Clear Sky Science · en
A genetic toolkit for the human gut bacterium Mediterraneibacter gnavus identifies capsular polysaccharides as a competitive colonization factor
Why a gut microbe’s “coat” matters
Our intestines are home to trillions of microbes that can help or harm us. One common resident, the bacterium Mediterraneibacter gnavus, is often found in higher numbers in people with inflammatory bowel diseases like Crohn’s disease. Yet not all strains of this species behave the same way. This study asks a simple but important question: which genes make some M. gnavus strains better at living in the gut, and how does that affect inflammation?
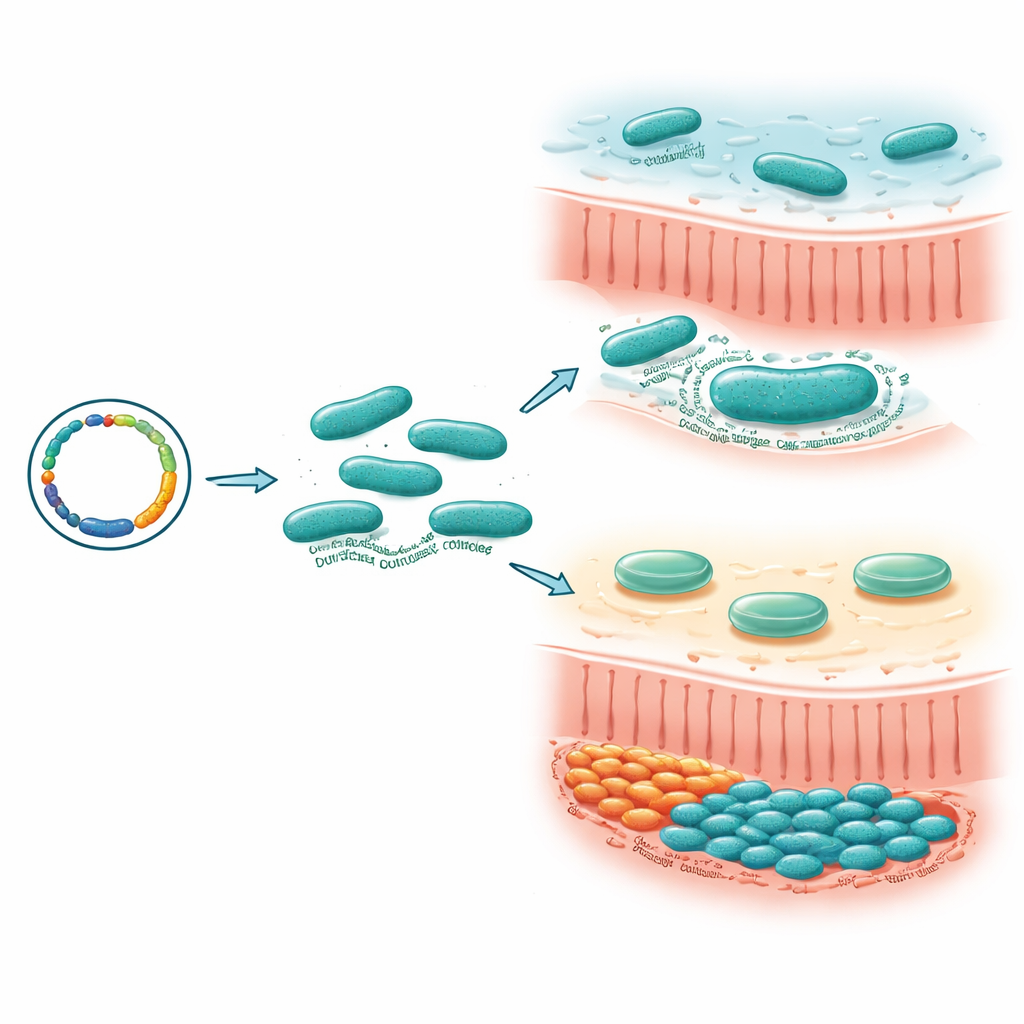
Building a toolbox to edit a stubborn microbe
Many gut bacteria are difficult to tinker with genetically, which has slowed efforts to understand what their individual genes do. The researchers first solved this technical roadblock for M. gnavus. They created shuttle plasmids that can move DNA back and forth between E. coli and M. gnavus, along with switches to turn genes on only when needed and a set of bright fluorescent tags to track bacterial cells. They also adapted two systems for permanently changing the genome: a mobile DNA insertion method and a CRISPR/Cas9-based approach. Together, these tools allowed them to delete or disrupt specific genes, mark strains with color, and check the effects in both lab dishes and mice.
Pinpointing genes that shape the bacterial surface
Armed with this toolkit, the team focused on genes called sortases, which act like molecular staplers that attach proteins and sugars to the bacterial cell wall. M. gnavus carries eight predicted sortase genes. By knocking out most of them one by one, the scientists showed that one enzyme, SrtB3, is essential for fastening powerful immune-activating proteins—called superantigens—to the cell surface. When SrtB3 was lost, these proteins were no longer anchored; instead, they leaked into the surrounding medium, and antibody binding on the cell surface dropped sharply. This confirmed that individual sortase types specialize in handling distinct sets of surface molecules.
