Clear Sky Science · en
Development and validation of an activatable PET radiotracer reporting extracellular myeloperoxidase activity for the detection of unstable atherosclerotic plaque
Why this matters for heart health
Most heart attacks and strokes are triggered not by the biggest artery blockages, but by small, fragile patches of fatty buildup that suddenly tear or erode. Doctors currently struggle to see which plaques are about to cause trouble. This study introduces a new medical imaging agent for PET scans that is designed to light up only those “dangerous” plaques driven by a particular inflammatory enzyme, potentially helping doctors find high‑risk areas in arteries before they cause an emergency.
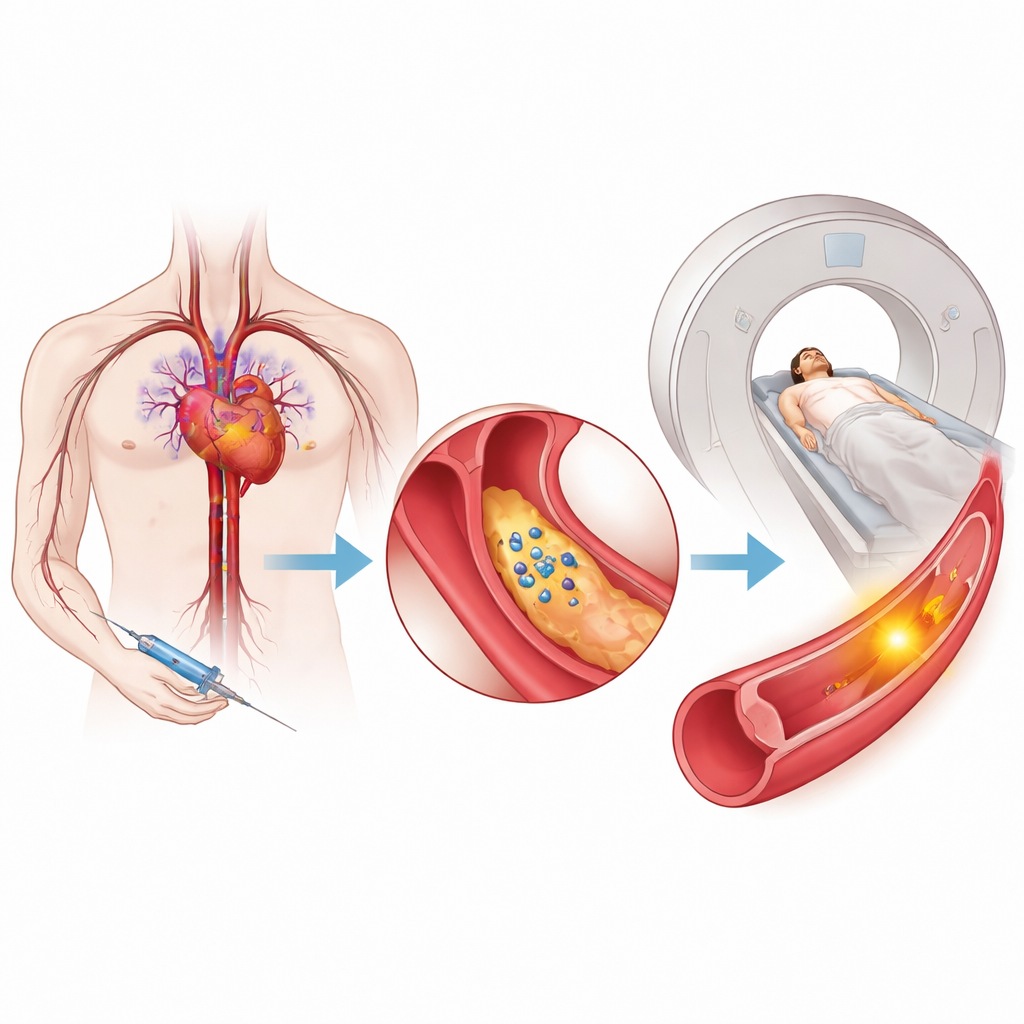
The hidden danger inside artery plaque
Atherosclerosis—fatty buildup inside arteries—is a leading cause of disability and early death worldwide. Not all plaque is equally harmful: some deposits are relatively stable, while others become thin‑capped, inflamed, and prone to rupture, triggering clots that can block blood flow to the heart or brain. Standard scans usually show how narrow an artery is, or how much plaque is present, but they do not reliably reveal whether that plaque is actively damaged and unstable. Existing PET tracers mostly highlight general immune activity, and cannot clearly distinguish between inflammation that is harmful and inflammation that is part of healthy repair.
An enzyme that marks truly dangerous inflammation
The researchers focused on myeloperoxidase (MPO), an enzyme released by certain white blood cells when they mount an aggressive, tissue‑damaging response. MPO helps kill microbes, but when it spills into the space around artery plaque, it can thin the protective cap, erode the surface, and make clots more likely. Animal and human studies have shown that plaques with high MPO activity are more likely to be unstable, and that blocking MPO can stabilize plaques without necessarily changing how many immune cells are present. That makes extracellular MPO activity a promising marker of genuinely dangerous plaque behavior, rather than just the presence of inflammation.
