Clear Sky Science · en
BMAL1 regulates circadian rhythms via phase separation–mediated transcriptional hub formation
Why Our Internal Clocks Matter
Almost every cell in our body keeps time, quietly coordinating sleep, hunger, hormone surges, and even how we respond to medicine. At the heart of this 24-hour timing system lies a protein called BMAL1, long known as a key gear in the biological clock. This study reveals that BMAL1 does something surprising inside our cells: it gathers into tiny, liquid-like droplets that appear and disappear with time of day, and these droplets act as control hubs that help keep our daily rhythms stable.
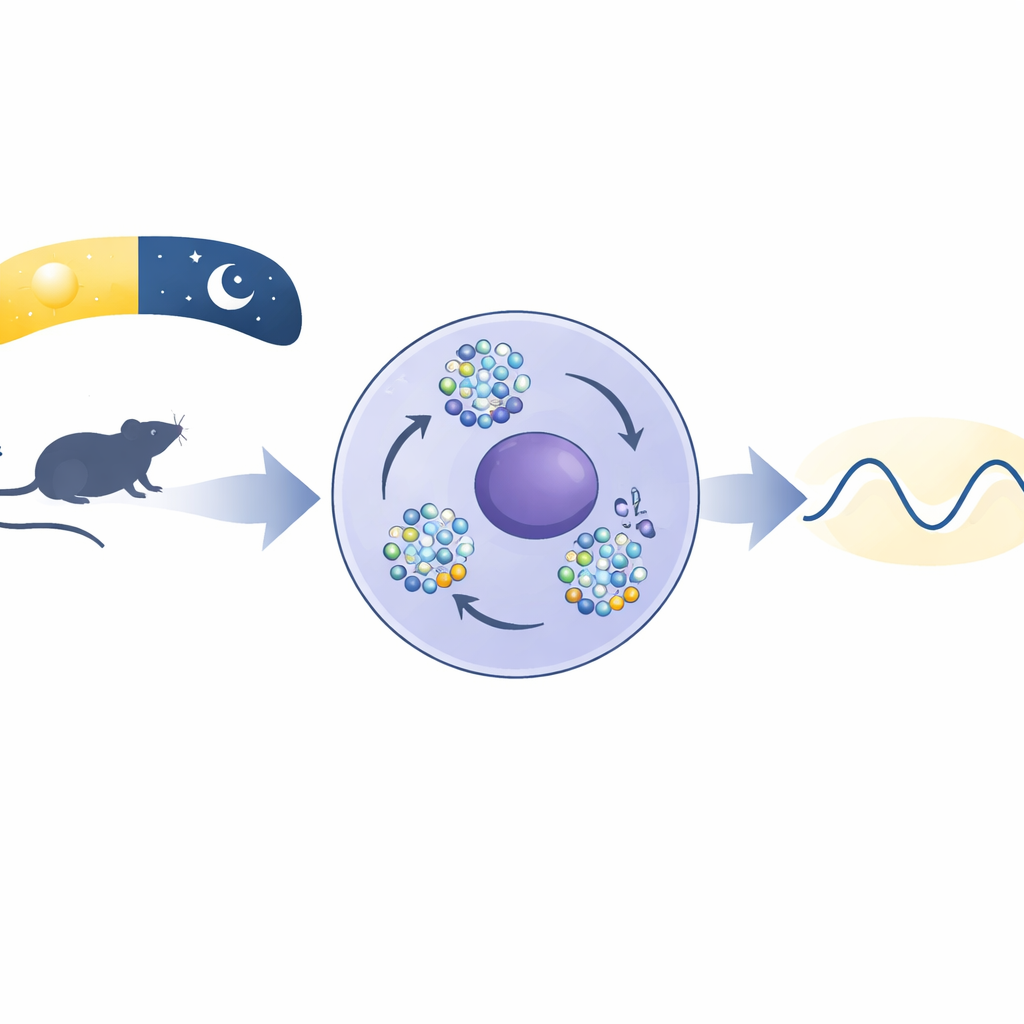
Tiny Droplets Inside the Cell
When the authors looked in mouse brain cells from the master clock region and in liver cells, they found that BMAL1 wasn’t spread evenly in the nucleus. Instead, it clustered into bright puncta—small spots that waxed and waned over the day. These spots peaked at specific times that lined up with when BMAL1 binds DNA to turn genes on. Using live-cell microscopy, the team saw the puncta merge and split like droplets of oil in water, recover their shape after being bleached by laser light, and dissolve when exposed to a chemical that disrupts weak molecular interactions. All these behaviors are hallmarks of “phase separation,” a process by which molecules self-organize into membraneless compartments.
A Flexible Tail That Switches the Droplets
To figure out what makes BMAL1 able to condense, the researchers dissected its structure. They homed in on a floppy N‑terminal stretch of about 90 amino acids that lacks a rigid shape. Such intrinsically disordered regions are known to drive droplet formation in many proteins. When this N‑terminal segment was deleted, BMAL1 lost its ability to form droplets and became diffusely spread in the nucleus. In purified protein solutions, BMAL1 formed droplets under specific salt and pH conditions, confirming that it can phase‑separate on its own. The team also showed that chemical changes—phosphorylation “marks” added to this flexible tail—tune how easily droplets appear and what they look like, making the condensates more or less stable without needing to change BMAL1’s overall amount.
