Clear Sky Science · en
A new integrated genetic and transcriptomic approach for investigating DUX4 and DUX4C
Genes at the Edge of Our Chromosomes
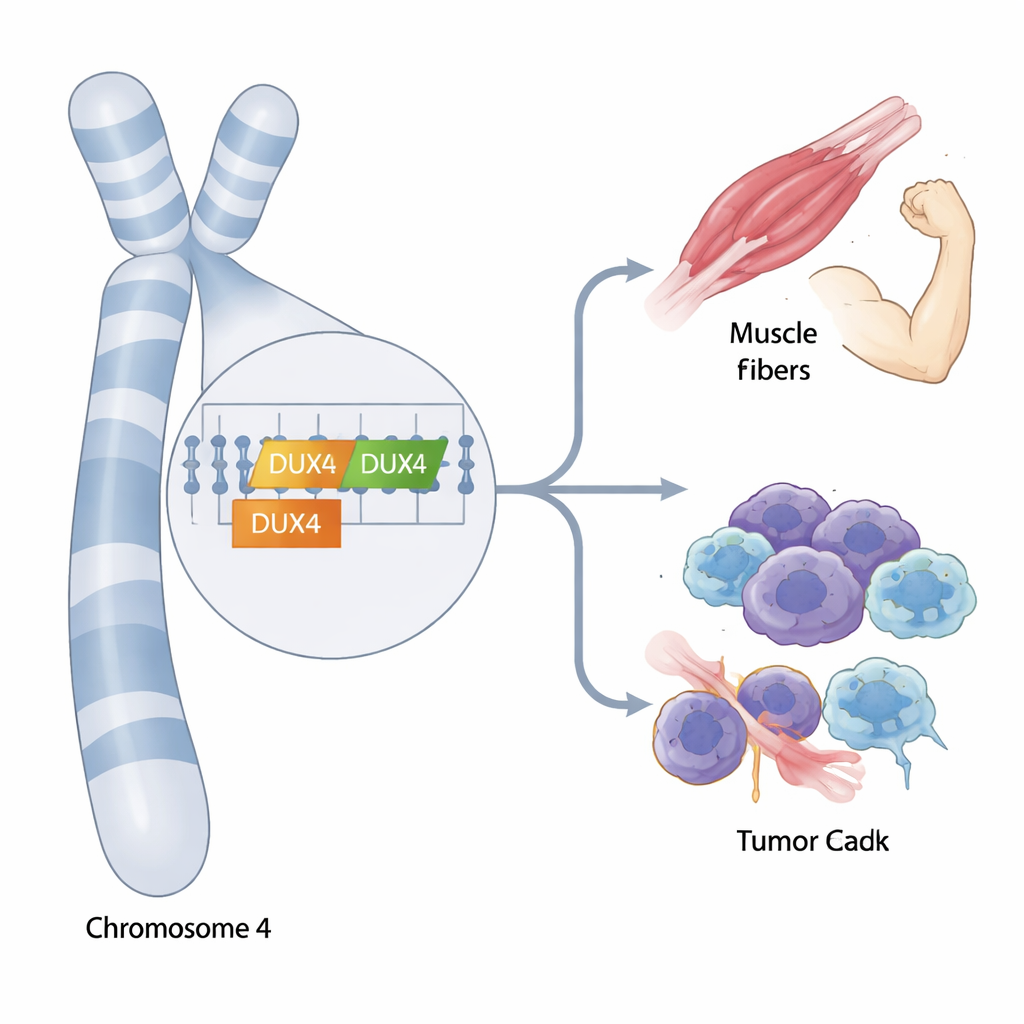
At the far ends of our chromosomes lie stretches of highly repeated DNA that are notoriously difficult to study. Two genes nestled in this maze, called DUX4 and DUX4C, have been linked to a form of muscular dystrophy and to several cancers, yet their precise roles have remained murky because standard genetic tools struggle to tell them apart. This study introduces new ways to read and interpret these confusing regions, revealing how different versions of DUX4 and DUX4C may shape disease risk and immune behavior.
