLX6-AS1–positive stem cells from human dental pulp. In laboratory dishes, they exposed the cells first to a cocktail of WNT proteins and then, after several days, to the NOTCH activator JAG1, mimicking the developmental relay. This two‑step treatment drove the cells to mature into odontoblast‑like cells and to deposit mineralized, dentin‑like matrix much more effectively than standard protocols. When combined with epithelial cells and implanted under the kidney capsules of mice, only the WNT‑then‑NOTCH–treated cells produced organized, tubular dentin structures.
Repairing damaged teeth in animals
The researchers next created injuries in the molars of nude mice that mimic deep cavities reaching the pulp. They introduced human DLX6-AS1–positive dental pulp stem cells that had been primed with WNT signals, together with local NOTCH activation at the injury site. Over the following weeks, imaging and microscopic analysis revealed that these cells formed a well‑organized dentin bridge across the defect, with fine, parallel tubules resembling natural dentin rather than the disordered “patch” tissue usually seen in repair. The transplanted human cells lined up at the edge of the new dentin and expressed markers of mature odontoblasts, confirming that they were actively rebuilding the tooth.
What this means for future tooth repair
By mapping how epithelial and mesenchymal cells talk to each other over time, this study shows that a carefully timed handoff from WNT to NOTCH signals guides a special class of pulp stem cells to become dentin‑forming cells. Recreating that sequence in adult human dental pulp stem cells allowed them to regenerate high‑quality dentin in an animal model of tooth damage. While clinical application will require safer delivery methods and long‑term testing, the work lays a conceptual and practical foundation for regenerative dental therapies that could one day replace synthetic fillings with living, self‑repairing tooth tissue.
Citation: Wei, W., Wu, C., Sun, J. et al. Spatiotemporal interplay between epithelial and mesenchymal cells drives human dentinogenesis. Nat Commun 17, 2791 (2026). https://doi.org/10.1038/s41467-026-69545-3
Keywords: tooth regeneration, dental pulp stem cells, dentin repair, cell signaling, regenerative dentistry