Clear Sky Science · en
Noncanonical role of KDM5C in conferring bortezomib resistance via the PERK‒Nrf2 axis in multiple myeloma
Why this matters for patients and families
Multiple myeloma is a blood cancer that has become much more treatable thanks to modern drugs, especially the proteasome inhibitor bortezomib. Yet many patients eventually relapse because their cancer cells learn to resist the drug. This study uncovers a previously unrecognized way in which myeloma cells protect themselves from bortezomib, centered on a protein called KDM5C. Understanding this hidden survival trick points to new strategies for keeping treatment effective longer.
A stress problem inside antibody‑making cells
Myeloma arises from plasma cells, the body’s antibody factories. Because they produce huge amounts of protein, these cells constantly strain a cellular compartment called the endoplasmic reticulum, which folds and processes new proteins. When the workload becomes too heavy, a protective system known as the unfolded protein response is triggered. Bortezomib exploits this vulnerability by blocking protein breakdown, further overloading the system and pushing myeloma cells into lethal stress. However, cancer cells can rewire this stress machinery to survive. The authors suspected that KDM5C, a protein that usually modifies DNA‑associated proteins to control gene activity, might be one of the key switches in this rewiring.
A protein that predicts aggressive disease and poor drug response
Using large patient datasets and bone marrow samples, the researchers found that KDM5C levels were much higher in myeloma cells than in normal cells. Patients whose tumors expressed more KDM5C were more likely to have advanced disease, to have relapsed after treatment, and to have shorter overall survival. In laboratory myeloma cell lines that had been made resistant to bortezomib, KDM5C was strongly elevated. When KDM5C was reduced or deleted in these cells, their growth slowed and they became markedly more sensitive to bortezomib, both in culture dishes and in mouse tumor models. These observations position KDM5C as both a driver of myeloma progression and a molecular marker of bortezomib resistance.
How KDM5C builds a pro‑survival stress circuit
