Clear Sky Science · en
Hybrid macrophage-mitochondria extracellular vesicles for mitochondrial ROS regulation in diabetic wounds
Why stubborn diabetic wounds matter
For many people with diabetes, a small blister or scratch on the foot can spiral into a chronic wound that refuses to heal, sometimes ending in infection, hospitalization, or even amputation. These hard‑to‑treat sores are fueled by runaway inflammation inside immune cells that sit in the wound. In this study, scientists built a highly targeted "nanovesicle"—a tiny, cell‑like bubble—that sneaks directly into the energy factories of these immune cells to calm harmful chemistry without completely shutting it down, speeding healing in diabetic mice.
When healing stalls instead of progressing
Normal wound healing moves through a tidy sequence: brief inflammation, tissue regrowth, and long‑term remodeling. In diabetes, that first phase often gets stuck. Macrophages—the immune cells that clean debris and coordinate repair—stay locked in an aggressive, inflammation‑driven state instead of switching to a nurturing, rebuilding mode. A major culprit is excessive reactive oxygen species made by mitochondria, the tiny power plants inside cells. Too much of this chemically reactive oxygen damages mitochondrial DNA, fats, and proteins, destabilizing the cell and preventing macrophages from switching into a repair‑friendly, anti‑inflammatory role.
A smart antioxidant that waits for trouble
Simply flooding wounds with antioxidants has not solved the problem; it can blunt both bad and good signals and miss the mitochondria entirely. The researchers instead designed a “prodrug” version of a natural antioxidant called caffeic acid phenethyl ester. This prodrug is chemically locked in a stable, inactive form that becomes active only when it encounters high levels of oxidants, such as the ones