Clear Sky Science · en
Production of the recombinant spider silk MaSp2 protein using the marine purple photosynthetic nonsulfur bacterium Rhodovulum sulfidophilum under autotrophic conditions
Turning Sunlight and Seawater into Strong Threads
Imagine making high‑performance “spider silk” without raising spiders, using only light, seawater, and gases from the air. This study explores how a marine bacterium can be turned into a tiny solar‑powered factory that spins the building blocks of spider silk while relying on carbon dioxide and nitrogen from the atmosphere. The work points toward new ways to produce advanced materials with far less dependence on farmland, freshwater, and fossil‑based ingredients.
A Tiny Worker from the Sea
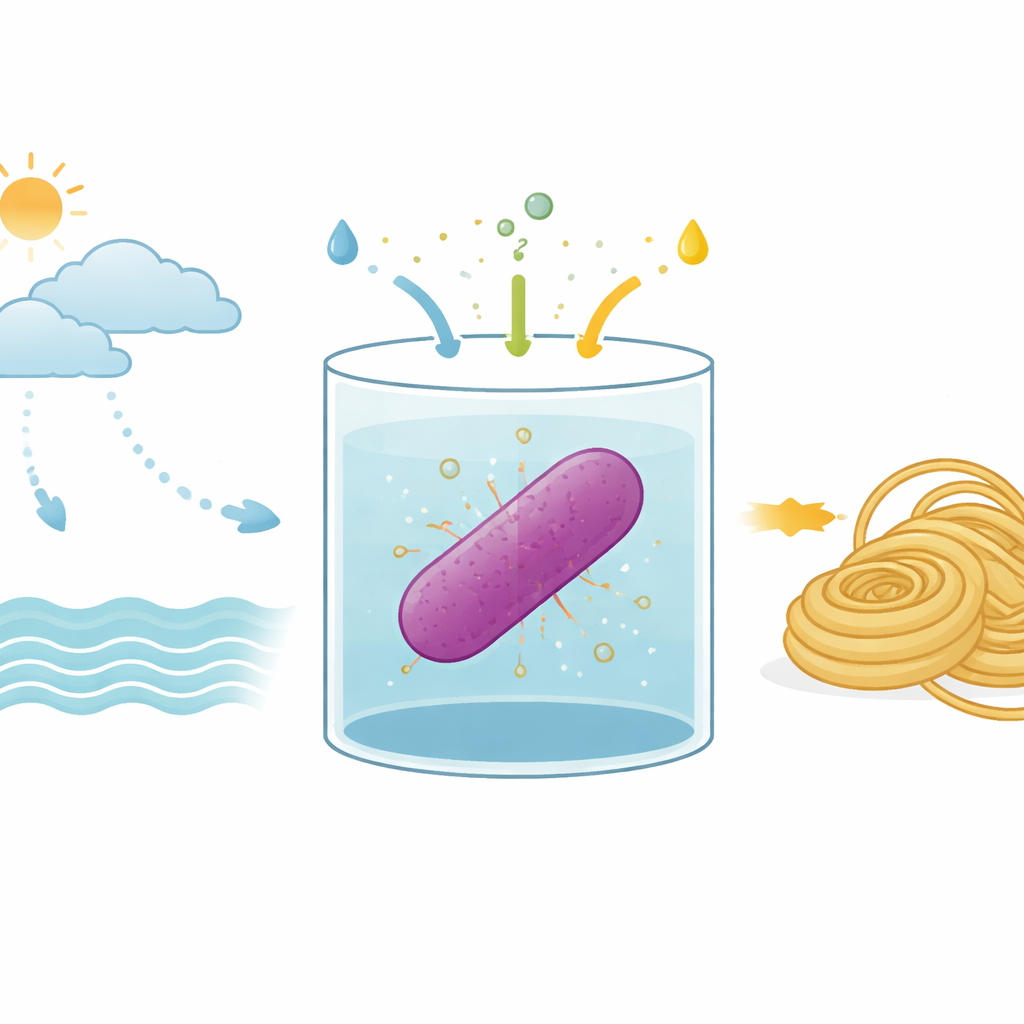
The researchers work with a microbe called Rhodovulum sulfidophilum, a purple bacterium that lives in seawater and harvests energy from light. Unlike plants, it does not release oxygen but still performs a form of photosynthesis. It can also capture nitrogen gas from the air, turning it into the nitrogen‑rich molecules life needs. Because it thrives in salty water and can use simple inorganic substances, this bacterium is a promising platform for making useful products without competing with agriculture for fresh water or fertile land.