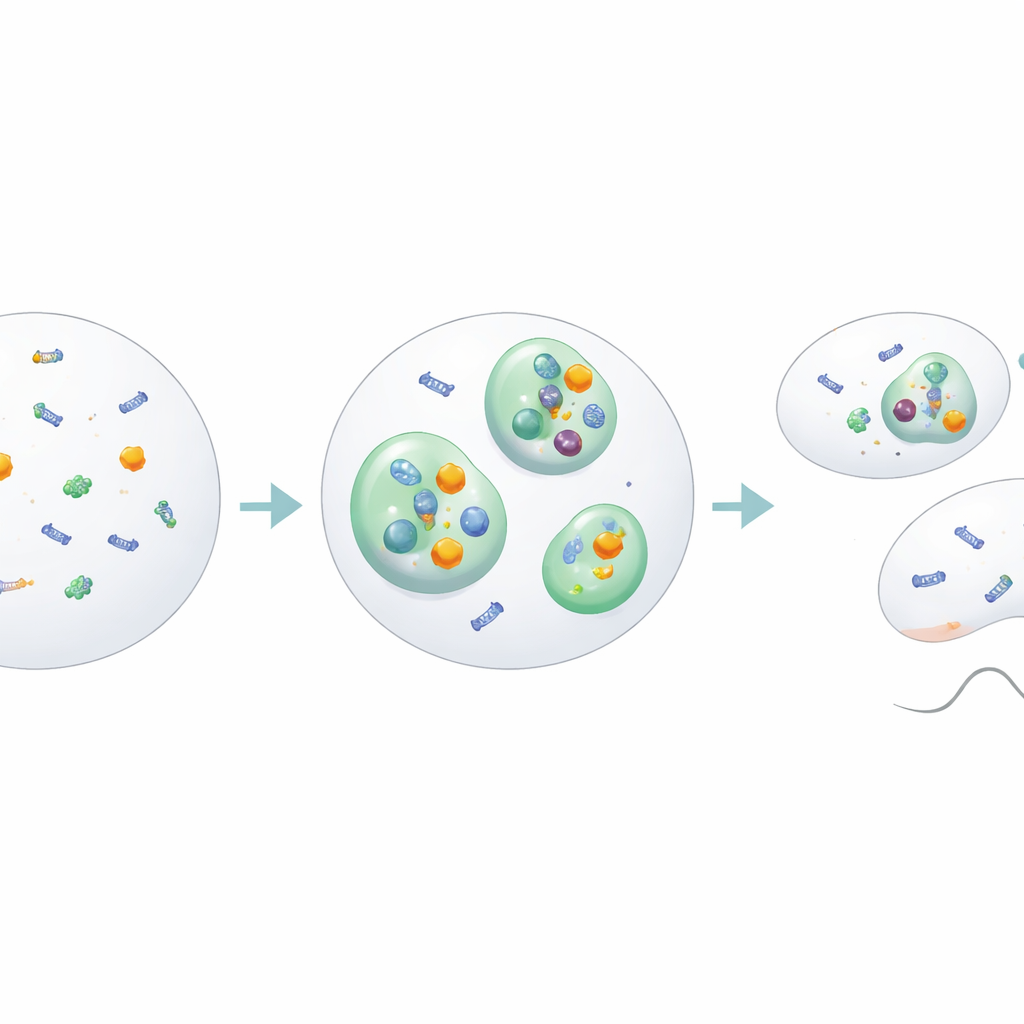
Building a Hub for Gene Control
Droplets are only interesting if they do something, and these BMAL1 condensates turned out to be busy hubs. Within the nucleus, they selectively drew in CLOCK, BMAL1’s main partner in driving circadian genes, as well as helper proteins that open up DNA and support transcription, such as the Mediator component MED1 and the co‑activator p300. Short pieces of DNA carrying BMAL1’s preferred binding sequence made droplets form more readily, suggesting that bits of the genome themselves help nucleate these hubs. At the same time, other transcription-related proteins stayed outside or around the droplets, hinting that BMAL1 condensates are specialized staging grounds where the first steps of gene activation are organized before full-scale transcription begins.
From Cells to Whole-Body Rhythms
The team then asked what happens when BMAL1 can no longer form these droplets. In cultured human cells lacking BMAL1, re‑adding the normal protein restored robust 24‑hour swings in the activity of clock genes and in newly made RNA. By contrast, a droplet‑deficient mutant missing the N‑terminal 90 amino acids left these rhythms flat, even though the protein was present. In these mutant cells, a key chromatin mark associated with active genes (H3K27ac) lost its normal daily rise and fall at circadian gene promoters, and the overall pattern of rhythmic gene regulation shifted toward more basic maintenance functions. In mice, removing BMAL1 specifically from the brain’s master clock region lengthened the animals’ daily activity period, weakened their rhythms, and altered their overall activity levels. Reintroducing normal BMAL1 rescued these behavioral rhythms, but the droplet‑deficient version did not, underlining that condensate formation is not just a microscopic curiosity but crucial for keeping the animal’s internal clock on time.
What This Means for Daily Health
Together, these findings recast BMAL1 as more than a simple on–off switch on DNA. It acts as an organizer that gathers key molecules and pieces of DNA into time‑dependent droplets, creating transcriptional “hotspots” that align gene activity with the 24‑hour day. When this droplet-forming ability is disrupted, cellular and behavioral rhythms become weak or misaligned. Understanding how such condensates shape circadian timing opens the door to future strategies that might adjust our clocks—for example, by designing drugs that slide into or dissolve specific droplets at chosen times of day—to improve sleep, metabolism, or treatment responses.
Citation: Gao, W., Zhu, L., Wei, Y. et al. BMAL1 regulates circadian rhythms via phase separation–mediated transcriptional hub formation. Sig Transduct Target Ther 11, 160 (2026). https://doi.org/10.1038/s41392-026-02711-7
Keywords: circadian rhythm, BMAL1, phase separation, biomolecular condensates, gene regulation