Clear Sky Science · en
ADAMTS4 elicits myeloid-derived immune cell recruitment and liver fibrogenesis in metabolic dysfunction-associated steatotic liver disease
Why liver scarring matters to everyday health
Metabolic dysfunction-associated steatotic liver disease (MASLD) is becoming increasingly common as waistlines grow around the world. In many people, this fat buildup in the liver quietly progresses to inflammation and scarring, eventually leading to cirrhosis, liver failure, or cancer. This study uncovers a previously underappreciated molecular player, a protein called ADAMTS4, that seems to help drive the vicious cycle of inflammation and scar formation in the fatty liver. Understanding this culprit could open the door to more precise blood tests and new treatments that interrupt liver damage before it becomes irreversible.
A hidden enzyme in the scarred liver
Researchers began by comparing liver tissue from nearly 300 people, ranging from those without MASLD to patients with advanced fatty liver inflammation and scarring. They found that levels of ADAMTS4, an enzyme that trims large proteins outside cells, rose steadily as disease worsened. The enzyme was especially abundant in activated stellate cells, a specialized cell type that normally stays quiet but, when switched on, transforms into a major producer of scar tissue. The team confirmed similar increases in ADAMTS4 in several mouse models of liver injury, whether triggered by unhealthy diets or toxic chemicals, and showed that related family members of this enzyme did not rise in the same way. 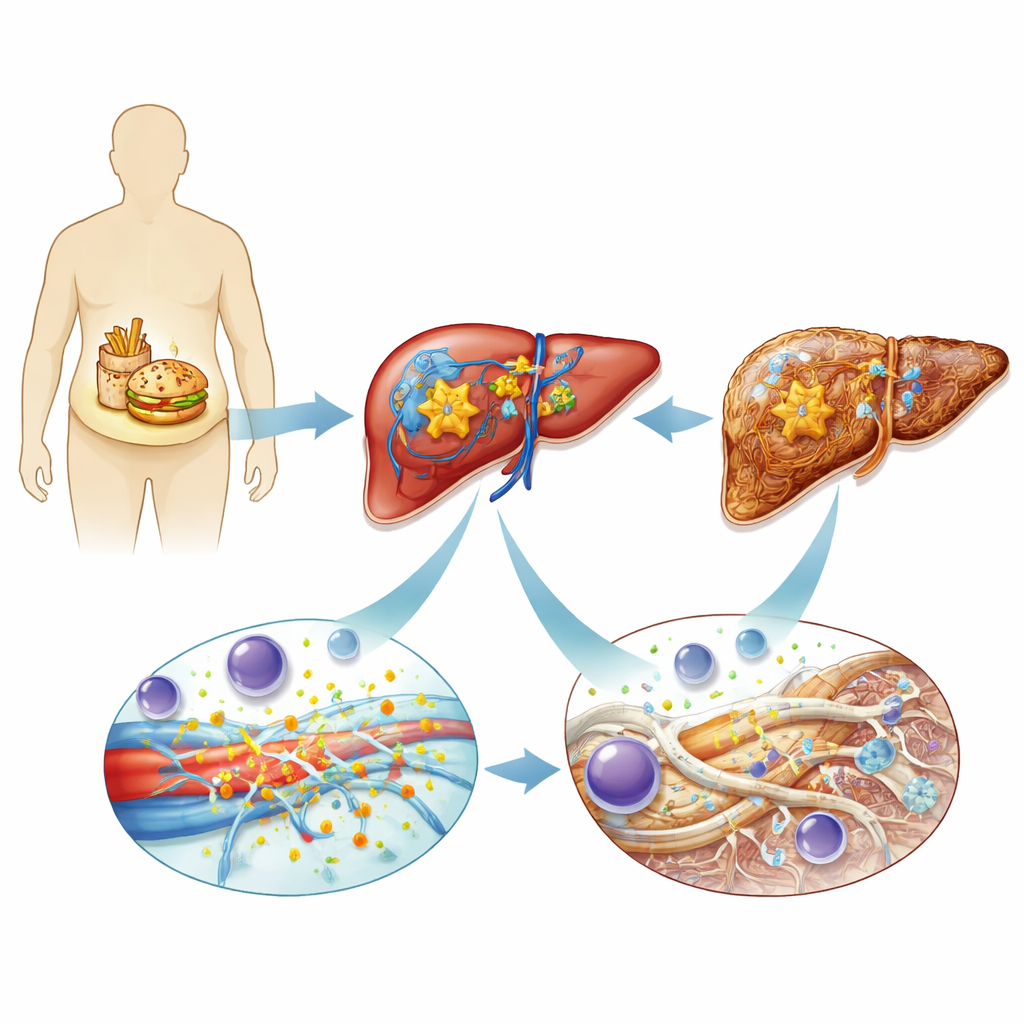
Switching off the enzyme eases liver injury
To test whether ADAMTS4 was simply a bystander or an active driver of disease, the scientists created mice lacking the gene for this enzyme. When these animals were fed fibrogenic high-fat, high-sugar diets, or exposed to chemical liver toxins, their livers showed less scarring, lower injury markers in the blood, and fewer fibrotic bands under the microscope than in normal mice. The improvement did not come from reduced fat accumulation, which remained similar, but from a marked drop in the influx of myeloid-derived immune cells, especially macrophages, which are key amplifiers of inflammation. Conversely, forcing the liver to overproduce ADAMTS4 in otherwise protected knockout mice restored heavy collagen buildup and strong inflammatory cell recruitment, underscoring that the enzyme actively promotes fibrosis.
