Clear Sky Science · en
Neoadjuvant immunochemoradiotherapy with nivolumab, paclitaxel, and cisplatin followed by esophagectomy for locally advanced esophageal squamous cell carcinoma
Why this matters for people facing esophageal cancer
Esophageal cancer is one of the deadliest cancers worldwide, and many patients are diagnosed only after the disease has become locally advanced. Doctors already use a tough combination of chemotherapy, radiation, and surgery to try to cure these patients, but survival remains poor. This study tested whether adding a modern immunotherapy drug, nivolumab, to standard pre-surgery treatment could better shrink tumors and improve outcomes for people with a common form of the disease called esophageal squamous cell carcinoma.
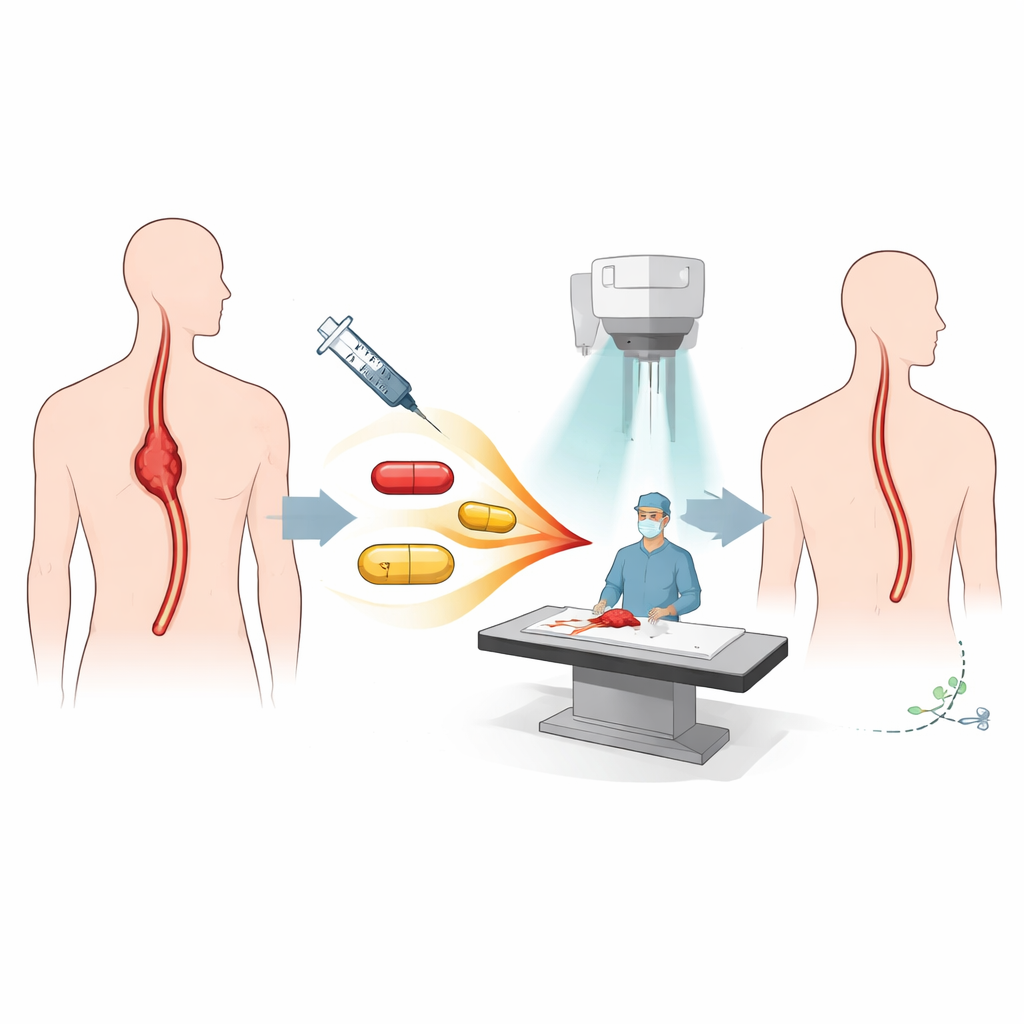
A closer look at the treatment strategy
The patients in this trial all had locally advanced esophageal squamous cell cancer confined to the chest and still considered removable by surgery. Before surgery, they received what doctors call "neoadjuvant" therapy: five weeks of chemotherapy (paclitaxel and cisplatin) given at the same time as radiation to the chest, plus four doses of nivolumab, an immune-boosting antibody drug that helps the body’s T cells attack cancer. After finishing this combined course, most patients went on to have minimally invasive esophagectomy, in which the diseased part of the esophagus is removed and the digestive tract is rebuilt.
What the researchers were hoping to see
The main measure of success was how many patients had a pathologic complete response—meaning that when surgeons removed the esophagus and lymph nodes and pathologists examined them under the microscope, no living cancer cells could be found. Previous work with chemoradiotherapy alone produces a complete response in about one quarter of such patients. The team designed the study so that if the new combination could raise that rate to about half of patients, it would be considered promising enough to pursue in a larger trial.
What actually happened in the trial
Seventeen patients were enrolled. The combined treatment was delivered largely as planned: nearly all received full-dose radiation, most received all nivolumab doses, and the chemotherapy was adjusted as needed for blood count side effects. Fourteen patients went on to surgery; three did not because their cancer progressed or other serious problems arose. Among those who had surgery, four had no remaining cancer in the removed tissue. That translates to a 24% complete response rate among all 17 treated patients—almost identical to historical results with chemoradiotherapy alone, and below the threshold needed to continue to a second stage of the trial. Survival times were also modest: half the patients saw their disease come back or progress by about a year, and half had died by just over two years.
