Clear Sky Science · en
Hypoxia inducible factor network reflects kidney disease progression in diabetes and sodium-glucose co-transporters inhibition
Why Low Oxygen in the Kidney Matters
For millions of people with type 2 diabetes, kidney damage quietly develops over years before symptoms appear. This study asks a deceptively simple question with big consequences: what happens inside kidney cells when they do not get enough oxygen, and can modern diabetes drugs nudge those cells back toward health? By following the activity of hundreds of genes controlled by the body’s oxygen-sensing machinery, the researchers built a detailed map of how diabetic kidney disease progresses and how a widely used class of drugs, SGLT2 inhibitors, may help protect the kidneys.
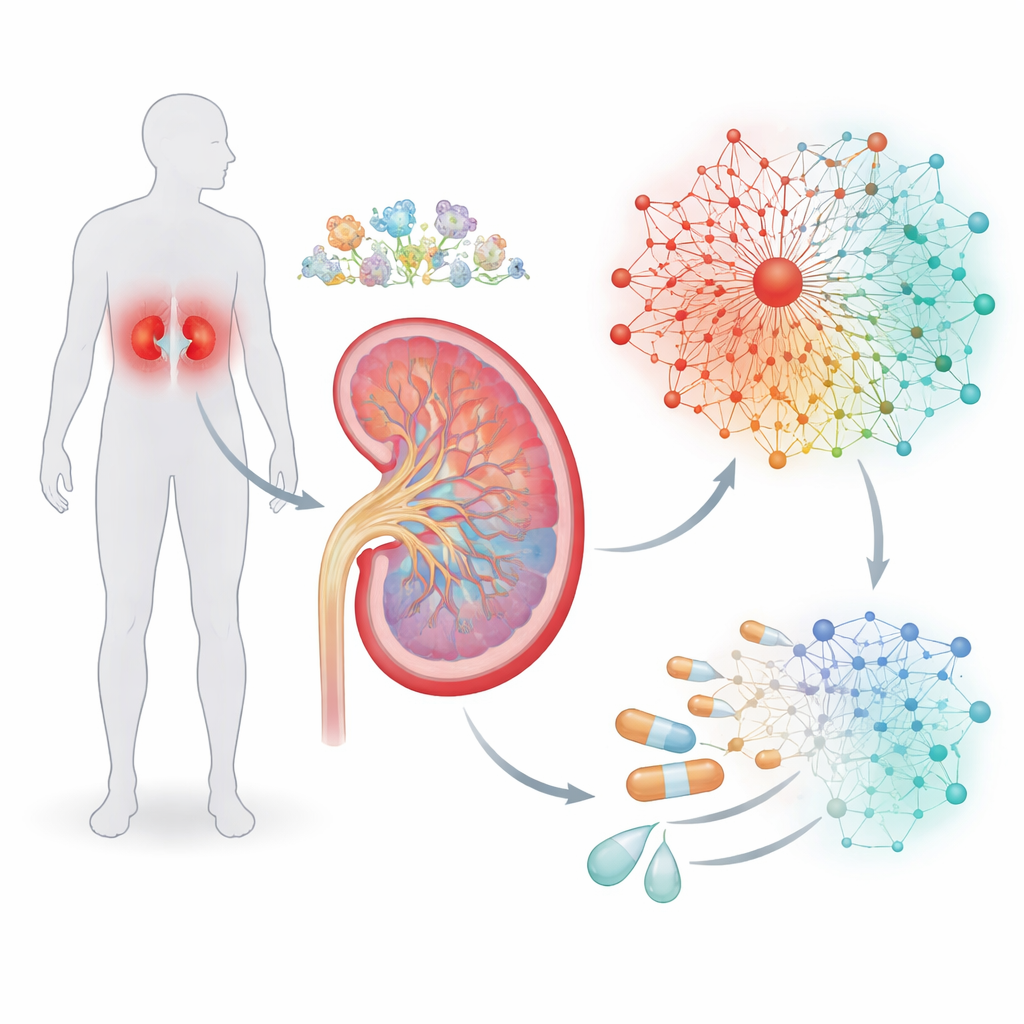
Tracing the Kidney’s Oxygen Alarm System
The kidney is one of the body’s busiest organs, constantly filtering blood and adjusting salt and water levels. That work consumes a lot of oxygen, and in diabetes the balance between oxygen supply and demand can tip toward chronic shortfall. Cells respond to low oxygen through a master switch called hypoxia-inducible factor, or HIF, which turns many other genes on or off. Instead of looking at HIF alone, the authors combed through previous studies to assemble a list of genes known to react to low oxygen. Using high-resolution single-cell RNA sequencing from kidney biopsies, they examined where in the nephron—different segments and cell types of the kidney’s filtering units—these hypoxia-responsive genes are most active.
Building a Gene Network that Mirrors Disease
Gene switches rarely act alone, so the team next asked which other regulators tend to cluster with HIF on the DNA. By scanning promoter regions—the stretches of DNA that control whether a gene is used—they searched for recurring patterns of binding sites, like grammatical phrases in a sentence. This analysis uncovered “cassettes” where HIF sites sit alongside sites for two other transcription factor families, called KLF and ETS. From these patterns, the researchers assembled seven interconnected signaling pathways containing 237 genes, forming a hypoxia-driven regulatory network. Importantly, data from a large kidney reference project showed that most of these genes lie in chromatin regions that are open and accessible, meaning kidney cells are primed to use this network when conditions demand it.