How ADAMTS4 stirs inflammation and builds scar tissue
Diving deeper, the team probed how ADAMTS4 reshapes the liver environment. Stellate cells rich in ADAMTS4 cut a large matrix molecule called versican into smaller fragments known as versikine. These fragments, produced mainly by stellate cells rather than by other liver cells, encouraged macrophages in cell culture to move toward the damaged area and adopt a more pro-inflammatory state, especially in the presence of additional immune triggers. In parallel, the researchers discovered that ADAMTS4 can directly boost production of collagen, the main component of scar tissue, in human stellate cells by activating a signaling protein called STAT3, without relying on the classic TGF-β pathway often blamed for fibrosis. When STAT3 was blocked, ADAMTS4 could no longer raise collagen levels, linking this enzyme to both immune-cell recruitment and direct scar formation. 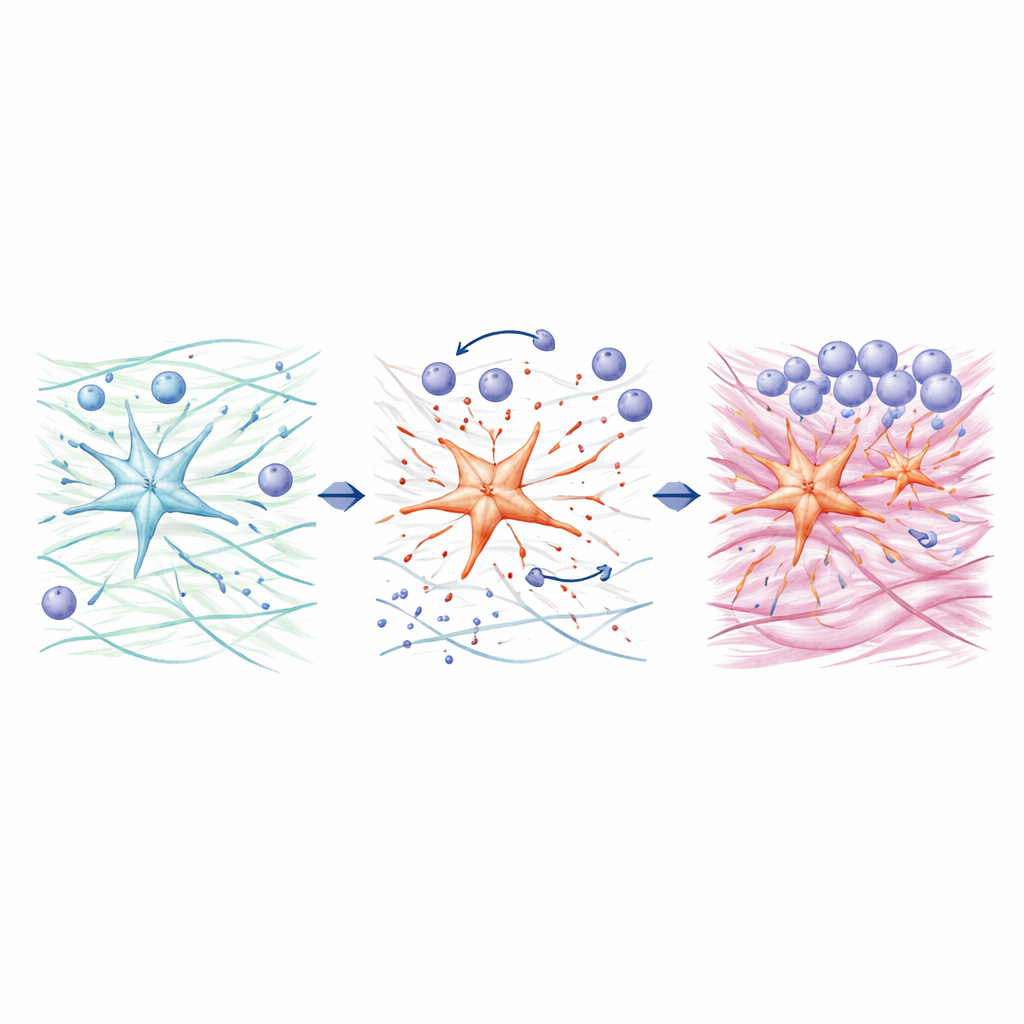
Genetic clues and who may benefit most
Not everyone with a fatty liver develops severe scarring, so the team looked for genetic variants that might tune ADAMTS4 activity. By integrating liver gene-expression data with DNA differences among patients, they identified specific single-letter changes in the genome that alter how strongly ADAMTS4 is turned on in MASLD. One variant, located upstream of the ADAMTS4 gene, was associated with particularly high enzyme levels in patients with fatty liver but not in unaffected individuals. Laboratory reporter assays showed that this DNA region can enhance gene activity in a manner that depends on which version of the variant is present. The findings hint that some people may be genetically predisposed to stronger ADAMTS4-driven responses and might benefit the most from therapies that target this enzyme.
What this means for patients
In plain terms, this work suggests that ADAMTS4 acts like a molecular accelerator pedal for liver scarring in fatty liver disease. It helps summon inflammatory immune cells and encourages scar-producing cells to lay down more collagen, tightening the organ over time. Because the enzyme shows up in blood and rises with disease severity, it might serve as part of a future blood test to flag high-risk patients. Even more importantly, drugs or antibodies designed to block ADAMTS4—or to interrupt its downstream effects on versican fragments or STAT3—could slow or stop the march from a fatty liver to cirrhosis, especially in individuals whose genetics make them particularly vulnerable.
Citation: Park, J., Kim, T., Shim, W.S. et al. ADAMTS4 elicits myeloid-derived immune cell recruitment and liver fibrogenesis in metabolic dysfunction-associated steatotic liver disease. Sig Transduct Target Ther 11, 168 (2026). https://doi.org/10.1038/s41392-026-02679-4
Keywords: fatty liver disease, liver fibrosis, ADAMTS4, immune cells, genetic risk