Clear Sky Science · en
Sexual dimorphism of COVID-19 inspires drug repositioning and host-targeting immunotherapy for viral pneumonia
Why a Sex Hormone Matters in a Lung Infection
Why do men tend to fare worse than women when infected with COVID-19? This study tackles that question by zooming in on testosterone, the main male sex hormone, and testing how it shapes the course of coronavirus pneumonia in hamsters. By carefully adjusting hormone levels and trying an existing drug that blocks testosterone signaling, the researchers show that the balance of sex hormones can strongly influence how much virus grows in the lungs, how intense inflammation becomes, and whether animals survive. Their findings hint that some medicines already used for prostate disease and hair loss might be repurposed to treat severe viral pneumonia in people.
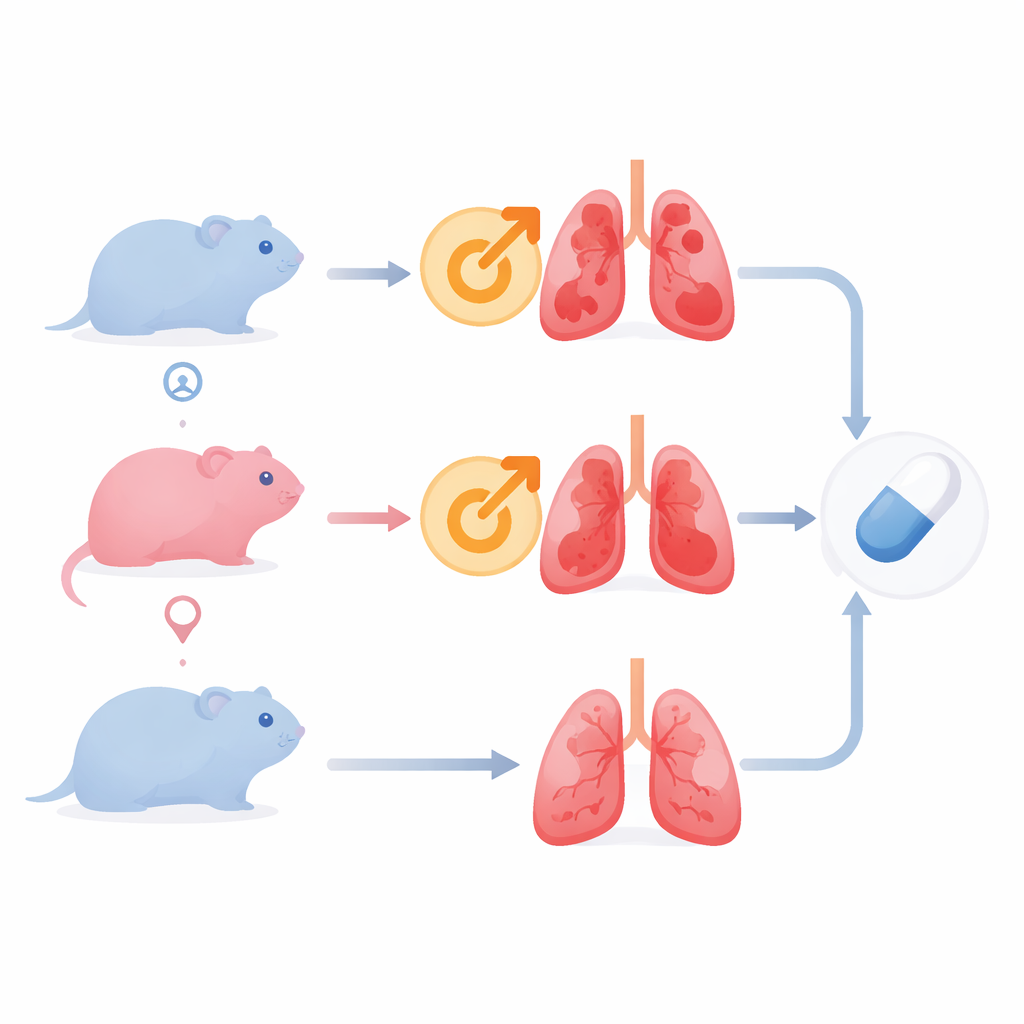
Different Outcomes for Males and Females
The team used golden Syrian hamsters, a well-established model that mimics many features of human COVID-19. They infected male and female animals with several SARS-CoV-2 strains, including early pandemic and newer variants such as Beta, Delta, and XBB. Across all viruses, male hamsters lost more weight, carried higher amounts of virus in their lungs, and developed more severe lung damage than females. As infection progressed, testosterone levels in the blood rose in both sexes, but the way these levels linked to disease differed: in males, higher testosterone went hand-in-hand with greater weight loss and higher viral load, whereas in females the pattern was reversed, hinting that the same hormone can have opposite effects depending on sex and baseline levels.