Untangling Two Look-Alike Genes
DUX4 and DUX4C sit close together near the tip of chromosome 4, surrounded by dozens of nearly identical repeat units. Short-read DNA sequencing, the workhorse of modern genetics, often cannot distinguish between these repeats, between DUX4 and DUX4C, or among their many cousins scattered across the genome. To overcome this, the researchers combined long-read and short-read sequencing to reconstruct the full sequences of the most common versions, or haplotypes, of DUX4 and DUX4C. They then built a custom reference genome, called D4Ref-T2T, that replaces the confusing repeat block with clearly defined copies of each major haplotype, allowing standard sequencing data to be mapped more precisely.
Common Gene Variants and Who Carries Them
Using large public datasets from European and Japanese populations, the team identified two main DUX4C haplotypes, which they termed 4qα and 4qβ. These versions differ at a handful of DNA changes in a regulatory region near the end of the gene, and both are found at similar frequencies in Europeans and Japanese people. When they examined DUX4, they confirmed that existing reference genomes accurately represent only some of its forms, and that one widely used reference misplaces parts of the disease-associated DUX4 haplotype. With their new D4Ref-T2T reference, they could genotype both genes more reliably and discovered that the DUX4C-4qα version tends to travel together with a particular DUX4 variant, called 4qB, showing a strong genetic linkage between the two loci.
New Messages from DUX4C
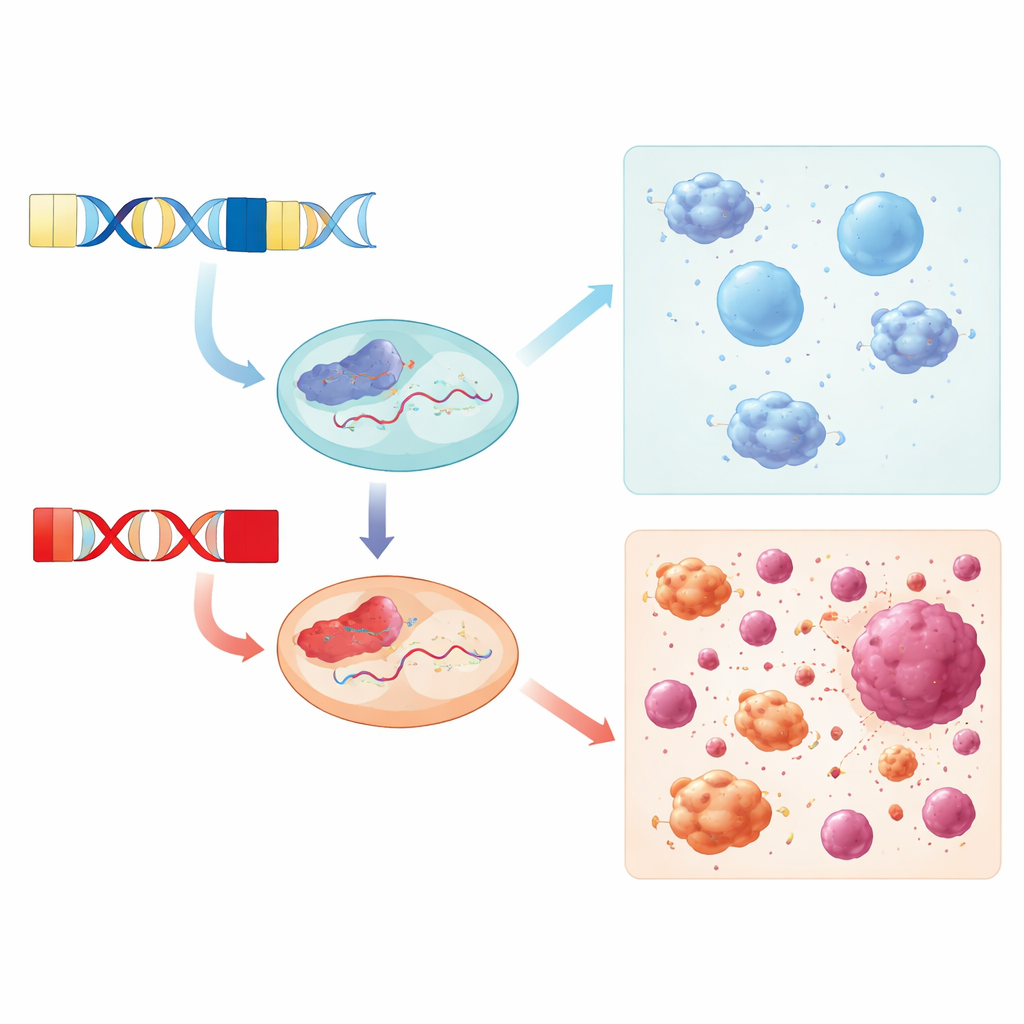
Beyond the DNA itself, the scientists explored how DUX4C is transcribed into RNA in living cells. Using long-read RNA sequencing in human immune cell lines stimulated with interferon, they reconstructed full-length DUX4C transcripts and discovered two previously uncharacterized RNA isoforms with well-defined start and end regions. These isoforms encode a protein that shares the same DNA-binding domains as DUX4 but lacks its classic activation tail. Instead, the tail of DUX4C is intrinsically disordered and packed with short sequence motifs that often serve as flexible docking sites for other proteins and signaling molecules, hinting that DUX4C may act as a regulatory hub rather than a simple on/off switch for genes.
Where and When the Genes Switch On