found inside damaged mitochondria. Careful chemistry linked the antioxidant to a reactive bond and a greasy tail, making the molecule water‑stable yet eager to tuck into fatty membranes. Tests showed that this prodrug stayed intact in mild conditions but broke apart and released strong antioxidants when exposed to higher oxidant levels, closely mimicking what happens inside stressed mitochondria.
Tiny hybrid shells that home in on immune power plants
To deliver this smart cargo exactly where it was needed, the team built artificial extracellular vesicles from real biological membranes. They stripped and purified outer membranes from macrophages and from their mitochondria, then fused these into hybrid shells. Because these membranes carry the same proteins as the original cells and organelles, the resulting vesicles can recognize macrophages and then their mitochondria, much like a matching key and lock. Experiments showed that when these drug‑loaded hybrid vesicles contacted macrophages, they were swallowed through natural uptake routes and then fused with mitochondrial membranes. Inside, the high oxidant environment clipped the chemical lock on the prodrug, releasing active antioxidant precisely where the excess damage originated. 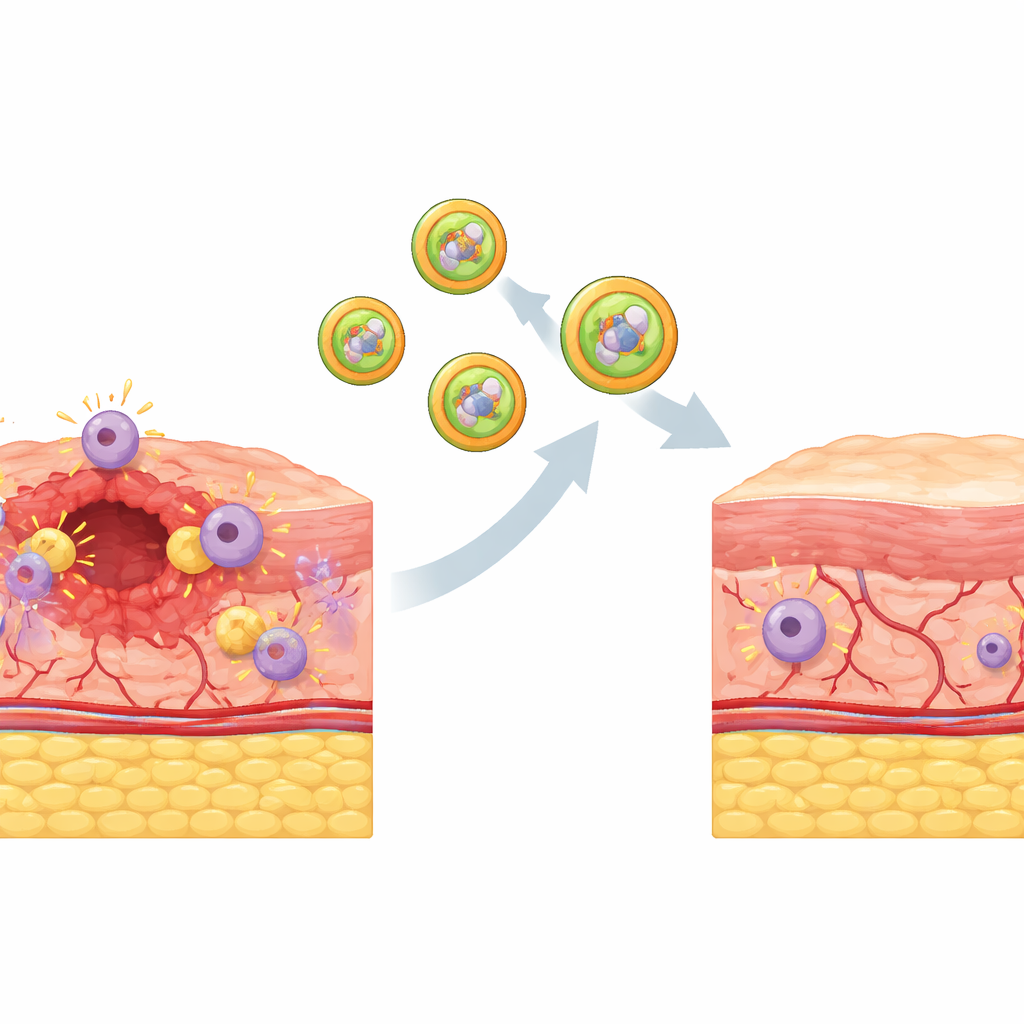
Rewiring immune cells and rescuing damaged tissue
In cell cultures, this targeted system sharply reduced harmful mitochondrial oxidants, limited fat damage inside mitochondria, restored the electrical potential they need to make energy, and improved ATP production. Electron microscopy revealed that swollen, misshapen mitochondria reverted toward slim, well‑organized structures once treated. These internal repairs had major consequences for cell behavior: macrophages shifted away from an inflammatory profile, dialing down genes and markers linked to attack mode and boosting those tied to a soothing, tissue‑repairing state. 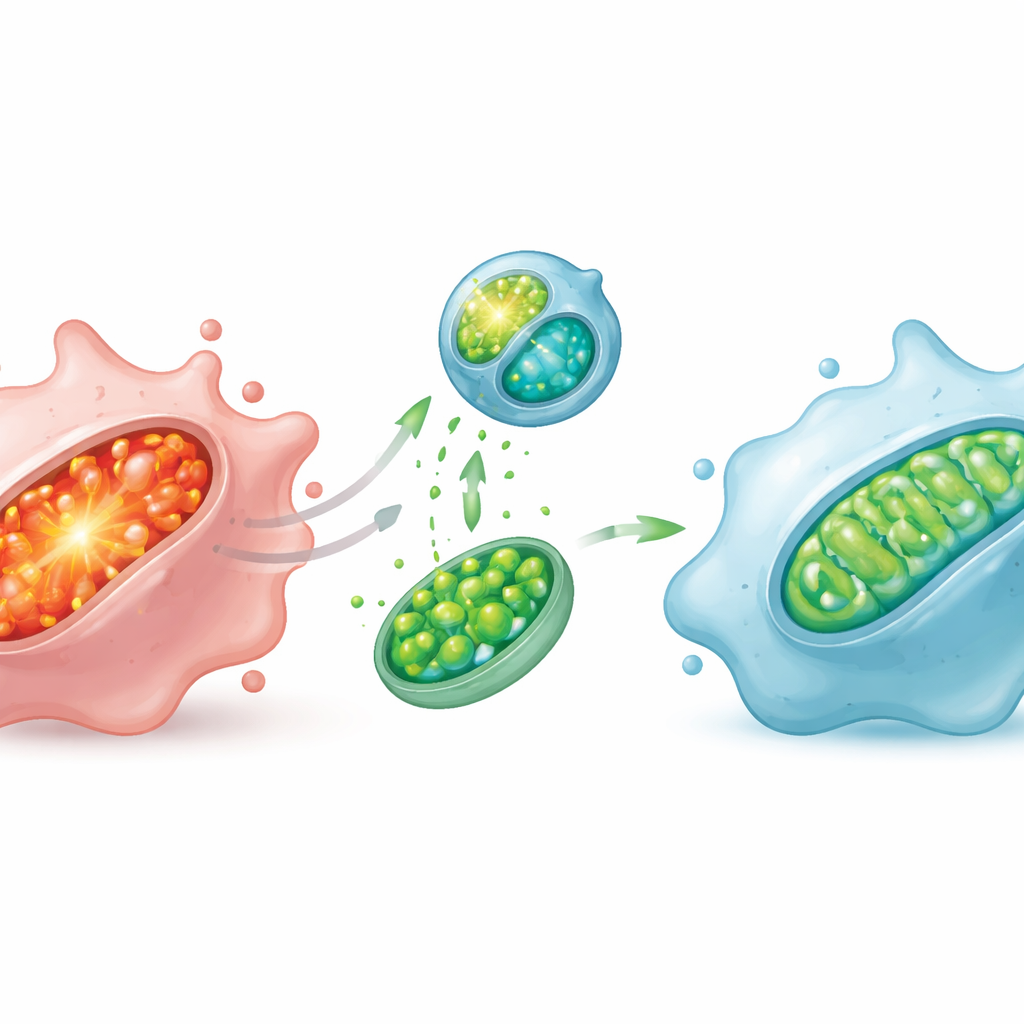