Turning the Hormone Knob Up and Down
To move from correlation to cause, the researchers actively changed testosterone levels. Surgically removing the testes of male hamsters sharply lowered their testosterone and dramatically softened the illness: castrated males had lower viral loads, less lung injury, and better weight recovery. Next, the team gave extra testosterone in graded doses to infected males and females. In males, any added testosterone made things worse in a dose-dependent fashion: more deaths, more weight loss, more virus in respiratory tissues, and more scarring and inflammation in the lungs. In females, a small testosterone boost slightly improved outcomes and reduced inflammatory signals, but higher doses flipped this benefit into harm, again increasing lung injury and viral growth. These experiments show that disease severity is tightly linked to how high and how long testosterone is pushed during infection.
Inside the Lungs: Inflammation and Immune Balance
Looking deeper, the scientists measured immune molecules and gene activity in lung tissue. In male hamsters, extra testosterone ramped up inflammatory messengers such as IL-6, TNF, and interferon-gamma, while dampening key antiviral programs that normally help cells block virus replication. Viral genetic material and live virus counts rose throughout the nose, windpipe, and lungs. Advanced RNA sequencing at both whole-tissue and single-cell levels revealed that testosterone reshaped which immune cells filled the lungs. Harmful inflammatory pathways, including IL-17 and HIF-1 signaling and cell-death programs, were more active, while beneficial antiviral defenses and certain structural cell pathways were disrupted. Importantly, when the researchers used inhaled RNA interference to tone down some of these overactive pathways, they could partly reverse the hormone-driven damage, suggesting that testosterone’s harmful effects are not fixed.
Repurposing a Common Drug
Because directly lowering testosterone by surgery is not practical in people, the study tested finasteride, a widely used drug that blocks the conversion of testosterone to a more potent form. Infected male hamsters received finasteride by mouth either starting at the time of infection or a few days later. Even short treatment courses reduced weight loss and sharply improved lung appearance under the microscope. Inflammatory molecules fell, antiviral genes became more active, and viral loads in the lungs decreased. Finasteride also altered the balance of sex hormones in the blood, lowering the strong androgen signal while modestly increasing estrogen-like activity. Gene expression analyses showed that finasteride reversed many of the testosterone-driven changes in immune cells and tissue pathways, calming inflammatory macrophages and restoring a healthier lung environment. Notably, this protective effect held across multiple SARS-CoV-2 variants, suggesting that targeting the host rather than the virus can overcome viral evolution.
