Clear Sky Science · en
Neuronal APOE4-induced early hippocampal network hyperexcitability in Alzheimer’s disease pathogenesis
Why restless brain circuits matter
Many people carry a common gene variant called APOE4 that greatly increases their risk of developing Alzheimer’s disease, often decades before symptoms appear. Yet what actually happens inside the brain during those quiet years has been a mystery. This study uses sophisticated recordings and genetic tools in mice to show that APOE4 makes specific memory circuits in the brain become overactive and unstable early in life, long before memory problems surface. Understanding this early phase may open a window for prevention, rather than waiting until damage is widespread.
Early electrical storms in memory hubs
The researchers focused on the hippocampus, a seahorse-shaped structure crucial for forming memories and mapping space. In mice engineered to carry human APOE4, they recorded the brain’s electrical activity while the animals moved freely. They looked for brief, high-amplitude bursts known as interictal spikes, a hallmark of overly excitable networks often seen in epilepsy and in people with Alzheimer’s. Young APOE4 mice showed many more of these events in two hippocampal regions, called CA3 and the dentate gyrus, than control mice carrying the lower-risk APOE3 form. A neighboring region, CA1, remained relatively quiet. Strikingly, the number of early spikes in CA3 and the dentate gyrus predicted how poorly the same mice would perform on a spatial learning task many months later, linking early network hyperactivity to future memory decline.
Smaller, jumpier neurons in vulnerable circuits
To uncover what drives this hyperactivity, the team sliced the hippocampus and measured how individual neurons responded to injected currents. In young APOE4 mice, CA3 pyramidal cells and a subset of dentate granule cells fired more easily and more often than those from APOE3 mice. These neurons required less current to spike, responded faster, and had a steeper firing response, all signs of intrinsic hyperexcitability. At the same time, they were physically smaller: measurements of electrical capacitance and direct microscopy showed reduced cell surface area and volume. Smaller neurons tend to have higher input resistance, so incoming signals push their voltage around more strongly, making them easier to trigger. In older APOE3 mice, neurons gradually "caught up"—they too became smaller and more excitable—suggesting that APOE4 accelerates an aging-like process rather than creating an entirely new one.
When balance between go and stop signals fails
