Clear Sky Science · en
Dual solvents with different coordination strengths for localized high concentration electrolytes in lithium metal batteries
Better Batteries for Everyday Life
Lithium metal batteries promise phone charges that last for days and electric cars that drive much farther between stops. Yet these powerful batteries are held back by the liquid inside them—the electrolyte—which can slowly damage the battery’s internal parts. This paper explores a new recipe for that electrolyte so lithium metal batteries can run at higher voltages, deliver more energy, and survive hundreds of charge–discharge cycles without failing.
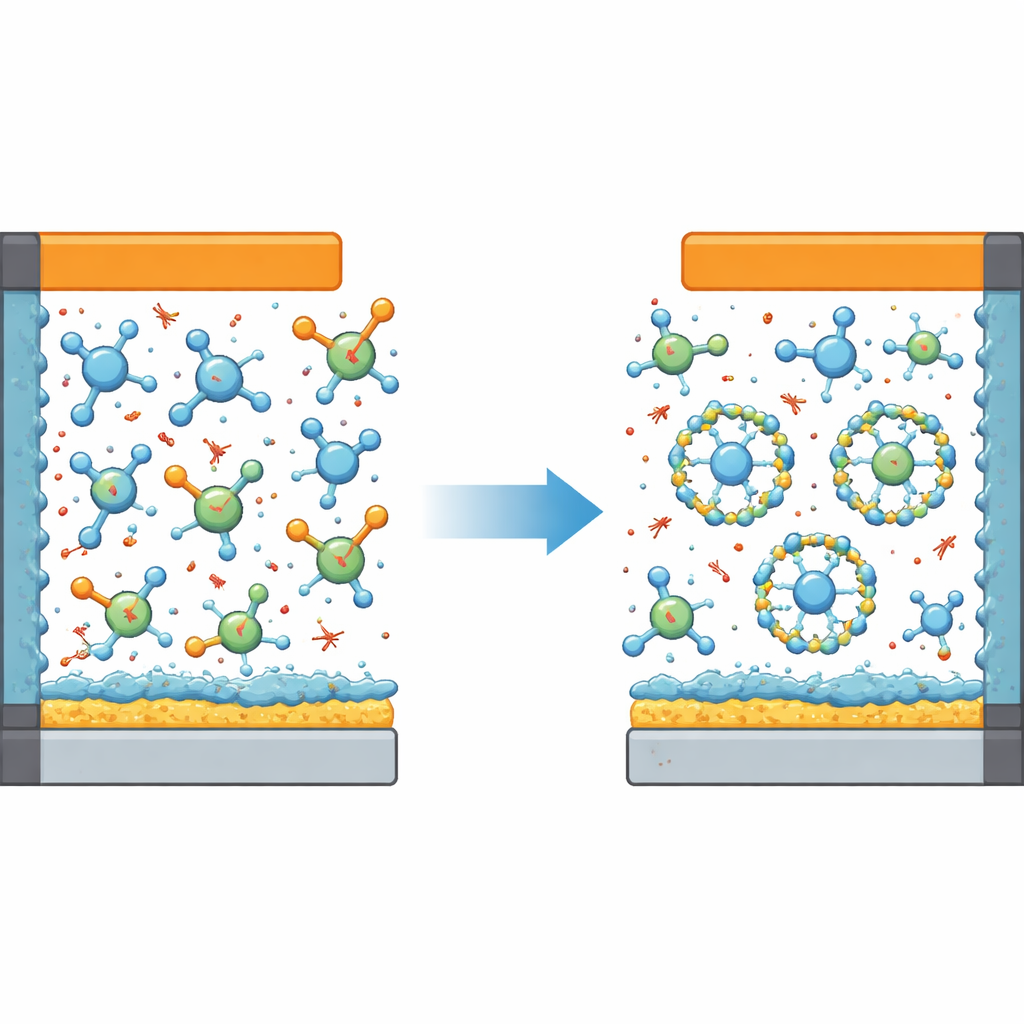
Why Lithium Metal Is So Hard to Tame
Lithium metal is an ideal battery material because it stores a lot of energy in a small, light package. But it is also highly reactive. When a battery charges and discharges, lithium metal can grow into needle‑like structures and react with the liquid around it, wasting lithium and risking short circuits. Modern designs try to control this behavior by carefully adjusting the electrolyte so it builds thin protective layers on both the lithium anode and the high‑voltage cathode. A popular approach, called a localized high‑concentration electrolyte, crowds negatively charged ions close to lithium ions, encouraging these protective films to be rich in tough, inorganic compounds such as lithium fluoride and lithium oxide.