Why Spider Silk Is So Appealing
Spider silk has long fascinated scientists and engineers because it is both very strong and remarkably flexible. The dragline silk used by spiders in their webs is built from special proteins called spidroins. In this study, the focus is on one such protein, MaSp2. In nature, spiders dissolve these proteins at high concentration and then spin them into fibers with a complex internal structure. Since spiders cannot be farmed easily—they tend to eat each other—researchers usually turn to engineered microbes like yeast or common lab bacteria to produce silk proteins. However, those systems typically depend on sugar‑rich, nitrogen‑rich media, which limits their sustainability.
Building a Solar‑Driven Silk Workshop
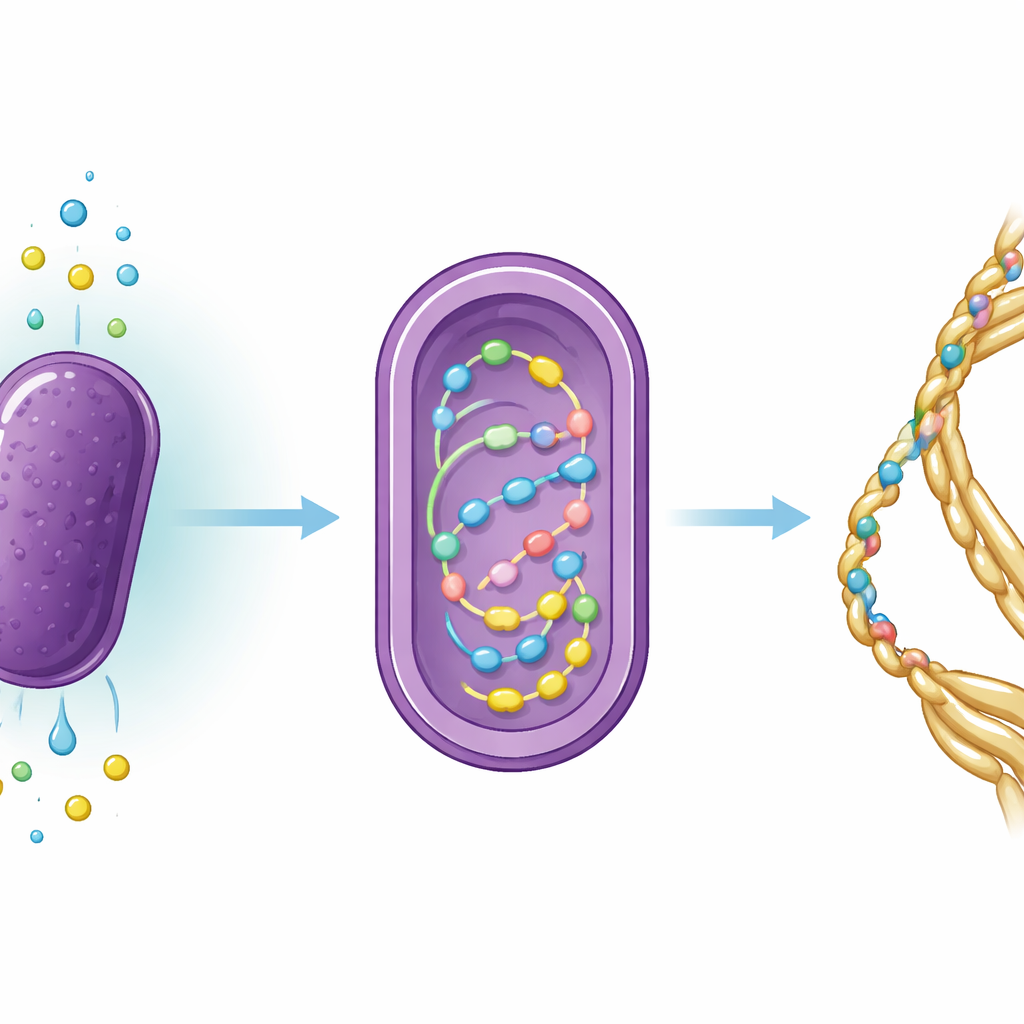
The team redesigned R. sulfidophilum so that it carries a ring of DNA encoding the MaSp2 silk protein. Then they set up growth conditions that force the bacteria to rely almost entirely on inorganic ingredients: carbon dioxide as the carbon source, nitrogen gas as the nitrogen source, and a reduced sulfur compound, sodium thiosulfate, as the electron donor that helps drive photosynthesis‑like reactions. In a 10‑liter vessel filled with artificial seawater, they bubbled in carbon dioxide and nitrogen, carefully controlled the pH so that the carbon dioxide would dissolve, and illuminated the culture with far‑red light tuned to the bacterium’s light‑harvesting machinery. When they added thiosulfate, cell growth increased markedly compared with earlier attempts that lacked this sulfur input.
Measuring Growth and Silk Output
Over several days, the researchers monitored how fast the bacteria multiplied and how much carbon and nitrogen moved from the seawater into the cells. The microbes grew for about four days before leveling off, and chemical analyses showed that they steadily took up both carbon and nitrogen. The ratio of these elements inside the cells suggested that nitrogen was in relatively short supply, hinting that the energy‑hungry step of fixing nitrogen gas was limiting overall productivity. The team then broke open the cells and used a metal‑binding purification method to isolate the engineered MaSp2 protein. Although the bacteria produced only microgram quantities of soluble silk protein per liter—tiny amounts by industrial standards—the work proved that fully inorganic, seawater‑based culture conditions can support both growth and recombinant silk production.