Neural circuits rely on a delicate balance between excitatory signals that drive activity and inhibitory signals that rein it in. The researchers examined this balance by recording tiny spontaneous currents coming into CA3 and dentate neurons. In young APOE4 mice, CA3 cells received extra excitatory input while inhibition remained stable, tilting the excitation–inhibition ratio upward and further amplifying their hyperactivity. In the dentate gyrus, the picture shifted with age: excitatory drive stayed similar, but inhibitory input declined in APOE4 mice, consistent with earlier work showing gradual loss of certain inhibitory interneurons there. This progressive drop in braking power left the dentate gyrus persistently overexcitable in older APOE4 animals, just when their memory performance worsened.
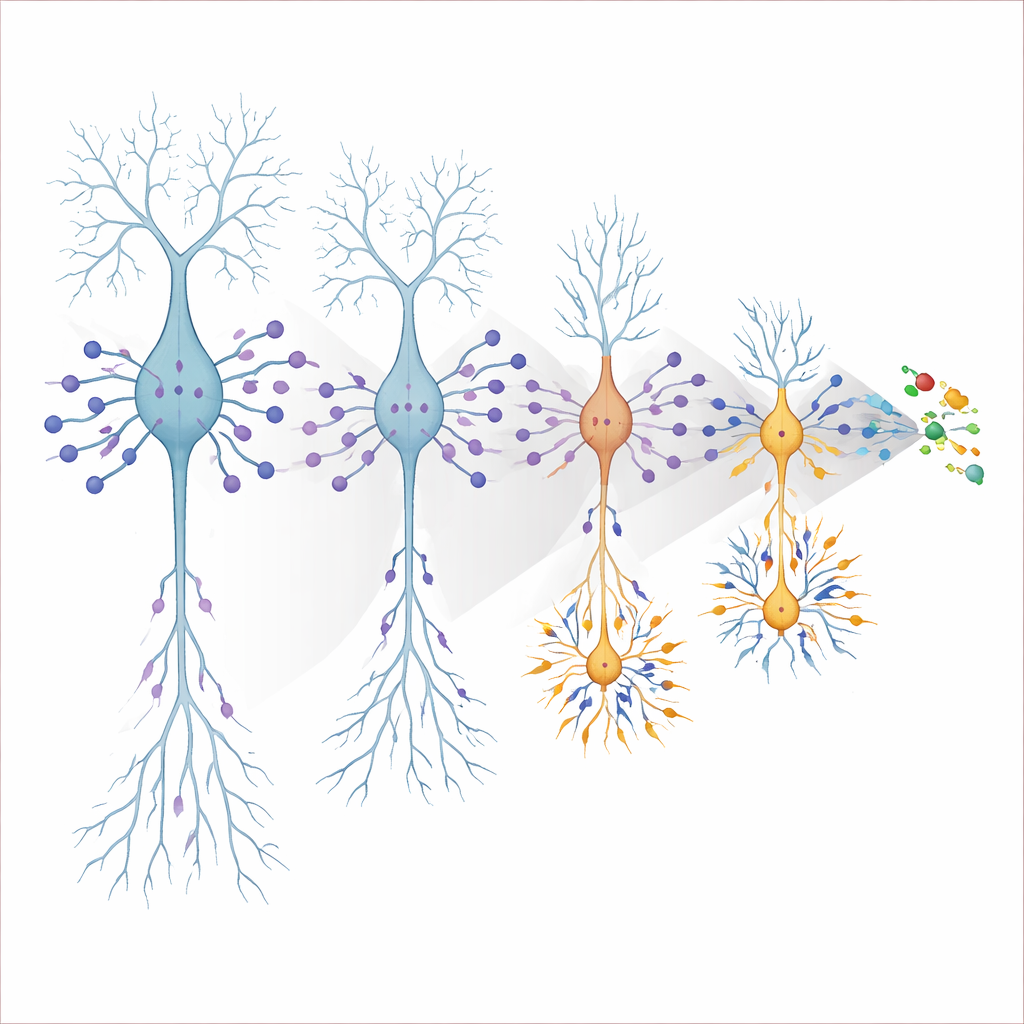
A harmful role for neuron-made APOE4 and a new molecular suspect
APOE is often thought of as a support protein made by star-shaped glial cells, but stressed neurons can make it too. By selectively deleting APOE4 only in neurons, the authors showed that this neuronal source was crucial: removing APOE4 from neurons completely normalized their size and firing behavior, while removing it from glia had little effect on excitability. To pinpoint downstream molecular players, they performed single-nucleus RNA sequencing across ages and cell types, then filtered for genes whose expression tracked with the early hyperexcitable state. One standout was Nell2, a protein previously linked to neuron growth and synapses and found at elevated levels in human Alzheimer’s brain and spinal fluid. Using a CRISPR interference approach to dial down Nell2 in APOE4 neurons, the team was able to enlarge these cells and quiet their firing back toward normal, without altering neighboring cells—strong evidence that excess Nell2 helps convert APOE4’s presence into structural shrinkage and electrical overactivity.
What this means for future Alzheimer’s prevention
Taken together, the results outline a chain of events: neuronal APOE4 boosts Nell2 and other gene changes in select hippocampal cells; those neurons shrink and become hyperexcitable; local circuits in CA3 and the dentate gyrus enter a long-lasting overactive state; over time, inhibitory cells are lost and the network’s braking system fails, culminating in memory problems. For people who carry APOE4, this suggests that harmful changes may begin years before symptoms, when targeting network hyperexcitability or molecules like Nell2 could still protect vulnerable circuits. Rather than viewing Alzheimer’s solely as a late-life accumulation of plaques and tangles, this work highlights it as a disorder of early, subtle circuit instability—one that might be steadied if we intervene soon enough.
Citation: Tabuena, D.R., Jang, SS., Grone, B. et al. Neuronal APOE4-induced early hippocampal network hyperexcitability in Alzheimer’s disease pathogenesis. Nat Aging 6, 886–904 (2026). https://doi.org/10.1038/s43587-026-01096-0
Keywords: APOE4, hippocampal hyperexcitability, Alzheimer’s disease, dentate gyrus, Nell2