Clear Sky Science · en
Zinc accumulation-induced integrated stress response triggers β-cell identity loss
Why this research matters for people with diabetes
Type 2 diabetes is often explained as the body "running out" of insulin-producing cells, but many of these cells do not simply die—they change who they are. This study uncovers a surprising culprit in that identity crisis: too much zinc inside the insulin-making cells of the pancreas. By tracing what happens in both human and mouse cells, and in stem cell–derived mini–islets, the authors map out how zinc overload stresses these cells and pushes them to become the wrong cell type, and how this process might be slowed or even reversed.
When insulin cells forget their job
In healthy people, pancreatic islets contain different hormone-producing cells arranged in a delicate balance, with insulin-producing beta cells playing the starring role. In type 2 diabetes, doctors have noticed fewer beta cells and more alpha cells, which make glucagon, a hormone that raises blood sugar. The team dug into large single-cell datasets from human pancreases and found a smooth "trajectory" from typical beta cells toward alpha-like cells, including hybrid cells that carry features of both. Tissue samples from people with and without diabetes, along with diabetic mouse models, confirmed that under high blood sugar, more beta cells lose their original identity and start to look and behave like alpha cells instead of simply disappearing.
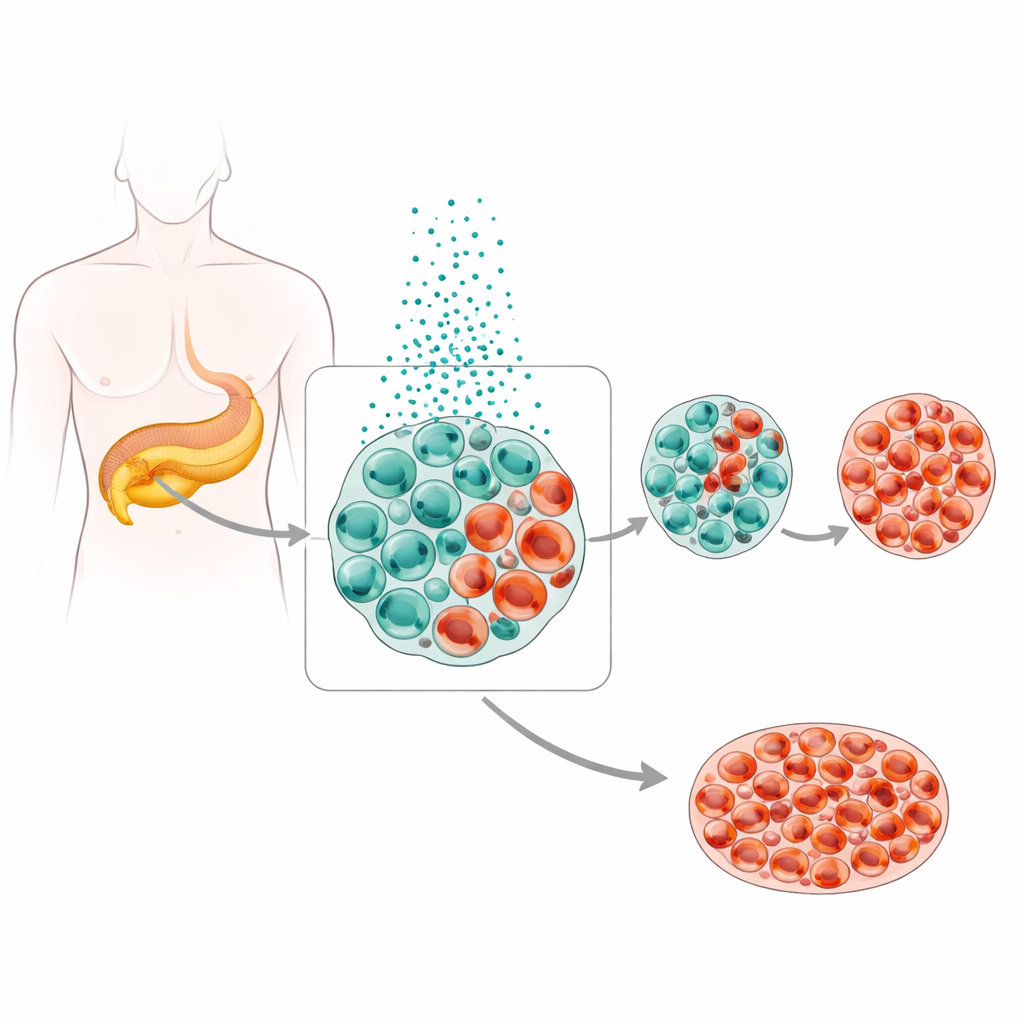
A metal with two faces
Zinc is essential for beta cells because it helps package insulin for storage and release. But these cells already carry zinc at levels far above those in most other tissues, and the study shows that pushing zinc even higher becomes dangerous. A specific transporter protein, ZnT8, normally moves zinc into insulin granules. In islets from people with type 2 diabetes and in diabetic mice, ZnT8 levels were markedly higher, and measurements of zinc inside the cells showed clear accumulation. When the researchers forced human islets to make more ZnT8, or bathed them in extra zinc or a zinc-carrying compound, beta cells shrank in number while alpha cells and mixed insulin–glucagon cells increased, both in dishes and in live mice. Genetic tricks that reduced ZnT8, by contrast, kept zinc levels lower and better preserved beta-cell identity.
