Clear Sky Science · en
NEO-STIM advances personalized neoantigen-specific adoptive T cell therapy
Turning the Immune System into a Custom Cancer Drug
Cancer often slips past our immune defenses by disguising itself as normal tissue. This study describes a way to turn a patient’s own immune cells into a custom-made living drug that homes in on the unique mutations in that person’s tumor. For readers following advances in cancer treatment, it shows how researchers are moving beyond one-size-fits-all therapies toward highly tailored cell treatments built from a simple blood draw rather than surgery.
Why Targeting Tumor “Typos” Matters
Every cancer carries genetic “typos” that can alter proteins inside tumor cells. Fragments of these altered proteins, called neoantigens, can appear on the cell surface and mark the cancer as foreign. They are attractive targets because they are unique to the tumor and largely absent from healthy tissue, which means immune cells aimed at neoantigens should, in principle, spare normal organs. Existing immune therapies called tumor-infiltrating lymphocyte (TIL) therapies already benefit some people by expanding T cells that naturally seep into tumors. But TIL therapy demands a sizable tumor sample from surgery, captures only a small fraction of all possible neoantigen targets, and often relies on T cells that are already worn out by life inside the tumor’s hostile environment.
From Blood Draw to Personalized T Cell Product
To overcome these hurdles, the authors developed NEO-STIM, a stepwise lab method that builds a personalized T cell treatment entirely from peripheral blood. Instead of starting with tumor tissue, they begin with white blood cells collected by leukapheresis, a sort of “immune cell harvest” from a vein. They first remove cell types known to blunt immune responses, then rely on naturally circulating dendritic cells—highly efficient sentinels of the immune system—to present carefully