Clear Sky Science · en
p21+TREM2+ senescent macrophages fuel inflammaging and metabolic dysfunction-associated steatotic liver disease
Why aging and liver health are closely linked
As people live longer and rates of obesity rise, more adults develop fatty liver disease and low‑grade, body‑wide inflammation. This study asks a deceptively simple question with big implications: which specific cells in our organs keep the immune system smoldering as we age, and can turning them off—or removing them—reverse damage in the liver?
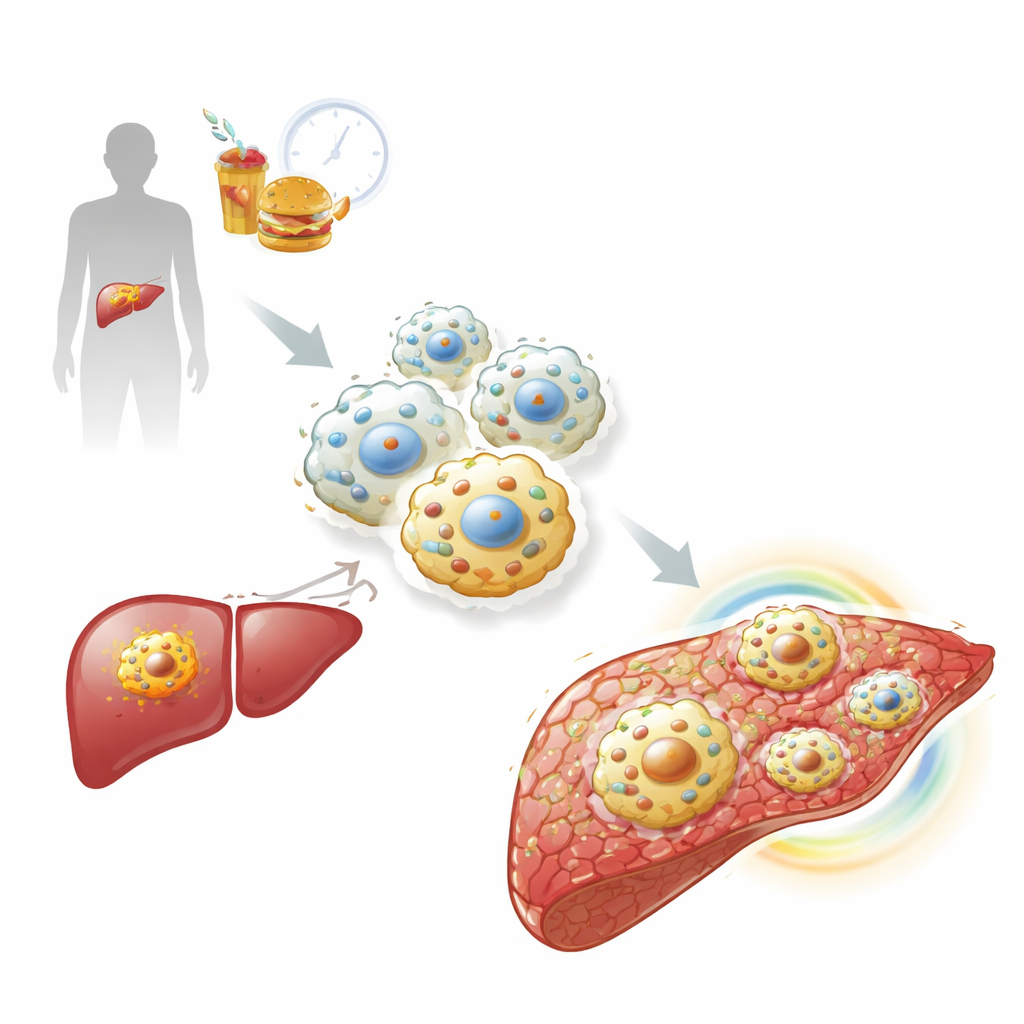
The immune cells that grow old and stay put
Our bodies are patrolled by macrophages, immune cells that swallow debris, dying cells, and microbes. Unlike many other cells, tissue‑resident macrophages can live for years in the same organ. The authors show that these long‑lived sentinels can enter a deeply aged state called cellular senescence. In laboratory experiments, mouse and human macrophages exposed to DNA‑damaging stress or high cholesterol stopped dividing, grew larger, and displayed classic features of senescence. These "aged" macrophages formed a distinct group, different from the usual pro‑inflammatory (M1) or healing (M2) types, and were marked by high levels of the protein p21 and the surface receptor TREM2.