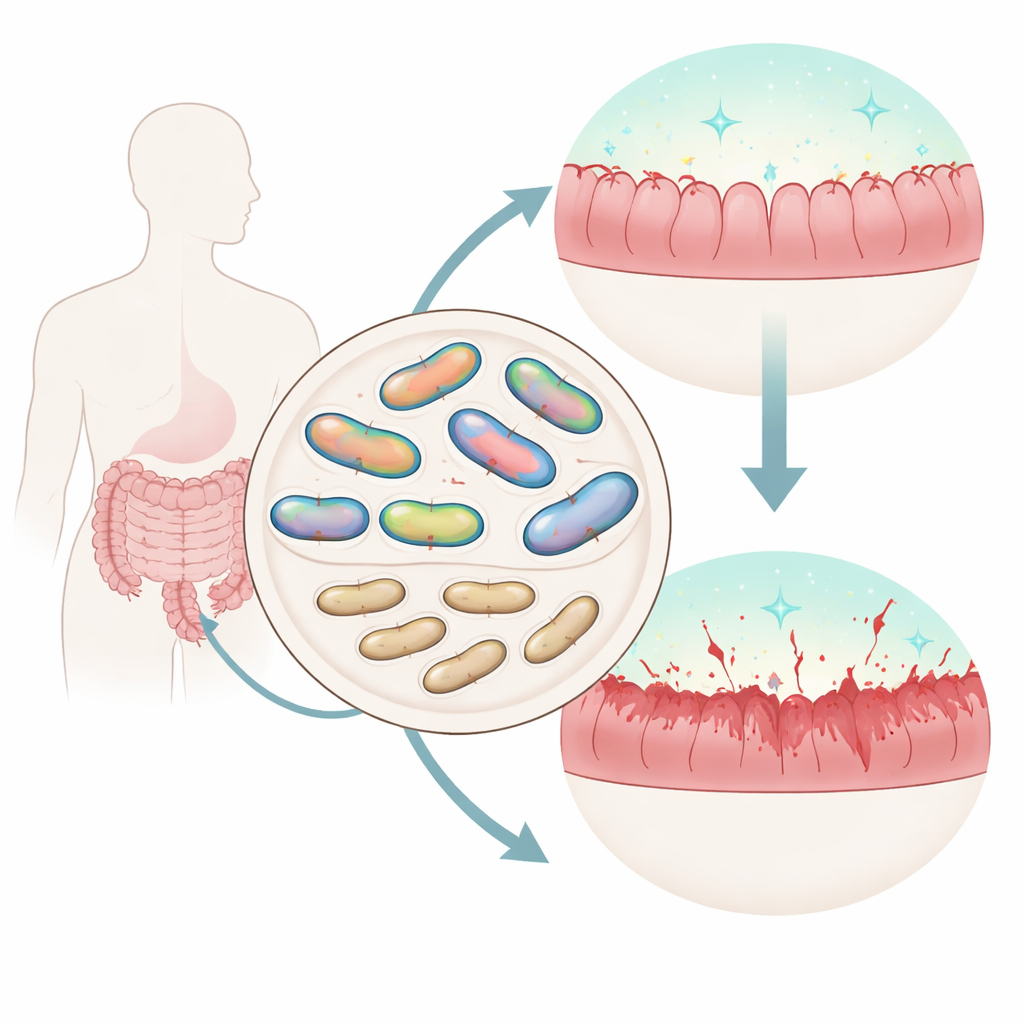
A sugary capsule that boosts gut colonization
The most striking findings centered on another sortase, SrtB4. Under the microscope, normal M. gnavus cells appear surrounded by a pale “halo”: a capsule made of complex sugars that forms a soft coat around each bacterium. When the srtB4 gene was disabled, this halo vanished. Chemical staining and electron microscopy confirmed a major loss of capsular polysaccharide, and the cells clumped tightly instead of forming loose pellets. Looking next to srtB4 on the chromosome, the researchers uncovered a cluster of genes predicted to build and attach this capsule. Disrupting several of these neighbors produced the same capsule-deficient look, tying the whole cluster to capsule manufacture.
From mouse gut competition to inflammation
To test why the capsule matters, the team pitted normal and mutant strains against each other inside germ-free mice. When capsule-deficient strains were mixed with their capped counterparts and fed to the animals, the mutants rapidly lost ground and almost disappeared from the gut, even though they grew just fine in lab culture. This showed that the capsule gives M. gnavus a sharp competitive edge during real intestinal colonization. But the story did not end with colonization. In cell culture, capsule-lacking strains triggered much higher levels of inflammatory signals from mouse immune cells than the capped strain. In a mouse model of chemically induced colitis, animals colonized with the capsule-deficient mutant suffered greater weight loss and more severe disease scores, even though overall bacterial numbers were similar.
Links to human disease and what it may mean
Finally, the authors compared many complete genomes of M. gnavus strains isolated from people. They found that the capsule-related gene cluster containing srtB4 was present much more often in strains from healthy individuals than in strains from Crohn’s disease patients. Other strains lacking this cluster also failed to make a visible capsule in the lab. Although more sampling is needed, this pattern suggests that capsule-bearing strains may be better long-term residents that provoke less inflammation, whereas “bare” strains without the capsule may be more damaging to the gut lining.
Take-home message for gut health
This work shows that a single patch of genes controlling a sugary outer coat can decide whether M. gnavus wins the battle to colonize the gut and how strongly it stirs up inflammation. The new genetic toolkit now makes it possible to dissect many other features of this microbe and its relatives. For non-specialists, the key idea is that not all members of a bacterial species are equal: subtle genetic differences in surface structures, like capsules, can tip the balance between a helpful companion and a harmful trigger of disease.
Citation: Obana, N., Nakato, G., Nomura, N. et al. A genetic toolkit for the human gut bacterium Mediterraneibacter gnavus identifies capsular polysaccharides as a competitive colonization factor. Nat Commun 17, 3855 (2026). https://doi.org/10.1038/s41467-026-69022-x
Keywords: gut microbiome, Mediterraneibacter gnavus, bacterial capsule, inflammatory bowel disease, CRISPR genetics