Stress inside the cell: a molecular tipping point
To understand how excess zinc changes cell fate, the team turned to stem cell–derived islets, a human model they could manipulate in detail. Under prolonged high glucose, these lab-grown beta cells gradually built up zinc and began to shift toward alpha-like features. Single-cell gene profiling revealed that, as zinc rose, beta cells switched on a cellular alarm system called the integrated stress response. This pathway reduces general protein production but strongly boosts a factor called ATF4. The researchers showed that ATF4 can latch directly onto the control region of ARX, a master switch that normally defines alpha cells, and turn it on. When ATF4 or zinc were increased, ARX appeared in beta cells and the cells converted toward an alpha identity; when the stress pathway was blocked with a small molecule inhibitor, this conversion was largely prevented.
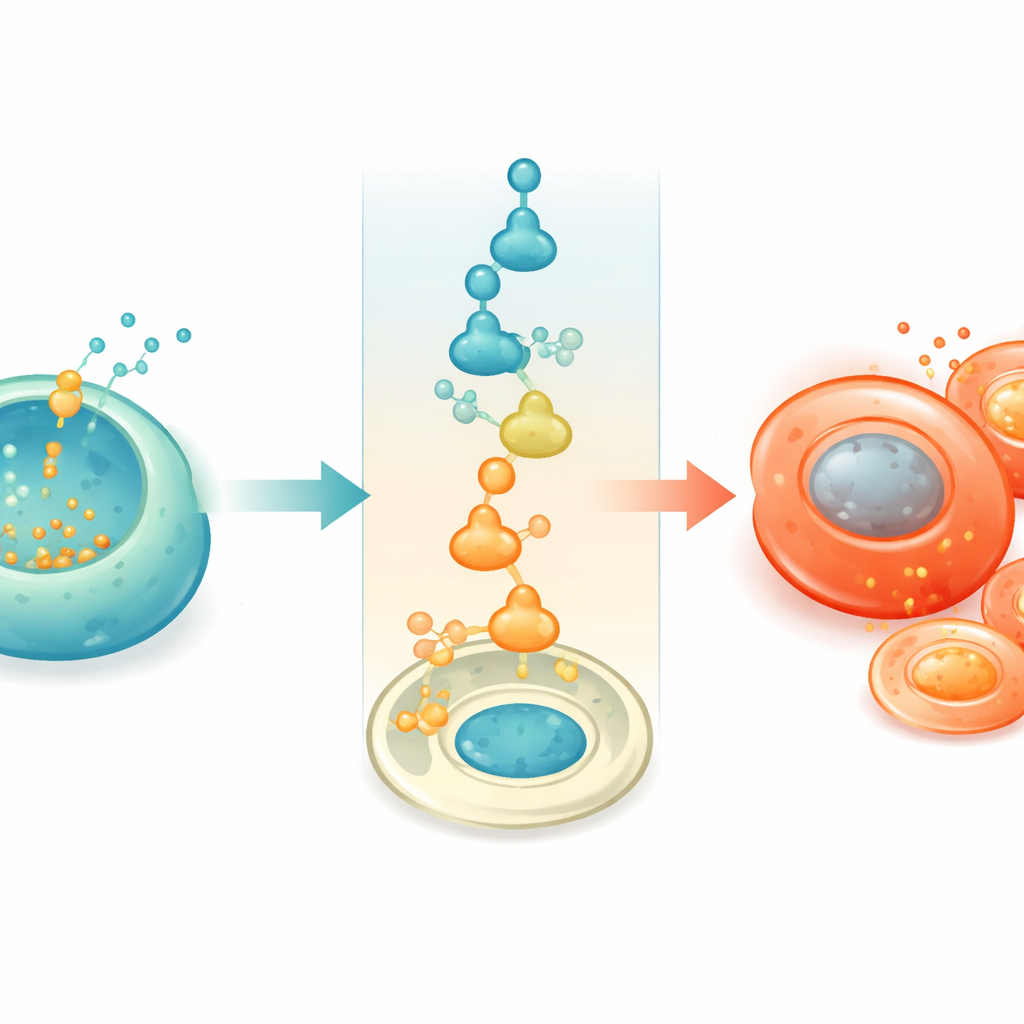
Protecting replacement cells in a diabetic body
The same zinc-driven identity loss appeared when human islets or pure stem cell–derived beta cells were transplanted into diabetic mice. In the high-sugar environment, grafted beta cells accumulated zinc, lost their defining features, and produced more glucagon-like cells, which weakened blood sugar control. Interfering with the zinc–stress axis changed this outcome. Giving mice an inhibitor of the stress response preserved more beta-like cells and improved insulin release and glucose tolerance. Likewise, using beta cells engineered to lack ZnT8, or treating cells with a low dose of the drug anisomycin that selectively reduced ZnT8 and zinc build-up, helped maintain beta-cell identity and made transplanted cells more effective at lowering blood sugar, all without obvious toxicity in the animals studied.
What this means for future diabetes treatments
This work paints a clear picture: in type 2 diabetes, chronic high blood sugar encourages beta cells to hoard zinc, which flips on an internal stress system that activates an alpha-cell gene program. Instead of dying, many beta cells are essentially reprogrammed into the wrong hormone-producing type, undermining the body’s ability to control glucose. By targeting zinc handling or the downstream stress response, it may be possible to keep beta cells "on script," both in a patient’s own pancreas and in transplanted or stem cell–derived cells used for therapy. While much work remains before these strategies reach the clinic, the study offers a concrete molecular roadmap for protecting the cells that make insulin.
Citation: Ma, Q., Xu, W., Wang, X. et al. Zinc accumulation-induced integrated stress response triggers β-cell identity loss. Cell Res 36, 359–376 (2026). https://doi.org/10.1038/s41422-026-01222-y
Keywords: type 2 diabetes, beta-cell identity, zinc accumulation, pancreatic islets, stem cell islets