designed neoantigen peptides to T cells. Under a controlled mixture of growth signals, these encounters prime “naïve” T cells that have never seen the tumor as well as amplify rare memory cells already tuned to cancer. Two staggered rounds of stimulation over about four weeks yield billions of neoantigen-reactive T cells ready to be frozen as a drug product.
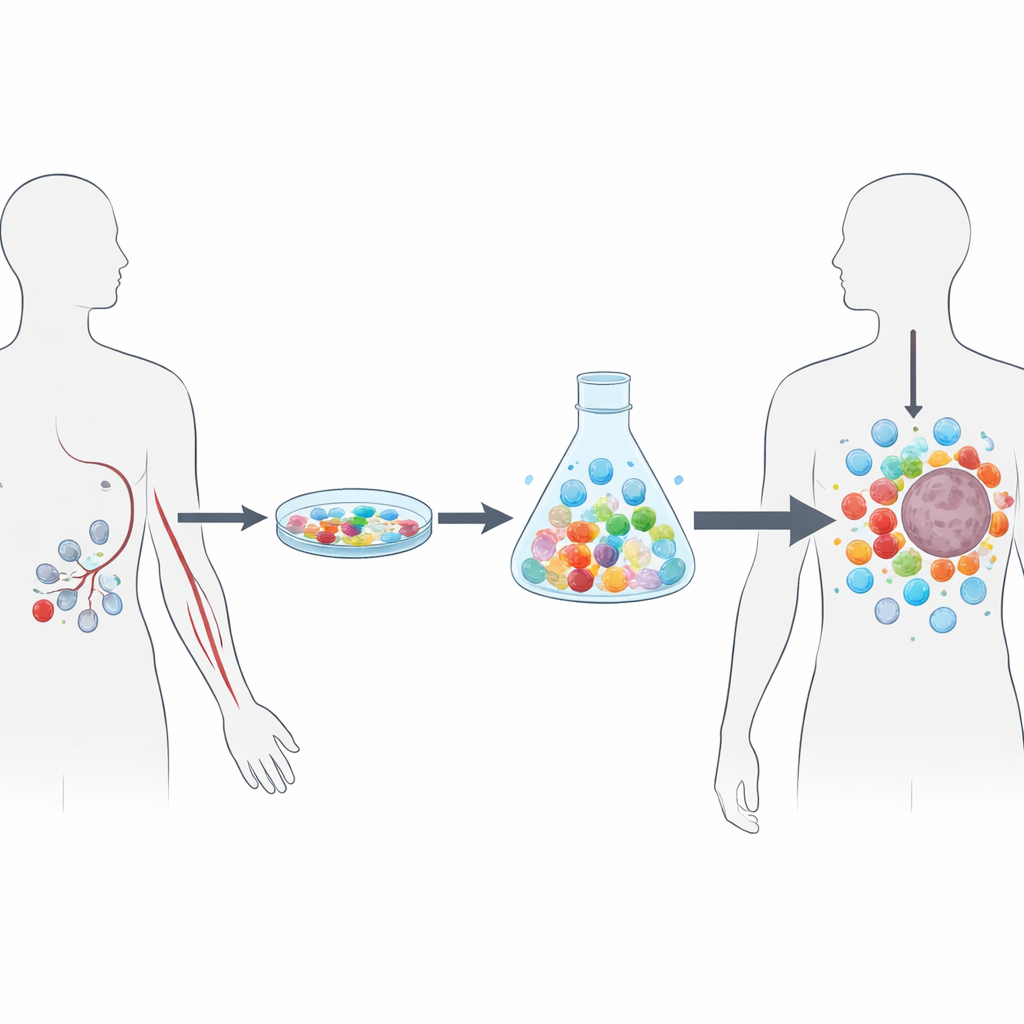
Building a Diverse, Functional Army of T Cells
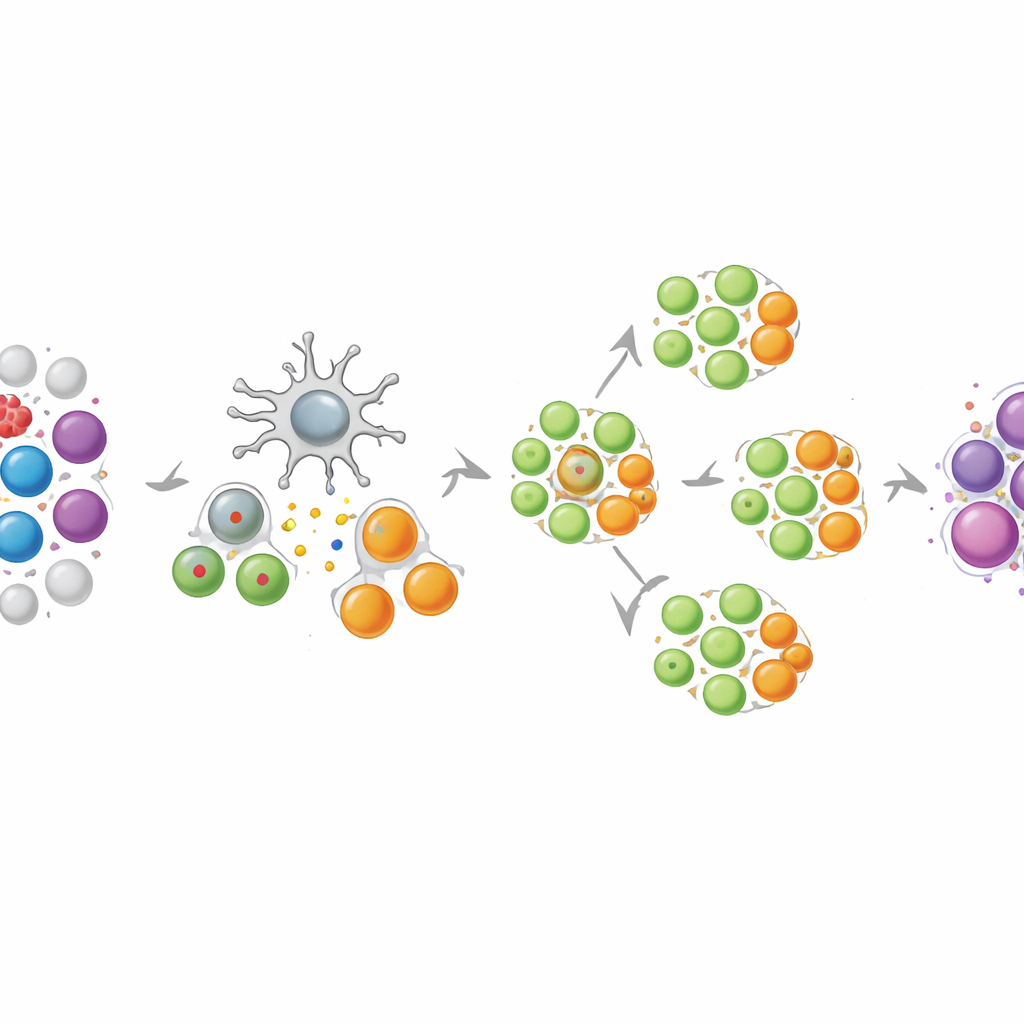
Using blood from healthy volunteers and from patients with melanoma or ovarian cancer, the team showed that NEO-STIM consistently generates a broad mix of T cell responses. The final products contained multiple CD8 T cells, which can directly kill cancer cells, and CD4 T cells, which coordinate and sustain immune attacks. Many of these responses were “de novo,” meaning they were undetectable before culture and thus newly raised in the dish. When the researchers challenged these cells with the very neoantigen fragments used for training, they saw strong “polyfunctional” behavior: single cells often released several defense molecules at once and displayed markers of active attack. Importantly, most responses were highly selective for the mutated form of each target and either ignored or strongly favored it over the normal version, reducing the risk of collateral damage to healthy tissue.
Putting the Cells to the Test Against Tumors
Lab tests went beyond recognition of isolated fragments. The scientists engineered tumor cell lines to naturally process and display the same neoantigens, then co-cultured them with NEO-STIM–derived T cells. In most cases, the T cells not only degranulated—a sign they were releasing toxic molecules—but also triggered early death signals inside the tumor cells, indicating true killing activity. For a subset of patients, the team obtained live cells from their actual tumors. Neoantigen-specific T cells produced by NEO-STIM recognized and responded to this autologous tumor material, even when no extra peptide was added, showing that they could sense physiologic levels of their targets as they would in the body.