The Hidden Problem with a Workhorse Solvent
Many of these advanced electrolytes rely on a common solvent, dimethoxyethane (DME), which holds lithium ions very well and supports fast charging. Unfortunately, DME breaks down easily at the high voltages used with energy‑dense cathodes like nickel‑rich NCM811. In conventional formulations, some DME molecules are not tightly bound to lithium; they wander freely, especially near the cathode and aluminum current collector. There, they decompose or corrode metal surfaces, eating away at capacity and shortening battery life. Simply reducing the DME content is not enough, because too little DME slows ion movement and hurts battery performance.
A Third Ingredient that Brings Order
The researchers tackled this dilemma by adding a carefully designed third liquid to the mixture: a weakly coordinating, heavily fluorinated ether called HFMTFP. Their new “ternary” electrolyte combines DME, a non‑solvating fluorinated diluent, and HFMTFP. Computer simulations and spectroscopic measurements show that HFMTFP subtly reshapes the microscopic environment around lithium ions. DME stays locked into the immediate neighborhood of lithium, while HFMTFP competes just enough to slow the constant swapping of DME molecules in and out. This energetic hierarchy suppresses the population of free DME molecules that would otherwise roam and decompose at high voltage.
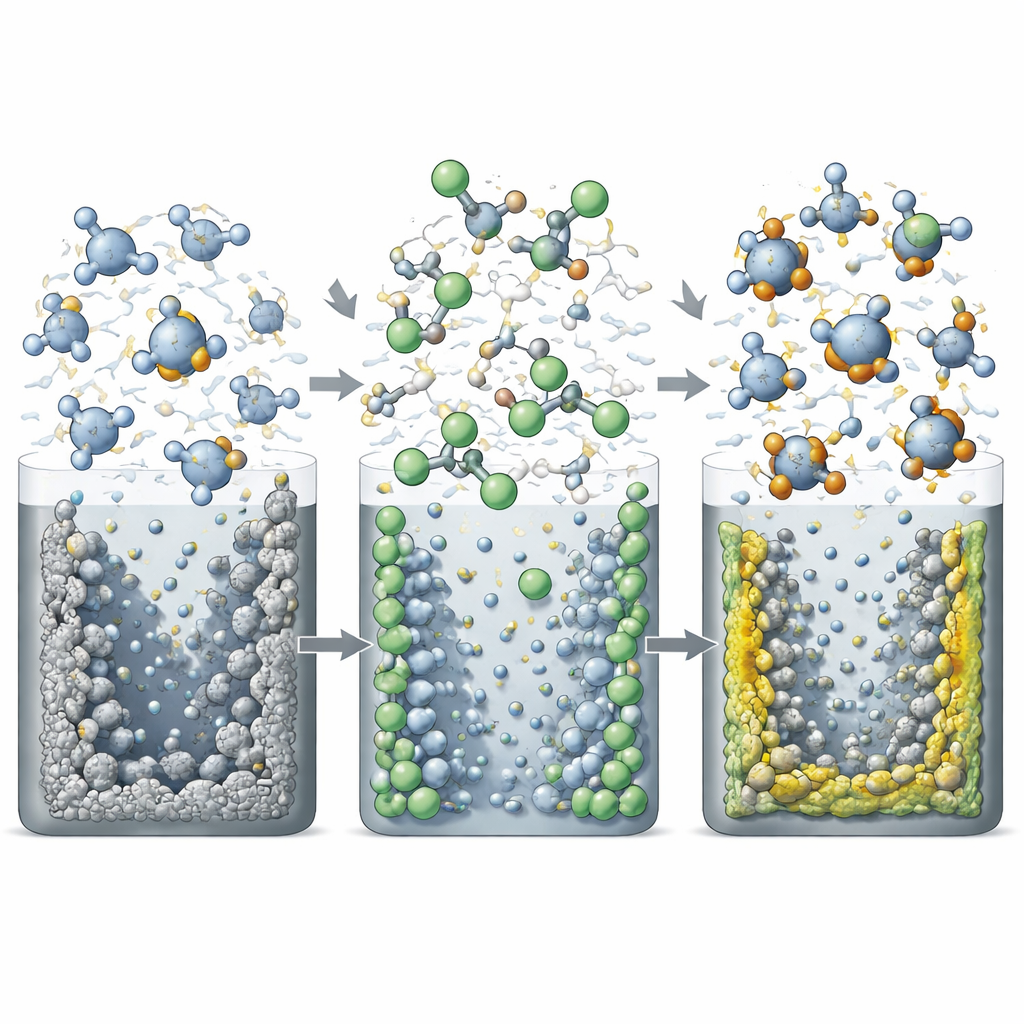
Self‑Forming Protective Coats on Both Electrodes
HFMTFP also serves a second, crucial role. Because of its fluorinated structure and its different behavior when free versus when bound to lithium, it decomposes preferentially at both electrodes in ways that are actually helpful. At the lithium metal surface, coordinated HFMTFP and the anion in the salt break down into inorganic compounds rich in lithium fluoride and lithium oxide, building a thin, uniform, and mechanically strong protective layer. At the high‑voltage NCM811 cathode, free HFMTFP oxidizes to form a fluorine‑rich coating that shields the active material from the harsh electrolyte. Measurements confirm that these coatings are more inorganic and fluorine‑rich than those formed in standard electrolytes, and microscopy shows that lithium deposits remain compact rather than growing into fragile, mossy structures.
