Clear Sky Science · en
Neurotrophic keratopathy in childhood: advances in understanding of pathogenesis and management
Why this matters for children’s sight
Most of us blink or feel pain the instant something touches our eye. That quick warning signal comes from tiny nerves on the eye’s surface and quietly protects our sight every day. This article explains what happens when those nerves do not work properly from birth or early childhood – a condition called neurotrophic keratopathy – and how new imaging, medicines, and surgery are helping many affected children keep good vision.
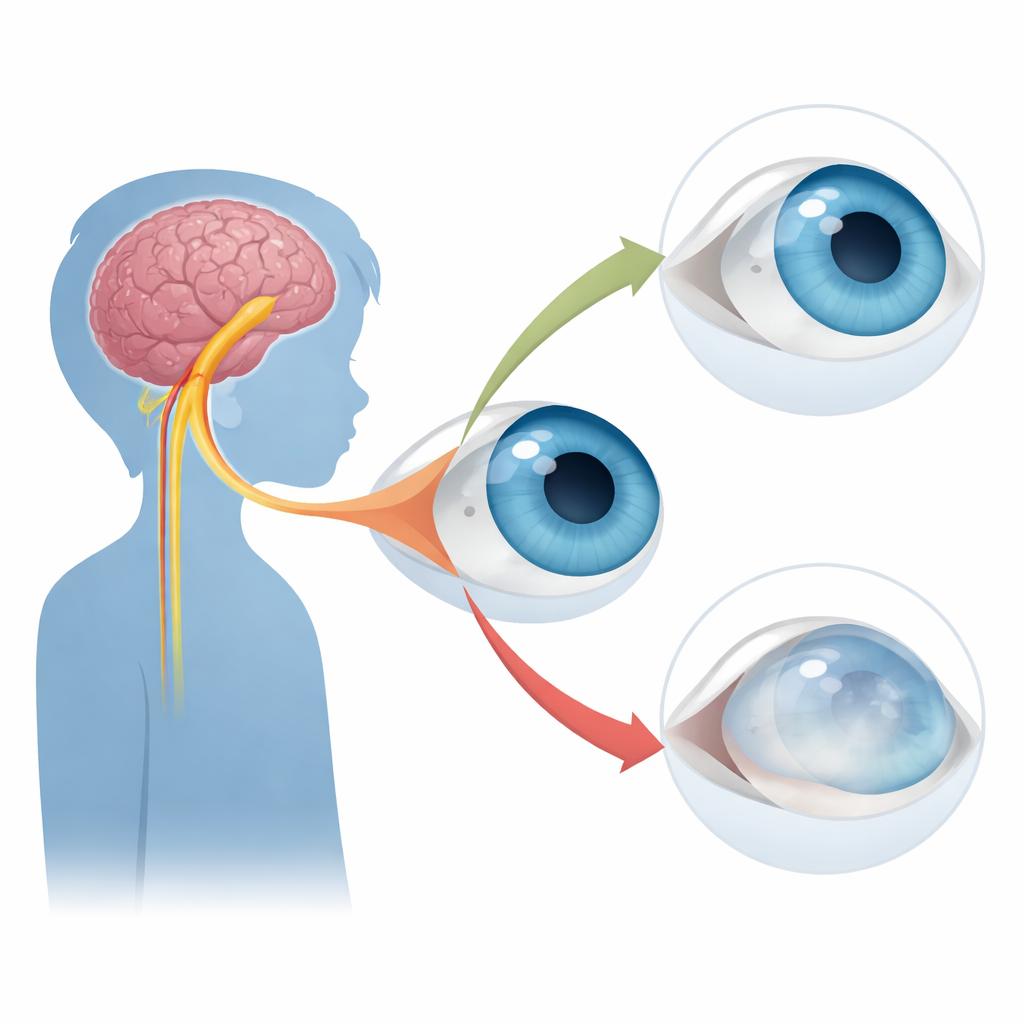
When the eye goes numb
In neurotrophic keratopathy, the sensory nerve that feeds the clear front window of the eye, the cornea, does not function properly. This nerve normally comes from a major head nerve called the trigeminal nerve and fans out into an intricate network of branches and endings in the cornea. These fibres do more than let a child feel pain or irritation: they help maintain a healthy, smooth corneal surface and trigger protective reflexes like blinking and tear production. When nerve input is lost, the surface can dry out, small defects may not heal, and in severe cases the cornea can thin, scar, or even perforate, threatening sight.
