Clear Sky Science · en
Beyond immune privilege: the brain as a dynamic immunological interface
Why the Brain’s Security System Matters
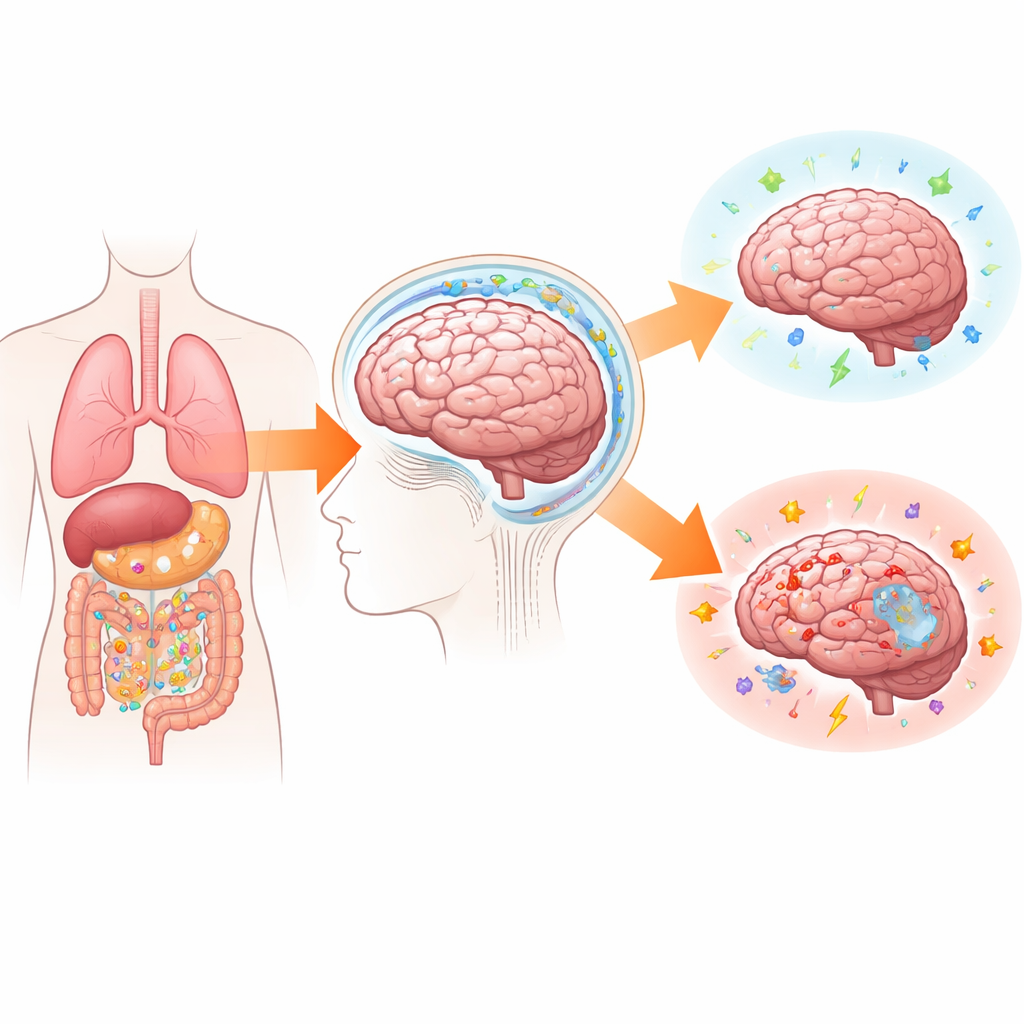
For decades, biology textbooks taught that the brain lived behind locked doors, largely invisible to the body’s immune defenses. This article overturns that simple story. It explains how the brain constantly exchanges signals, cells, and waste with the rest of the body—and how this hidden traffic shapes everything from infections and multiple sclerosis to Alzheimer’s and brain cancer. Understanding this new picture of the brain’s “border control” could lead to smarter treatments that calm harmful inflammation while boosting the body’s ability to protect and repair nerve cells.