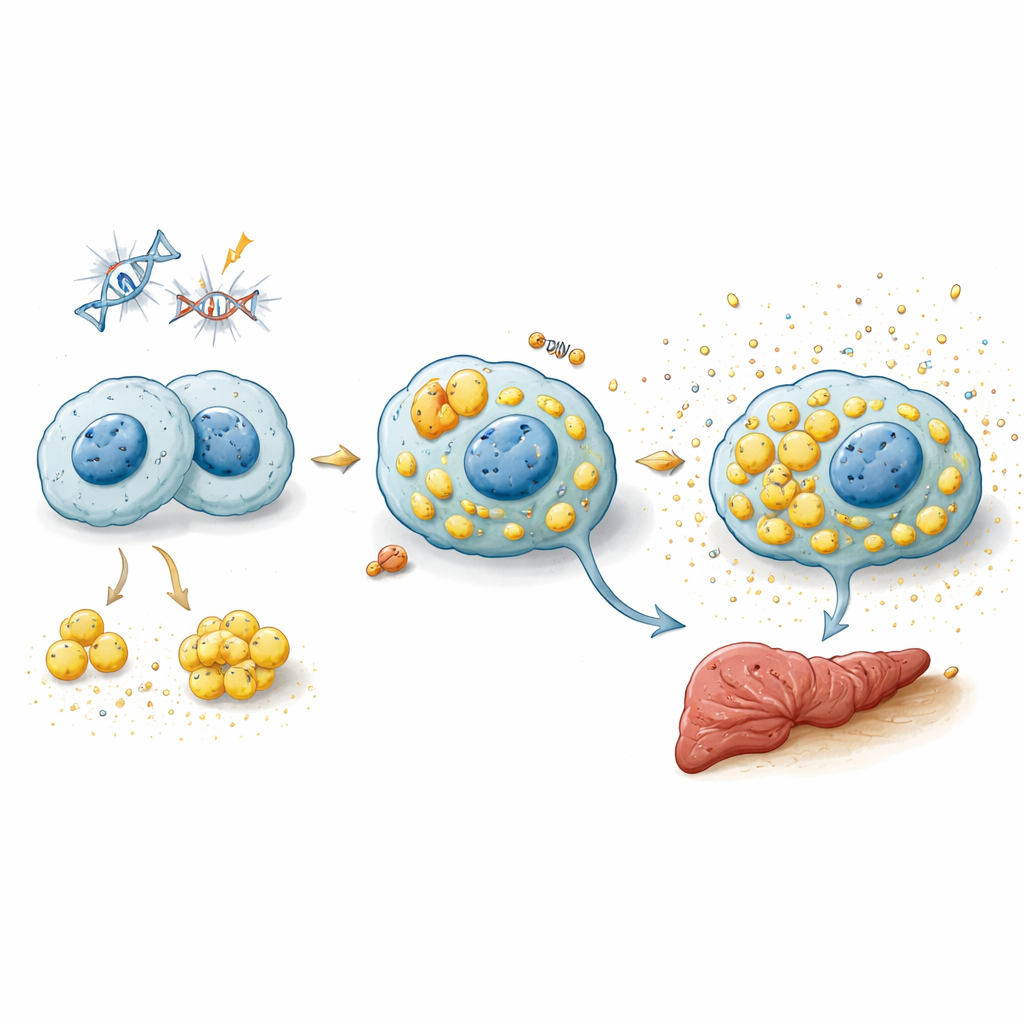
How stressed macrophages turn into inflammation factories
Senescent macrophages did more than simply stop dividing—they changed what they secreted. Using RNA sequencing and protein profiling, the team found a strong "secretory" pattern rich in inflammatory molecules and tissue‑remodeling enzymes, together known as the senescence‑associated secretory phenotype. A key driver was a signaling cascade triggered when damaged mitochondria leaked bits of their DNA into the cell interior. This loose DNA activated an alarm system called cGAS‑STING, which in turn boosted type I interferon responses, priming macrophages to overreact to additional triggers. When the researchers disrupted a mitochondrial enzyme, CMPK2, they reduced this DNA leakage and dialed down the interferon and inflammatory programs, showing that this pathway helps lock macrophages into their senescent, inflammatory state.
Cholesterol turns helpful cells into fatty, harmful "foam" cells
Because aging often coincides with high blood cholesterol, the investigators asked whether fat overload could push macrophages into senescence. Detailed lipid measurements revealed that senescent macrophages accumulated cholesterol esters and other fats, resembling the foam cells seen in clogged arteries. When normal macrophages were experimentally stuffed with cholesterol‑rich particles, they developed large lipid droplets, upregulated p21 and TREM2, and acquired senescent features and an inflammatory secretory pattern. In aging mice, the liver's resident macrophages—Kupffer cells—showed the same p21‑ and TREM2‑rich signature and a senescent gene program the authors call the "MSen" profile. This signature was also elevated in mouse models of metabolic dysfunction‑associated steatotic liver disease (MASLD) and in human cirrhotic livers, indicating that cholesterol‑driven, senescent macrophages appear in real‑world liver disease.
Removing senescent macrophages to protect the liver
If senescent macrophages drive chronic inflammation, could selectively eliminating them improve liver health? The team tested a drug called ABT‑263 (navitoclax), a senolytic compound that forces certain stressed cells into programmed death by blocking anti‑apoptotic proteins. In cell culture, ABT‑263 killed senescent macrophages far more efficiently than normal or simply activated ones. In aged mice, intermittent dosing sharply reduced the proportion of p21‑positive macrophages in the liver, lowered inflammatory gene expression, and shrank fat droplets in liver tissue. In a cholesterol‑driven MASLD mouse model, the same treatment reduced liver size, improved fatty changes, lowered systemic inflammatory markers, and partially restored levels of NAD⁺, a vital metabolic molecule often depleted in aging tissues.
What this means for aging, fatty liver, and future treatments
For a layperson, the main takeaway is that a specific subclass of long‑lived immune cells—p21⁺TREM2⁺ senescent macrophages—acts as a persistent spark for inflammation in the aging, cholesterol‑burdened liver. These cells accumulate fat, stop renewing themselves, and constantly release inflammatory signals that promote fatty liver disease and metabolic decline. By pinpointing their molecular fingerprints and showing that a senolytic drug can selectively clear them, this study suggests a new therapeutic angle: rather than only lowering cholesterol or suppressing broad inflammation, doctors might one day target these senescent macrophages to cool chronic inflammation, improve liver health, and perhaps influence other age‑related diseases driven by similar cells.
Citation: Salladay-Perez, I.A., Avila, I., Estrada, L. et al. p21+TREM2+ senescent macrophages fuel inflammaging and metabolic dysfunction-associated steatotic liver disease. Nat Aging 6, 792–815 (2026). https://doi.org/10.1038/s43587-026-01101-6
Keywords: cellular senescence, macrophages, fatty liver disease, inflammaging, cholesterol metabolism