Different causes in children than in adults
In adults, corneal numbness usually appears after infections such as herpes, long-standing diabetes, or surgery and other acquired problems. In children, however, it is most often present from birth or determined by genes. Some children belong to rare “pain loss” syndromes in which small nerve fibres all over the body develop abnormally. These include hereditary sensory and autonomic neuropathies, where genetic changes disrupt a key growth signal called the neurotrophin pathway. Depending on which piece of this pathway is affected, children may not feel pain, may not sweat normally, or may damage their eyes and skin without realising it. Other children have broader brainstem or cranial nerve development disorders, such as pontine tegmental cap dysplasia or Möbius syndrome, in which several head nerves, including those serving the cornea, are underdeveloped. Still others have complex birth defect patterns like oculo-auriculo-vertebral spectrum, where eye, ear, and spine abnormalities can go hand-in-hand with a missing trigeminal nerve branch.
Spotting a quiet disease
Recognising this condition early in young children is difficult because they may not complain of pain and can be challenging to examine. Doctors therefore rely on a careful general and family history, testing of touch sensation on the cornea and surrounding skin, and a detailed microscope examination of the eye. Traditional staging systems for this disease were built around visible surface damage and did not account for children who already lack corneal feeling but still have a perfectly clear, healthy-looking eye. The authors propose a simple modification: adding an early stage reserved for eyes with complete loss of corneal sensation but otherwise normal appearance. This change should help researchers compare patients fairly in future trials and encourage closer monitoring of children before overt damage appears.
New ways to protect and restore the surface
Treatment is tailored to how advanced the corneal changes are. For children whose corneas remain intact, frequent lubricating drops and close follow-up may be enough to prevent ulcers and scarring. Once the surface starts to break down, more active measures are used, such as partially sewing the eyelids together or inducing a protective droop of the upper lid to shield the cornea. Doctors can also use drops made from the patient’s own blood serum or diluted insulin, which contain natural growth factors that nourish the surface. A major advance is the availability of eye drops containing a laboratory-made form of nerve growth factor. In many adults and some children, this treatment helps stubborn ulcers heal by boosting the survival and regrowth of corneal nerves, although it is unlikely to help when the nerve’s receptor is missing due to certain genetic mutations.
A surgical reset for corneal feeling
Perhaps the most dramatic development is corneal neurotisation, a microsurgical technique that brings a healthy sensory nerve to the numbed cornea. Surgeons either move a nearby forehead nerve directly or connect it to a nerve graft that is then tunneled to the eye’s surface. Over months, new nerve fibres grow into the cornea, restoring feeling and its protective “trophic” support. This operation can reduce recurrent ulcers and scarring and is particularly attractive for children facing a lifetime of disease, including those with both eyes affected. Early reports and a pooled analysis show meaningful improvements in corneal sensation and vision, but questions remain about the best surgical method and the ideal timing during visual development.
Looking ahead for young patients
The authors conclude that although childhood neurotrophic keratopathy is rare, it carries a high risk of infection, scarring, and lazy eye if not recognised promptly. With better brain imaging, refined corneal staging, nerve-supporting medicines, and nerve-transfer surgery, many children can now maintain or regain useful vision. Future work will focus on long-term results of these treatments, next-generation nerve growth factor–like drugs, and, in the longer term, gene-based therapies that correct the underlying nerve defects. For families and clinicians, the key message is that a “quiet” or numb eye in a child should never be ignored, because timely diagnosis and tailored care can make the difference between clear sight and preventable vision loss.
Citation: Jiang, J., Ashton, C.B.J. & Larkin, D.F.P. Neurotrophic keratopathy in childhood: advances in understanding of pathogenesis and management. Eye 40, 758–764 (2026). https://doi.org/10.1038/s41433-026-04278-7
Keywords: neurotrophic keratopathy, corneal anesthesia, pediatric eye disease, nerve growth factor, corneal neurotisation