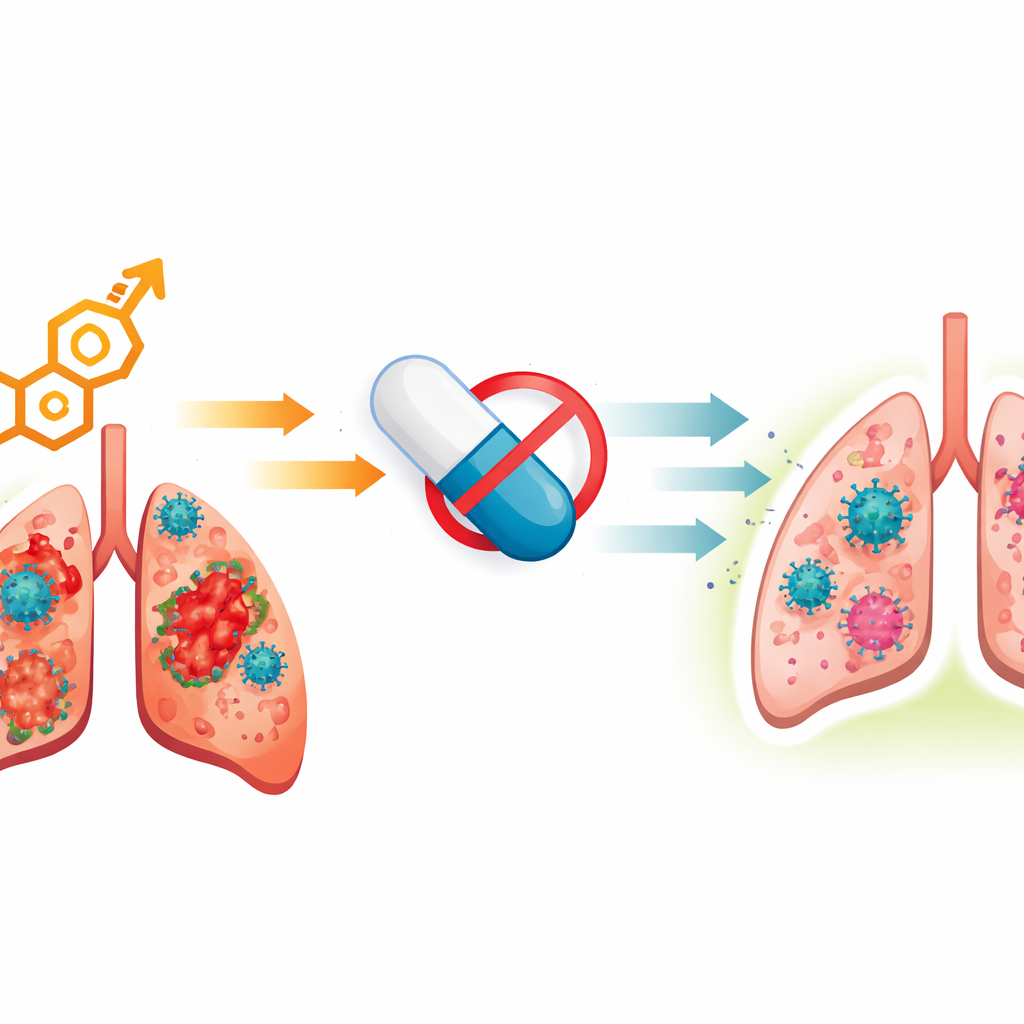
What This Means for Future Treatments
For a non-specialist, the take-home message is that sex hormones are not just background factors in COVID-19; they can actively tilt the odds toward mild illness or lethal pneumonia. In this animal work, high testosterone worsened coronavirus lung disease in males and, at high doses, in females, while blocking a key androgen pathway with an existing drug markedly improved survival and lung health. Although hamsters are not humans and finasteride has known side effects when used long term, the results support a broader idea: medications that fine-tune hormone signaling and the body’s own immune responses could become valuable additions to our toolkit against severe viral pneumonia, especially in high-risk male patients.
Citation: Yuan, L., Xiao, H., Liu, X. et al. Sexual dimorphism of COVID-19 inspires drug repositioning and host-targeting immunotherapy for viral pneumonia. Sig Transduct Target Ther 11, 147 (2026). https://doi.org/10.1038/s41392-026-02636-1
Keywords: testosterone, COVID-19, sex differences, finasteride, viral pneumonia