Clear Sky Science · en
Spatiotemporal alignment of hole transfer and water oxidation for highly efficient photocatalytic water splitting
Turning Sunlight and Water into Fuel
Splitting water into hydrogen and oxygen using only sunlight is a long-sought goal for clean energy, because hydrogen can serve as a carbon‑free fuel made directly from abundant water. This paper explores why a particular material, aluminum‑doped strontium titanate, comes remarkably close to doing this with almost no wasted light, and reveals how its internal structure carefully guides charges to the right places at the right times.

A Special Crystal for Splitting Water
Overall water splitting means using light to drive both halves of the reaction: making hydrogen and oxygen from pure water with no added chemicals. Many photocatalysts can do one half efficiently, but very few handle both simultaneously without losing most of the absorbed energy. Aluminum‑doped strontium titanate (SrTiO3:Al) is a striking exception, reaching apparent quantum efficiencies near 100%, meaning nearly every absorbed photon leads to useful chemical change. The authors use samples that achieve over 90% efficiency as a model system to ask: what, exactly, is aluminum doing inside this crystal that makes it so effective?
Shaping the Crystal from the Outside In
The team compares crystals made by different preparation routes and with different aluminum contents. They find that performance does not hinge on obvious traits like particle size or light absorption. Instead, the key is where the aluminum atoms end up. In the best samples, aluminum is concentrated in a thin shell near the particle surface, while the bulk contains only a small, uniform amount. This “gradient” arrangement subtly shrinks the lattice and, crucially, suppresses defects such as oxygen vacancies and titanium in an unwanted charge state; these defects would otherwise act as recombination centers that waste photogenerated charges. When aluminum is distributed poorly—clustered only at corners, or spread too uniformly—the water‑splitting efficiency drops sharply.
Guiding and Storing Charges in Time and Space
Using advanced surface photovoltage imaging, the authors map how charges move inside single particles under illumination. The gradient in aluminum concentration creates an internal electric field that pushes positively charged holes from the interior toward the surface. At the same time, aluminum‑related sites at or near the surface act as traps that hold these holes for unusually long times—extending their lifetime from around one hundred billionths of a second