The Brain’s Fence Is a Smart Gate
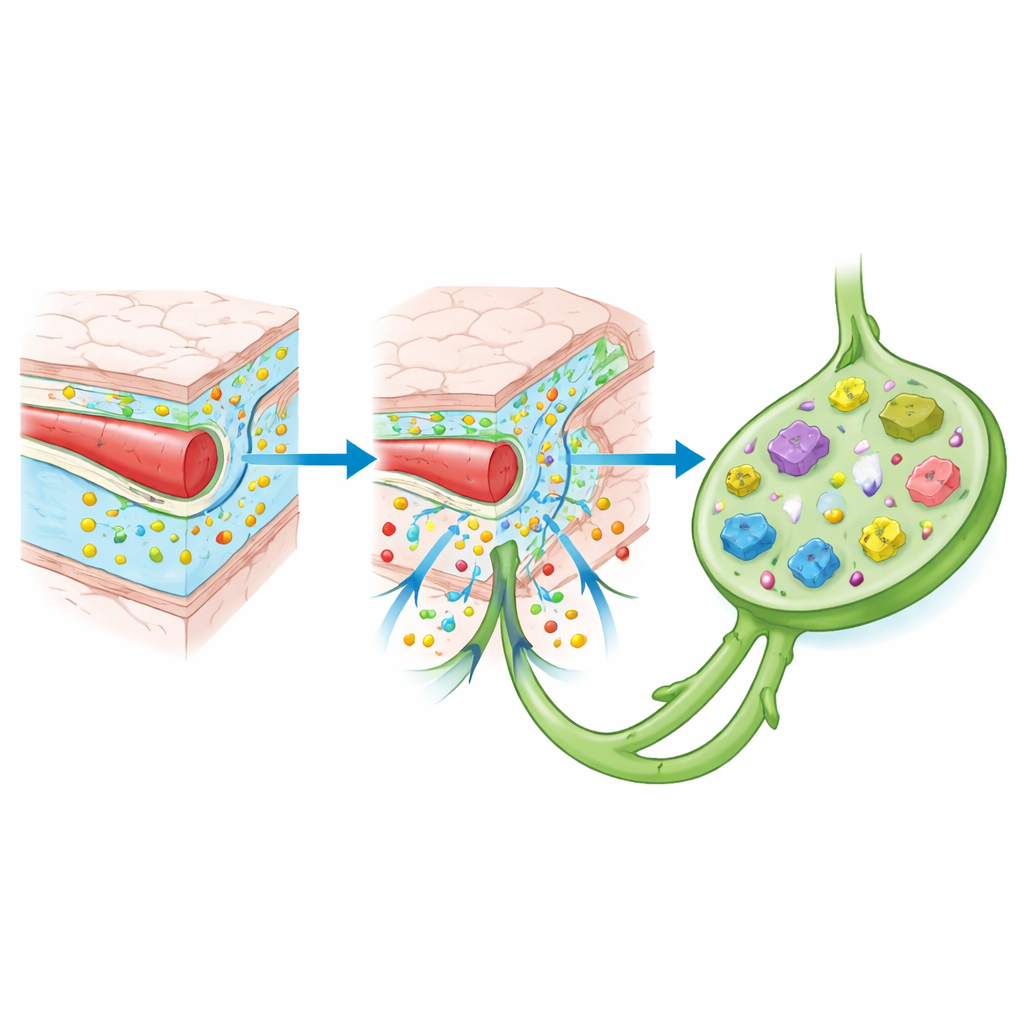
The blood–brain barrier is often compared to a fortress wall that keeps invaders out. In reality, it behaves more like a smart border crossing. Thin layers of tightly linked blood-vessel cells are wrapped by support cells that decide which molecules and immune cells may pass. When the brain is healthy, only limited traffic gets through. During infection or injury, those same cells display sticky docking sites and release chemical “scents” that guide selected immune cells inside. At the same time, a recently recognized plumbing system—the glymphatic system—flushes fluid through brain tissue, washing out waste proteins that can otherwise pile up in diseases such as Alzheimer’s. This fluid, carrying bits of debris and potential warning signals, leaves the brain along special routes that connect to the body’s immune hubs.