Safety and side effects of the combination
From a safety standpoint, the intensified pre-surgery regimen was manageable but not trivial. Almost nine in ten patients had some treatment-related side effects, and about one in four developed severe drops in white blood cells from the chemotherapy and radiation. Immune-related problems from nivolumab, such as skin rash, mild colitis, or hormone gland changes, occurred in nearly half the patients but were all low grade; none required strong steroids or led directly to treatment-related death. Surgery itself remained technically feasible after the combined therapy, though several patients experienced serious surgical complications, including two with major leaks at the reconnection site of the digestive tract.
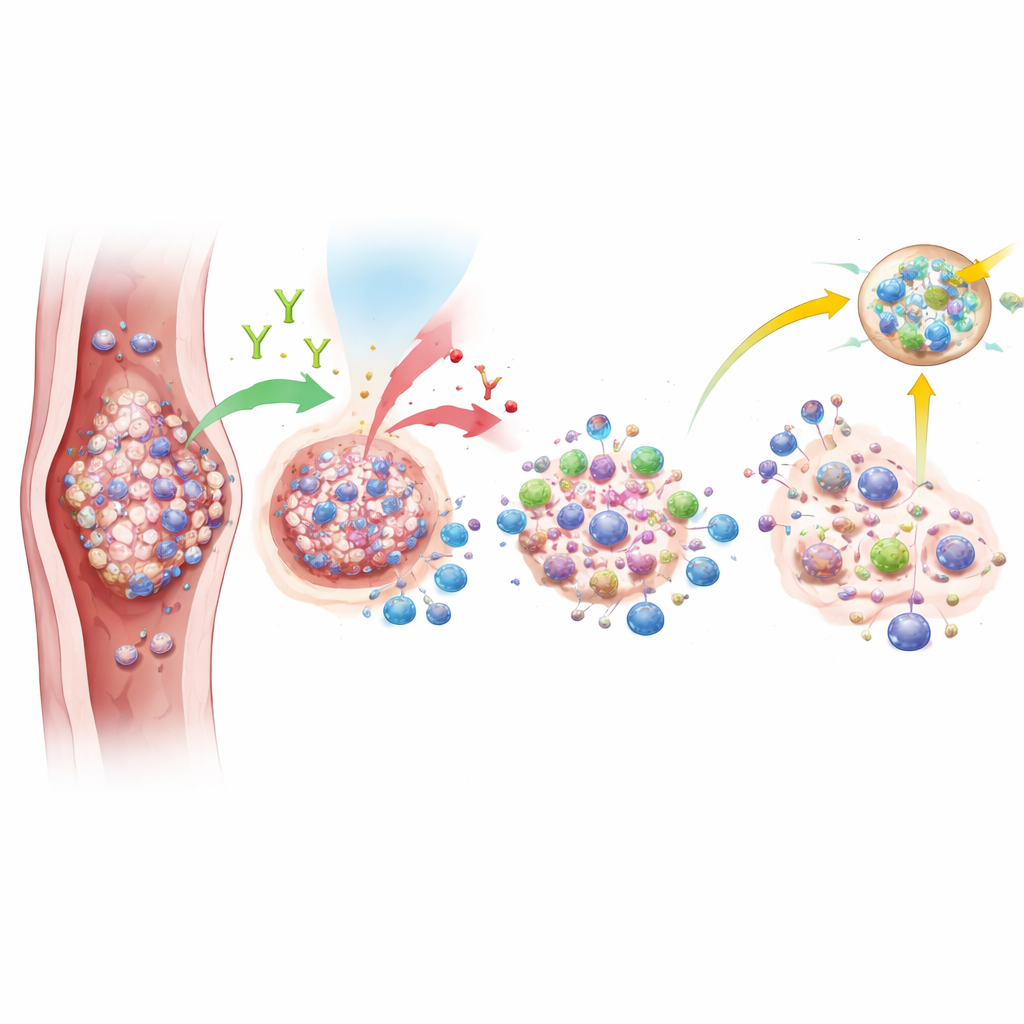
Signals from the immune system
Although the overall benefit was limited, the researchers did see intriguing clues that some patients’ tumors were far more sensitive to this approach than others. Tumor samples taken before treatment were examined for PD-L1, a protein that can indicate how visible a cancer is to the immune system, and for signs of specialized immune cell clusters called tertiary lymphoid structures. Patients whose tumors had high PD-L1 levels were much more likely to achieve complete disappearance of cancer after therapy. They also tended to live longer. Gene tests suggested that tumors which responded completely had more active immune cells and fewer suppressive cells, as well as stronger signals related to B cells and cancer-killing functions. These findings hint that certain immune features in the tumor may help predict who really benefits from adding nivolumab.
What this means going forward
For the average patient with locally advanced esophageal squamous cell cancer, this small trial suggests that simply adding nivolumab to standard chemoradiotherapy before surgery does not dramatically improve cure rates and does not yet justify changing routine practice. However, it also shows that the combination can be given safely and that a subset of patients—those whose tumors strongly express PD-L1 and show specific immune signatures—might gain more from this strategy. Larger, carefully designed trials and better tests to select likely responders will be needed to decide whether tailoring immunotherapy around these immune markers can finally move the needle for this challenging disease.
Citation: Huang, TC., Guo, JC., Lin, CC. et al. Neoadjuvant immunochemoradiotherapy with nivolumab, paclitaxel, and cisplatin followed by esophagectomy for locally advanced esophageal squamous cell carcinoma. Br J Cancer 134, 1403–1412 (2026). https://doi.org/10.1038/s41416-026-03349-6
Keywords: esophageal squamous cell carcinoma, neoadjuvant therapy, immunotherapy, chemoradiotherapy, PD-L1 biomarker