Tracking the Cells Inside Patients
The approach has already entered a first-in-human clinical trial in people with advanced melanoma. By sequencing the T cell receptors—the unique molecular “ID tags” of each clone—the researchers tracked individual neoantigen-specific lineages from the lab dish into the bloodstream and tumors after infusion. Both newly induced and pre-existing clones were detectable weeks later. Inside tumors, these cells bore signatures of activation, cytotoxicity, and partial exhaustion, consistent with active engagement of cancer cells. Clinically, in the early trial, most patients achieved disease stabilization, and several showed measurable tumor shrinkage, supporting the potential of this strategy while larger studies proceed.
What This Means for Future Cancer Care
In everyday terms, NEO-STIM is a manufacturing recipe for turning a tube of blood into a personalized army of cancer-hunting cells that recognize each patient’s unique tumor mutations. The process reliably produces large numbers of active, mutation-focused T cells that can see and attack tumor material, and early clinical experience suggests it is feasible and tolerable. While questions remain about how long these cells persist and how best to combine them with other treatments, this work outlines a scalable path from genetic analysis of a tumor to a bespoke cell therapy, moving cancer care closer to highly individualized, mutation-directed immunotherapy.
Citation: Lenkala, D., Kohler, J., McCarthy, B. et al. NEO-STIM advances personalized neoantigen-specific adoptive T cell therapy. Nat Commun 17, 3683 (2026). https://doi.org/10.1038/s41467-026-68680-1
Keywords: cancer immunotherapy, neoantigen T cells, personalized medicine, adoptive cell therapy, melanoma