Hidden Drainpipes and Watch Posts
One of the most dramatic shifts in thinking came with the discovery of real lymphatic vessels—drainage channels—running in the tough outer coverings of the brain. These vessels collect fluid, immune cells, and molecular scraps from brain tissue and route them to lymph nodes in the neck, where immune cells patrol for trouble. Nearby, large blood-filled spaces in the skull lining act as “border stations” where immune cells constantly sample material arriving from the brain’s fluid. The skull itself is not just bone: its inner marrow sends immune cells directly into these coverings through tiny channels, creating a local reservoir that can respond quickly to brain distress without relying solely on cells traveling through the bloodstream.
Resident Caretakers and Long-Distance Influences
Inside the brain, resident support cells double as immune sentries. Microglia, the brain’s own immune cells, constantly extend and retract their branches, sensing damage, infection, or protein buildup. Astrocytes, star-shaped support cells, help maintain the blood–brain barrier and release chemical messengers that can either fan the flames of inflammation or help resolve it. Together, they decide whether outside immune cells are called in, and how powerful their response will be. Strikingly, the state of these cells is influenced by signals from far away: the gut microbiome, sleep patterns, aging, and general body inflammation all alter the “tone” of brain immunity by acting at these border zones.
Different Brain Neighborhoods, Different Rules
The brain is not a single uniform immune zone. The protective layers, the fluid-filled spaces, the spinal cord, and the deep brain tissue each follow slightly different rules for what can pass and how strongly they respond. Some regions flood with immune cells after injury, while others stay more guarded. The gut–brain connection adds another layer: microbes in the intestines produce small molecules that reshape immune cells in the blood and, indirectly, in the brain. Imbalances in these microbes have been linked to worsened inflammation in disorders like multiple sclerosis and Parkinson’s disease. This patchwork of compartments and outside influences means that future treatments may need to target specific brain regions or fluid routes, rather than relying only on broad, body-wide drugs.
Disease, Therapy, and a New Playbook
When this carefully tuned system goes awry, the results can be dramatic. In multiple sclerosis, misdirected immune cells cross into the brain and spinal cord, stripping insulation from nerve fibers. In infections such as meningitis or cerebral malaria, waves of immune cells and inflammatory molecules can cause swelling and tissue damage. In Alzheimer’s and Parkinson’s disease, chronic, smoldering immune activity around protein deposits may both help clean up debris and, if unchecked, harm neurons. Modern treatments already exploit this new understanding: drugs that block immune cell entry can slow multiple sclerosis, and immune-based therapies for brain tumors aim to awaken or redirect immune cells within the brain’s unique environment. The challenge is to harness helpful immune actions—clearing infections, removing toxic proteins, killing tumor cells—without tipping the balance toward lasting injury.
What This New View Means for All of Us
The article’s central message is that the brain is not an off-limits sanctuary but a tightly regulated part of the body’s overall immune network. Fluid drainage pathways, lymphatic vessels in the brain’s coverings, and resident support cells together create a system that constantly samples brain health and reports to the rest of the body. In everyday life, this system helps preserve clear thinking and stable nerve function; in disease, it can either protect or destroy. By mapping these checkpoints and learning how to nudge them—through drugs, lifestyle, or even gut microbiome changes—researchers hope to design therapies that slow neurodegeneration, improve recovery after injury, and make immune-based cancer treatments safer and more effective.
Citation: Kobeissy, F., Salzet, M. Beyond immune privilege: the brain as a dynamic immunological interface. Cell Death Dis 17, 408 (2026). https://doi.org/10.1038/s41419-026-08561-z
Keywords: brain immunity, blood–brain barrier, glymphatic system, meningeal lymphatics, neuroinflammation