Clear Sky Science · en
MEKK3 bridges gut-brain communication and cerebral cavernous malformation pathogenesis
Why the Gut Might Matter to Brain Bleeds
Cerebral cavernous malformations are clusters of fragile blood vessels in the brain that can leak or bleed, causing seizures, headaches, and strokes. This review article explores a surprising twist: tiny microbes in our intestines and the chemical signals they release may influence whether these abnormal vessels form or worsen. By following the chain from gut bacteria to blood to brain, the authors highlight a signaling hub called MEKK3 inside blood vessel cells as a key relay point that may turn gut disturbances into dangerous brain lesions.
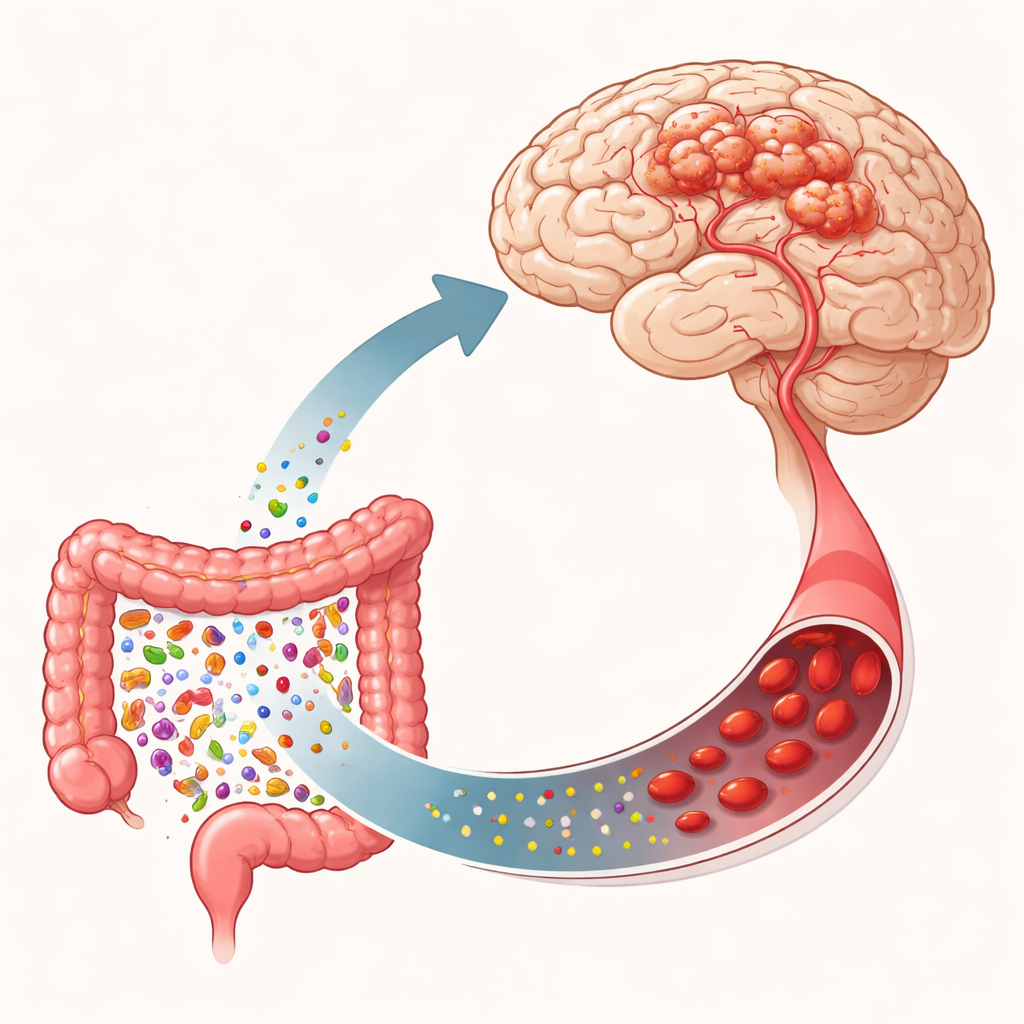
Hidden Weak Spots in Brain Blood Vessels
Cerebral cavernous malformations (CCM) affect about one in a few hundred people, though many never know they have them. These malformations are made of thin-walled, balloon-like vessels that can rupture. Some cases are sporadic, appearing as single lesions, while others run in families and involve multiple lesions. Inherited forms usually stem from damaging changes in one of three genes (often called CCM1, CCM2, and CCM3) that help keep the lining of blood vessels tight and stable. When these genes fail, the junctions between vessel cells loosen, vessels widen abnormally, and cavern-like tangles can form. Additional mutations in growth pathways, such as PIK3CA, can drive these fragile cells to multiply and remodel, making lesions larger and more aggressive.