Helping wounds close in diabetic animals
When the researchers injected the hybrid vesicles under the skin around full‑thickness wounds in diabetic mice, the particles spread through the damaged tissue and were taken up mainly by macrophages, including their mitochondria. Over days, mitochondrial oxidant levels dropped, inflammatory signals waned, and repair‑oriented macrophages became more common. Compared with several control treatments—including the free drug, vesicles without the prodrug, and other membrane‑based particles—the hybrid vesicles produced faster wound closure, earlier regrowth of the outer skin layer, more new blood vessels, and denser, better organized collagen fibers. The wounds treated with the new system looked and functioned more like healed, healthy skin.
What this could mean for future care
The study shows that it is possible not just to block reactive oxygen species, but to tune them in the right place and at the right time by combining bio‑inspired targeting with on‑demand chemistry. By homing in on macrophage mitochondria and releasing antioxidants only under high‑stress conditions, these hybrid vesicles restore balance instead of merely suppressing signals. In diabetic mice, this precise control softens chronic inflammation and jump‑starts normal repair, pointing toward a new class of treatments for diabetic wounds and other inflammatory diseases tied to mitochondrial imbalance.
Citation: Fan, L., Zhang, C., Xu, Z. et al. Hybrid macrophage-mitochondria extracellular vesicles for mitochondrial ROS regulation in diabetic wounds. Nat Commun 17, 3285 (2026). https://doi.org/10.1038/s41467-026-69383-3
Keywords: diabetic wound healing, macrophage mitochondria, nanovesicles, oxidative stress, targeted antioxidants