To see how KDM5C exerts this effect, the team examined which genes changed when KDM5C was shut down. Many belonged to pathways that help cells cope with stress in the endoplasmic reticulum. One gene, PERK, stood out as especially reduced. PERK is a sensor on the endoplasmic reticulum that, when activated, sends signals to a factor called Nrf2, which in turn boosts antioxidant defenses and other survival programs. The study showed that KDM5C is needed to keep PERK levels high, allowing robust activation of Nrf2. When KDM5C was removed, PERK levels dropped, Nrf2 signaling weakened, and myeloma cells were less able to buffer stress and more easily killed by bortezomib. Restoring PERK in KDM5C‑deficient cells rescued both Nrf2 activity and cell growth, proving that PERK is a crucial link between KDM5C and drug resistance.
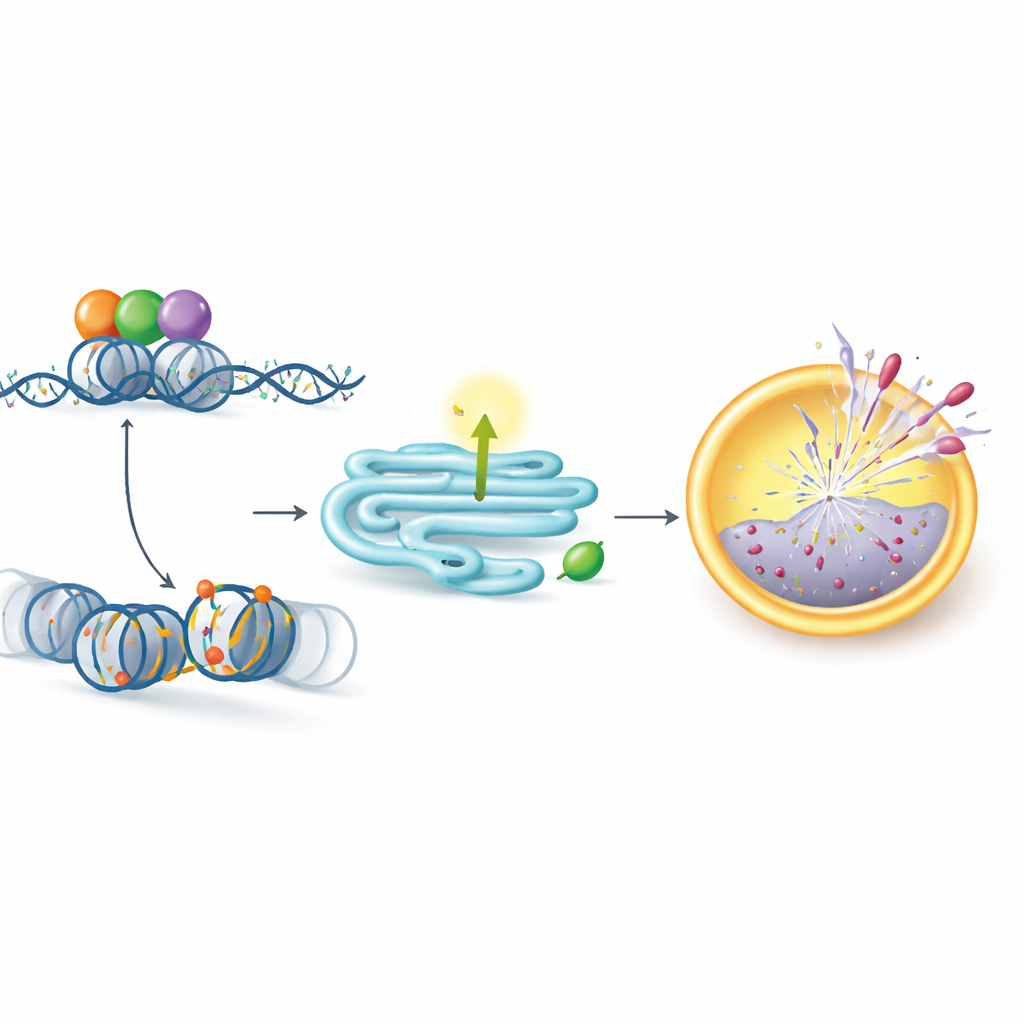
A scaffolding role that rewrites gene activity
Surprisingly, KDM5C did not act through its traditional chemical function. Instead, it behaved as a scaffold that brings together two other proteins: the master growth regulator MYC and the enzyme CBP, which places an “on” mark (acetylation) on histone proteins at key gene switches. The authors showed that KDM5C, MYC, and CBP form a large complex that binds near the PERK gene and increases acetylation at this site, turning PERK on. Removing any one of the three partners reduced this acetylation and lowered PERK expression. Mutating a specific region of KDM5C, called the PHD2 domain, disrupted its ability to assemble the complex and to activate PERK, even though the rest of the protein remained intact. Notably, a drug that blocks KDM5 enzymes’ usual activity did not affect PERK, underscoring that this resistance pathway depends on KDM5C’s structural role rather than its classic enzyme function.
What this could mean for future treatment
For people living with multiple myeloma, these findings highlight KDM5C as a promising new target to combat relapse and bortezomib resistance. By helping MYC and CBP switch on PERK and Nrf2, KDM5C lets myeloma cells ride out the severe protein stress that would normally kill them. Because this support does not rely on its conventional chemistry, therapies may need to go beyond simple enzyme inhibitors and instead eliminate the KDM5C protein itself—for example, using emerging degrader technologies. Combining such approaches with bortezomib could weaken the cancer’s stress defenses, keep treatment working longer, and ultimately improve patient outcomes.
Citation: Lu, P., Shangguan, W., Qian, W. et al. Noncanonical role of KDM5C in conferring bortezomib resistance via the PERK‒Nrf2 axis in multiple myeloma. Cell Death Dis 17, 339 (2026). https://doi.org/10.1038/s41419-026-08591-7
Keywords: multiple myeloma, bortezomib resistance, KDM5C, PERK Nrf2 pathway, epigenetic regulation