A Cellular Switchboard Called MEKK3
At the center of this story is MEKK3, a protein inside blood vessel cells that acts like a switchboard for many incoming signals. Genetic studies show that specific changes in the MEKK3 gene (MAP3K3) are enough to trigger cavernous malformations in animal models, defining a distinct subgroup of patients. MEKK3 activity controls other factors, KLF2 and KLF4, which in turn influence how tightly cells stick together, how much they proliferate, and how leaky the vessel wall becomes. When MEKK3 is overactive, junctions between endothelial cells weaken, the barrier that normally separates blood from brain tissue becomes porous, and malformations are more likely to grow and bleed. MEKK3 does not act in isolation; it intersects with growth signals like VEGF and PI3K as well as pathways that respond to physical blood flow and inflammation.
How the Gut Talks to the Brain
The gut–brain axis is the communication network that connects the intestine to the nervous system through nerves, immune cells, hormones, and microbial chemicals. This review emphasizes one route in particular: the movement of microbial products from the intestine into the bloodstream, and then to the brain’s blood vessels. When the gut community becomes imbalanced, certain bacteria release lipopolysaccharide (LPS), a component of their outer membrane, into the circulation. LPS can loosen the intestinal barrier, further increasing its own passage into blood. Once in the brain’s vessels, LPS activates a surface sensor called TLR4 on endothelial cells, which then triggers MEKK3 and downstream inflammatory cascades. The result is more vessel leakage, immune cell recruitment, and progression of CCM lesions. Other gut-derived molecules, such as short-chain fatty acids, trimethylamine N-oxide (TMAO), and indole-related compounds, can either support barrier health or fuel inflammation, depending on their balance.
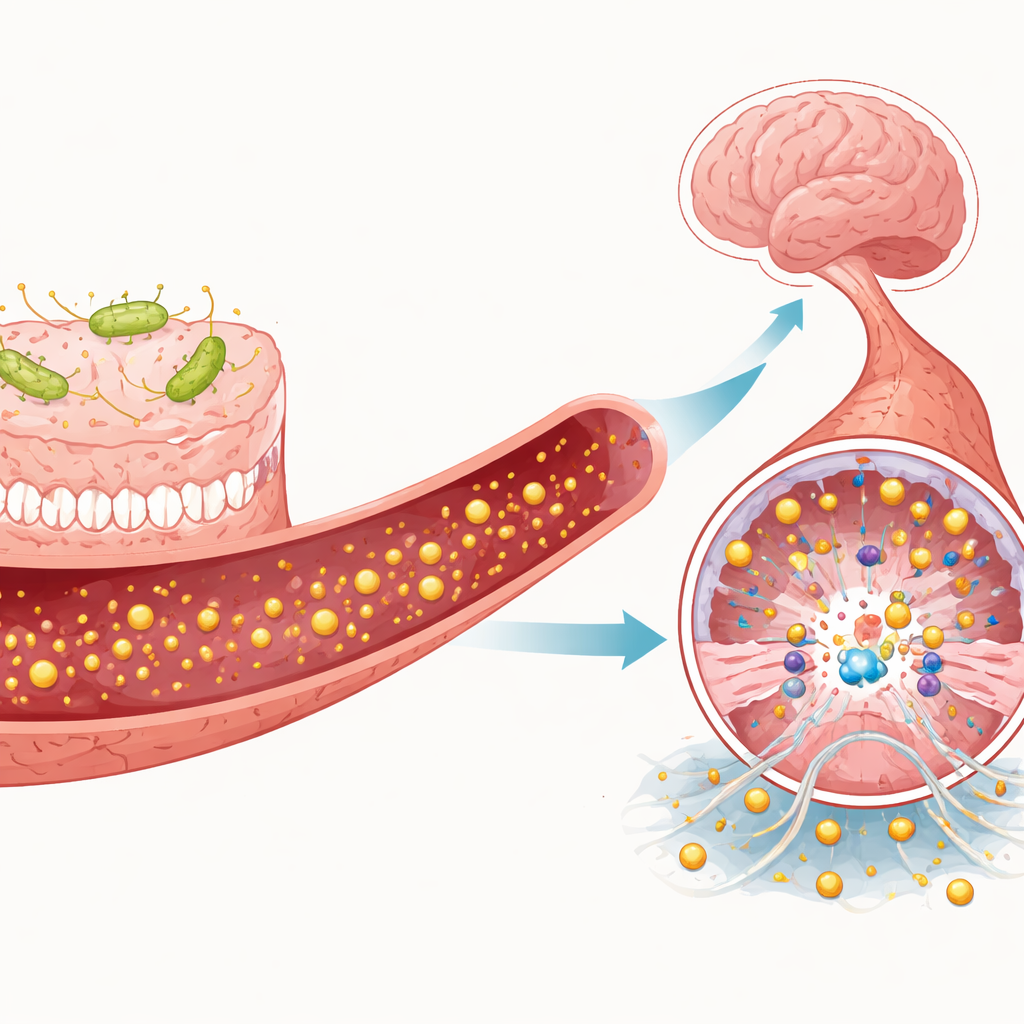
Helpful and Harmful Signals from Microbes and Immune Cells