Pathways to Better Performance
Beyond the immediate proof of concept, the authors outline several levers that could boost yields. Improving photosynthesis, for example by adjusting light intensity or nutrient balance, could generate more of the energy needed for nitrogen fixation and amino acid synthesis. Fine‑tuning the genetic control of the silk protein—for instance, turning expression on only after cells have grown dense, or optimizing the sequence for the bacterium’s translation machinery—might also reduce stress on the cells and increase output. The bacterium’s ability to use various reduced sulfur compounds suggests that industrial waste streams containing sulfur could someday feed such cultures, further tightening the loop of resource recycling. At the same time, the authors note that large‑scale use of genetically modified marine bacteria would require strict safety measures to prevent escape into the environment.
What This Means for Future Materials
In simple terms, this study shows that it is possible to get a marine bacterium to make spider silk protein while living almost entirely on air, light, seawater, and an inorganic sulfur source. The current yields are low, but the basic pathway works, and the authors identify clear steps to make the process more efficient. If improved, this kind of solar‑driven, seawater‑based production system could offer a way to manufacture strong, flexible protein materials with a much smaller footprint, helping to decouple advanced materials from traditional agriculture and fossil resources.
Citation: Suzuki, M., Numata, K. Production of the recombinant spider silk MaSp2 protein using the marine purple photosynthetic nonsulfur bacterium Rhodovulum sulfidophilum under autotrophic conditions. NPG Asia Mater 18, 13 (2026). https://doi.org/10.1038/s41427-026-00638-7
Keywords: spider silk, photosynthetic bacteria, sustainable biomanufacturing, seawater cultivation, recombinant proteins