Designing a smart tracer that sticks where trouble is
To tap into this signal, the team created a new PET tracer called [68Ga]Ga‑IEMA. Chemically, it is built so that it normally stays dissolved in blood and does not enter cells. In lab tests, the tracer was stable in human serum and had properties that prevent it from crossing cell membranes, meaning it mainly reports events outside cells. When it encounters active MPO in the presence of its usual oxidants, the tracer molecules link together and bind to nearby proteins, forming larger complexes that become trapped in the local tissue. This “activation‑and‑stick” behavior is what turns MPO activity into a bright PET signal at sites of unstable plaque.
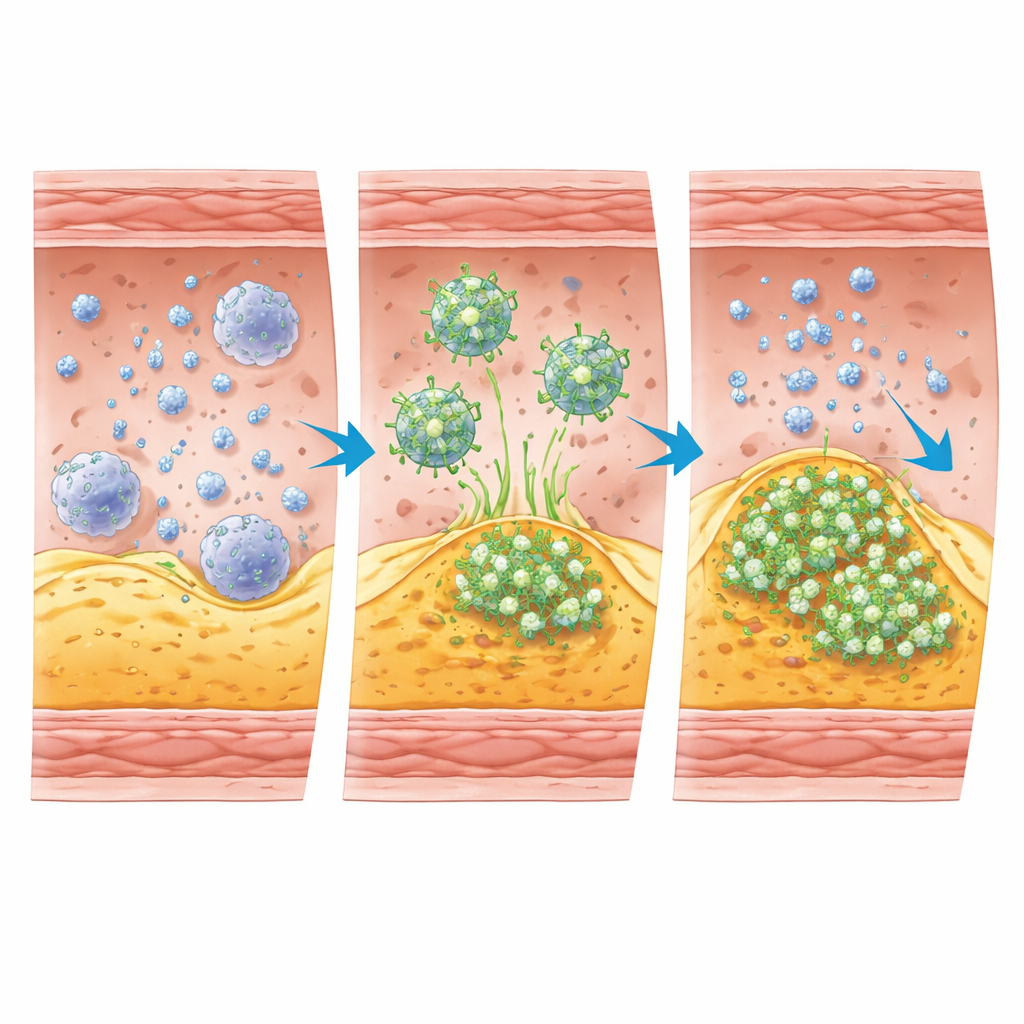
What the animal studies revealed
The scientists first confirmed that [68Ga]Ga‑IEMA could be produced quickly and cleanly, with high radiochemical purity, and that it cleared from the bloodstream mainly through the kidneys in mice. They then used a well‑established mouse model that develops both stable and unstable plaques in different artery segments. PET/CT scans showed that the tracer accumulated much more strongly in the artery region known to harbor unstable plaque than in regions with stable plaque or no plaque at all. In rabbits with advanced atherosclerosis, a non‑radioactive version of the tracer was found at higher levels in plaques that had ruptured or eroded and formed clots, compared with more stable plaques, again matching the pattern expected for high MPO activity.
Looking ahead to use in patients
Taken together, the results indicate that [68Ga]Ga‑IEMA acts as an imaging reporter for extracellular MPO activity, highlighting plaques that are more likely to be dangerous rather than simply large. Because it uses gallium‑68 and a chelator chemistry already familiar in clinical nuclear medicine, the tracer could be translated to human studies with relatively modest regulatory hurdles and radiation doses similar to commonly used PET agents. If future trials confirm its safety and performance in people, this tracer could help cardiologists pinpoint vulnerable plaques and assess whether anti‑inflammatory treatments are truly calming harmful arterial inflammation, moving care closer to a precision, risk‑based approach.
Citation: Keeling, G.P., Wang, X., Chen, W. et al. Development and validation of an activatable PET radiotracer reporting extracellular myeloperoxidase activity for the detection of unstable atherosclerotic plaque. npj Imaging 4, 23 (2026). https://doi.org/10.1038/s44303-026-00156-9
Keywords: atherosclerosis, PET imaging, myeloperoxidase, vulnerable plaque, radiotracer