to about a hundredth of a second. Detailed transient measurements show that this long‑lived trapped‑hole population barely decays over microsecond‑to‑millisecond timescales, meaning recombination with electrons is strongly suppressed. Electrons are instead drawn toward specific facets decorated with metallic cocatalysts, where hydrogen is produced, while holes accumulate where oxygen will form.
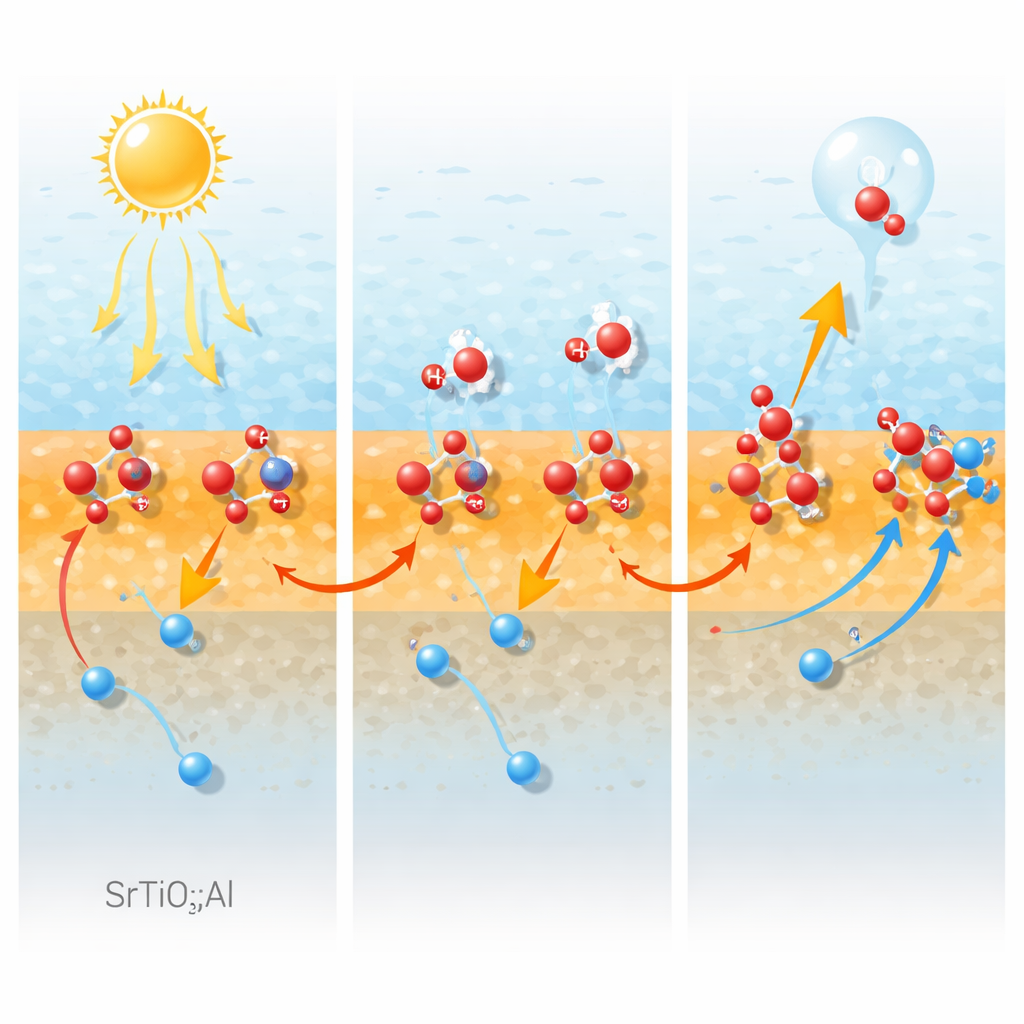
Creating the Right Spots for Water to React
To understand whether these hole‑trapping sites also help water itself, the researchers probe the local environment of aluminum using high‑field nuclear magnetic resonance and infrared spectroscopy. They identify two main types of aluminum centers: highly symmetric units buried in the bulk, and less symmetric surface units bound to hydroxyl groups. These surface "hydroxylated" aluminum sites turn out to be prime landing pads for water molecules. Their signals weaken when the samples are dehydrated or aged, and this loss closely tracks a drop in water adsorption and in oxygen‑evolution activity. Further tests show that the material can still oxidize water quite efficiently even without an added oxygen‑evolution cocatalyst, and that aluminum greatly boosts the intrinsic ability of the surface to carry out the difficult oxygen‑forming half‑reaction.
Linking the Microscopic Dance to the Big Energy Picture
Computational simulations support a mechanism in which neighboring hydroxyl groups on aluminum‑containing surface sites help water molecules couple and form oxygen–oxygen bonds while shedding protons. By aligning, in both space and time, long‑lived positive charges with these specially configured water‑binding sites, the material ensures that the slow, multi‑step oxygen‑forming reaction can keep up with the rate at which light supplies charges. In simple terms, aluminum does double duty: it builds an internal ramp that moves charges to the surface and it shapes the very sites where water is activated. This dual role explains how aluminum‑doped strontium titanate pushes water splitting efficiencies so close to the theoretical limit, and provides design principles for future photocatalysts that aim to turn sunlight and water into clean fuel with minimal losses.
Citation: Luo, Y., Chen, R., Dittrich, T. et al. Spatiotemporal alignment of hole transfer and water oxidation for highly efficient photocatalytic water splitting. Nat Commun 17, 2767 (2026). https://doi.org/10.1038/s41467-026-69276-5
Keywords: photocatalytic water splitting, solar hydrogen, strontium titanate, charge separation, oxygen evolution