Early Warnings in Stressed Tubule Cells
The investigators then asked whether this HIF-centered network changes as kidney disease advances. They focused on proximal tubule cells, which handle much of the kidney’s energy-hungry transport work and are heavily affected in diabetes. A particular stressed state of these cells, termed adaptive proximal tubule (aPT), has been linked to poor long-term outcomes. Comparing healthy tubule cells to aPT cells, the study found that nearly half of the network genes changed their activity, often accompanied by shifts in how tightly the local DNA was packed. Similar, and progressively larger, disruptions appeared when they examined bulk tissue from people with early and late diabetic kidney disease, and spatial transcriptomics confirmed that many of these altered genes cluster in visibly injured regions of the tubules.
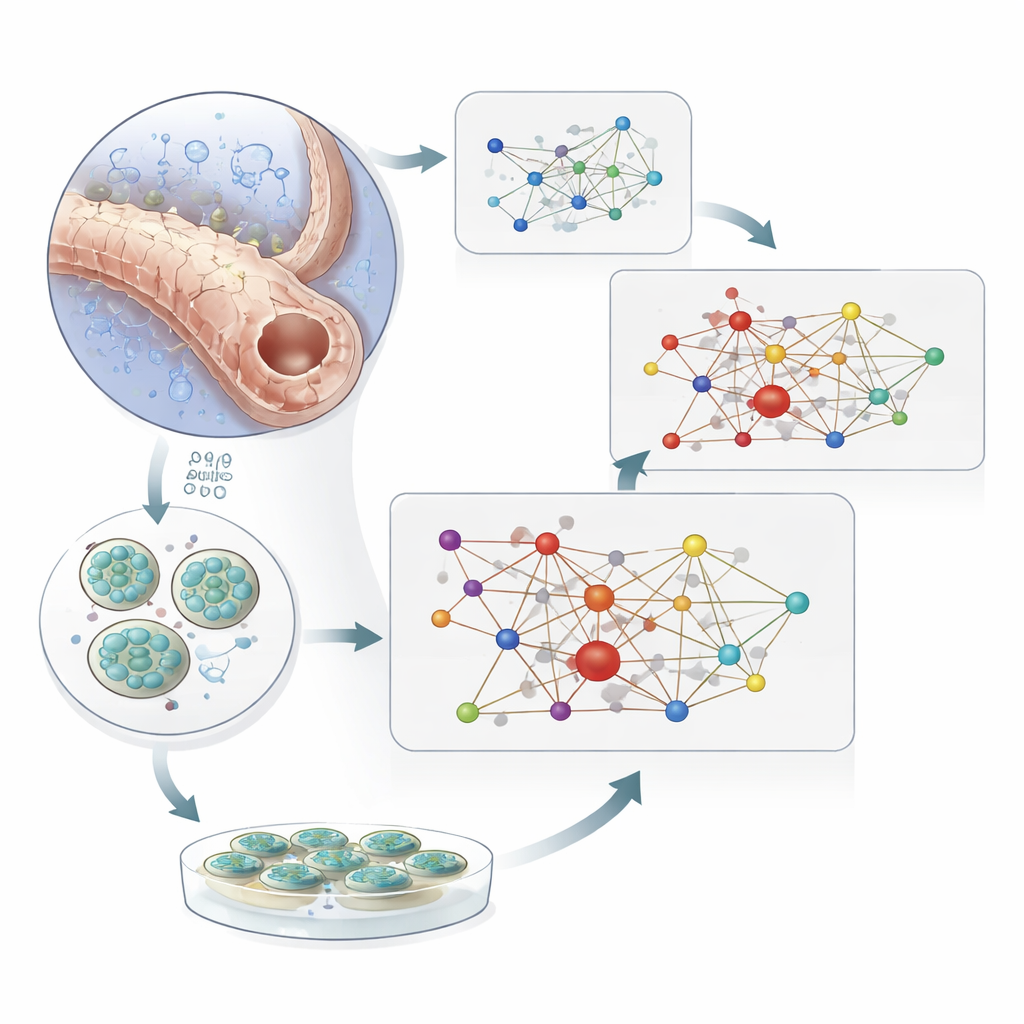
Testing the Network in Mini-Kidneys and with Drugs
To separate cause from consequence, the team turned to human kidney organoids—miniature, lab-grown kidney models derived from stem cells. When these organoids were exposed to very low oxygen, dozens of the network genes changed their expression, mirroring the patterns seen in diseased human tissue. Experiments in cultured kidney cells with HIF1A and HIF2A knocked down showed that many of these changes depend directly on HIF activity. Finally, the researchers examined people with type 2 diabetes who were or were not taking SGLT2 inhibitors, drugs that reduce the kidney’s workload by changing how sugar and salt are handled in the tubules. In proximal tubule cells from treated patients, more than a third of the network genes that had drifted away from healthy levels moved back toward a normal pattern. Similar protective shifts appeared when hypoxic organoids were exposed to an SGLT2 inhibitor.
What This Means for Patients and Therapies
Together, these findings outline a multi-gene “oxygen stress signature” that lights up early in diabetic kidneys, long before severe scarring is obvious under the microscope. Rather than relying on a single marker, the HIF-centered network provides a richer readout of how far kidney disease has progressed and how well therapies are working. The results support the idea that SGLT2 inhibitors help calm this stress network, potentially slowing damage to the tubules. While the study does not prove that lack of oxygen is the only trigger—metabolic shifts in diabetes likely play a role too—it shows that tracking this gene network may be a powerful way to monitor disease and test new drugs aimed at protecting the kidneys from the silent strain of diabetes.
Citation: Nair, V., Minakawa, A., Smith, C. et al. Hypoxia inducible factor network reflects kidney disease progression in diabetes and sodium-glucose co-transporters inhibition. Sig Transduct Target Ther 11, 144 (2026). https://doi.org/10.1038/s41392-026-02653-0
Keywords: diabetic kidney disease, hypoxia, HIF gene network, SGLT2 inhibitors, single-cell transcriptomics