Long‑Lasting Performance Under Realistic Stress
To see whether this molecular fine‑tuning truly matters in practice, the team tested full lithium metal cells with NCM811 cathodes under demanding conditions: high voltage (up to 4.4 V), high current (twice the normal rate), and realistic loading of active material. Compared with simpler electrolytes, the ternary formulation sharply reduced aluminum corrosion and high‑voltage side reactions. Cells using the new electrolyte kept more than 90% of their original capacity after 250 fast charge–discharge cycles, while conventional versions faded well below this mark much earlier. Structural probes of the cathode revealed that its layered crystal framework stayed remarkably intact, indicating that the protective coating successfully blocked damaging changes deep inside the material.
What This Means for Future Devices
In essence, the study shows that adding a modestly interacting, fluorinated cosolvent to the electrolyte can corral a reactive but useful solvent like DME, preventing it from misbehaving while turning its partner into a builder of protective films. By engineering how molecules arrange themselves around lithium ions, the researchers created an electrolyte that both resists breakdown at high voltage and naturally forms strong, inorganic skins on the battery’s electrodes. This strategy brings lithium metal batteries a step closer to everyday use in long‑range electric vehicles and high‑end portable electronics, where durability and safety must match their impressive energy density.
Citation: Kim, J., Lee, K., Kim, I. et al. Dual solvents with different coordination strengths for localized high concentration electrolytes in lithium metal batteries. npj Energy Mater. 1, 2 (2026). https://doi.org/10.1038/s44456-025-00002-0
Keywords: lithium metal batteries, electrolyte design, high voltage cathodes, fluorinated solvents, battery cycle life