The team then asked when DUX4 and DUX4C are actually expressed in human tissues. By reanalyzing RNA data from breast tumors, muscle biopsies from patients with facioscapulohumeral muscular dystrophy (FSHD), and interferon-stimulated immune cell lines, they found distinct patterns. Both DUX4 and DUX4C were elevated in breast tumors compared with normal breast tissue, whereas only DUX4 was consistently increased in FSHD muscle. DUX4C was largely silent in FSHD samples, though a few unaffected family members showed modest levels, suggesting it may be less damaging to muscle than DUX4 or could even play a protective or repair-related role. In immune cell lines, neither gene was strongly induced simply by interferon treatment alone.
Hints of an Immune-Directing Role
Finally, the researchers focused on a small set of immune cell lines that differed in both their DUX4C and DUX4 haplotypes and in whether DUX4C was expressed. Cells carrying the DUX4C-4qα / DUX4-4qB combination and actively expressing DUX4C showed increased activity of genes involved in immune cell movement, such as leukocyte and lymphocyte chemotaxis. This, together with the signaling motifs found in the DUX4C protein tail, suggests DUX4C may help guide immune cell migration or activation in certain genetic backgrounds, potentially influencing how tumors and inflamed tissues are surveilled by the immune system.
Why This Work Matters
By building tailored genetic and transcriptomic maps for DUX4 and DUX4C, this study provides tools to see clearly into one of the most repetitive and error-prone corners of the human genome. The results show that different versions of these genes are tightly linked and may work in opposition: DUX4 as a known driver of muscle damage and immune dampening, and DUX4C as a possible modulator of immune signaling and cell movement. These insights open the door to better genetic diagnosis of FSHD, more accurate interpretation of cancer sequencing data, and future work on how subtle differences at the chromosome tips can tilt the balance between tissue damage, repair, and immune defense.
Citation: Zhuang, Z., Ueda, M.T., Yamaguchi, K. et al. A new integrated genetic and transcriptomic approach for investigating DUX4 and DUX4C. J Hum Genet 71, 373–381 (2026). https://doi.org/10.1038/s10038-025-01450-x
Keywords: DUX4, DUX4C, facioscapulohumeral muscular dystrophy, breast cancer, immune chemotaxis