The article details how individual gut metabolites and immune cells can tip MEKK3 signaling toward damage or protection. Short-chain fatty acids like butyrate, produced when microbes digest dietary fiber, appear to strengthen the blood–brain barrier and may indirectly dampen TLR4 and MEKK3 activity. In contrast, TMAO and certain indole derivatives, such as indoxyl sulfate, promote inflammation, oxidative stress, and stronger TLR4 signaling, which may heighten MEKK3 activation. Immune cells also play a role. Neutrophils can release sticky extracellular webs that further stimulate TLR4 on vessel cells, while pro-inflammatory macrophages and T helper cells enhance MEKK3-linked pathways; anti-inflammatory counterparts may restrain them. Together, these signals create a dynamic tug-of-war over whether brain vessels remain stable or deteriorate into cavernous lesions.
New Paths Toward Gentler Treatments
Current treatments for cerebral cavernous malformations are limited, often relying on surgery or precise radiation for high-risk lesions. The authors argue that understanding how gut microbes and their products feed into MEKK3-centered signaling opens up new, less invasive possibilities. In principle, future therapies might combine microbiome-directed strategies—such as diet, probiotics, or targeted reduction of harmful metabolites—with drugs that temper TLR4 or MEKK3 activity in brain blood vessels. For a lay reader, the takeaway is that problems in the gut can have far-reaching effects on brain blood vessel health, and that a single cellular “hub” like MEKK3 could be the key to turning down this harmful conversation and preventing dangerous brain bleeds.
Citation: Cheng, P., Han, H., Huang, Y. et al. MEKK3 bridges gut-brain communication and cerebral cavernous malformation pathogenesis. Cell Death Discov. 12, 197 (2026). https://doi.org/10.1038/s41420-026-03062-6
Keywords: cerebral cavernous malformation, gut-brain axis, MEKK3 signaling, gut microbiota metabolites, neurovascular inflammation