Clear Sky Science · en
Irisin ameliorates obesity and insulin resistance via adipose tissue IL-33 and regulatory T cells
Why a Workout Molecule Matters for Weight and Blood Sugar
Obesity and type 2 diabetes now affect hundreds of millions of people worldwide, and many treatments focus mainly on eating less. This study explores a different angle: how a natural hormone released by exercising muscles, called irisin, talks to fat tissue and the immune system to burn more energy, calm inflammation and improve blood sugar—without cutting food intake or losing muscle.
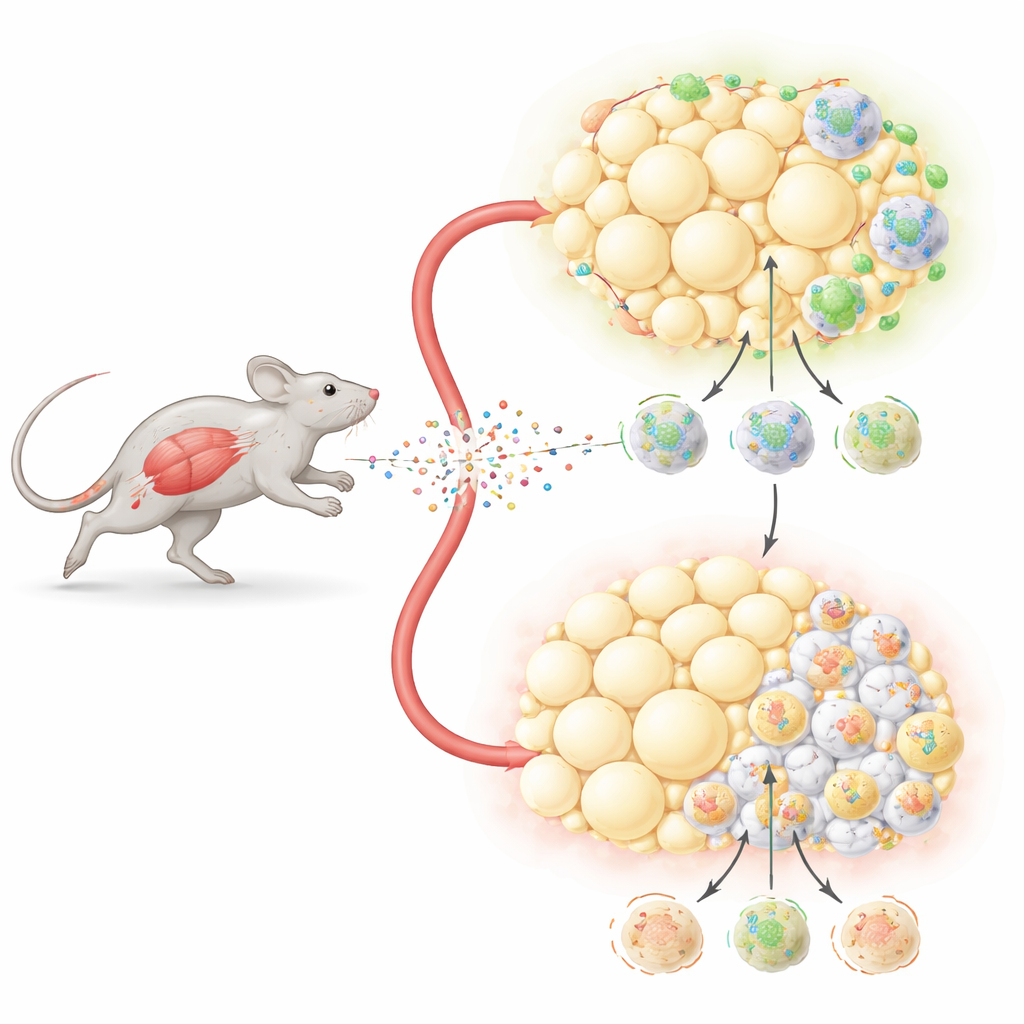
A Messenger Released by Working Muscles
When we exercise, our muscles release irisin into the bloodstream. Earlier work showed that irisin can turn energy-storing white fat just under the skin into a more active, heat-producing form. But it was unclear how this signal influenced the deeper belly fat that is closely linked to diabetes and heart disease. In this study, researchers boosted irisin levels in male mice using a harmless virus that made the liver continually release the hormone. The mice were then fed a high-fat diet for many weeks to induce obesity and insulin resistance, mimicking common human metabolic problems.
Less Fat, Better Blood Sugar, Same Appetite
Mice with long-term irisin elevation gained less weight on the high-fat diet than control animals, even though they ate the same amount of food and moved about just as much. Scans showed that the weight difference came mainly from reduced fat mass, not from loss of lean tissue like muscle. These mice also handled sugar and insulin better: after injections of glucose or insulin, their blood sugar levels dropped more quickly and to lower levels. Measurements of whole-body energy use revealed that the irisin-treated animals were burning more calories around the clock, suggesting that their fat tissue was behaving more like a furnace than a storage depot.
Tuning Down Harmful Inflammation in Belly Fat
Belly, or visceral, fat is not just a passive storage site; it is packed with immune cells that can become chronically inflamed in obesity and interfere with normal metabolism. The team carefully mapped these cells in the visceral fat of the mice. With a long-term high-fat diet, control animals showed the expected pattern: more aggressive immune cells and a marked loss of a protective cell type called regulatory T cells that normally helps keep inflammation in check. In contrast, mice with extra irisin maintained a robust population of these protective cells, especially a subset that carries a receptor called ST2, while showing fewer inflammatory cells. Sections of fat under the microscope also contained fewer “crown-like structures,” scars that mark dead fat cells surrounded by immune cells, indicating less ongoing damage.
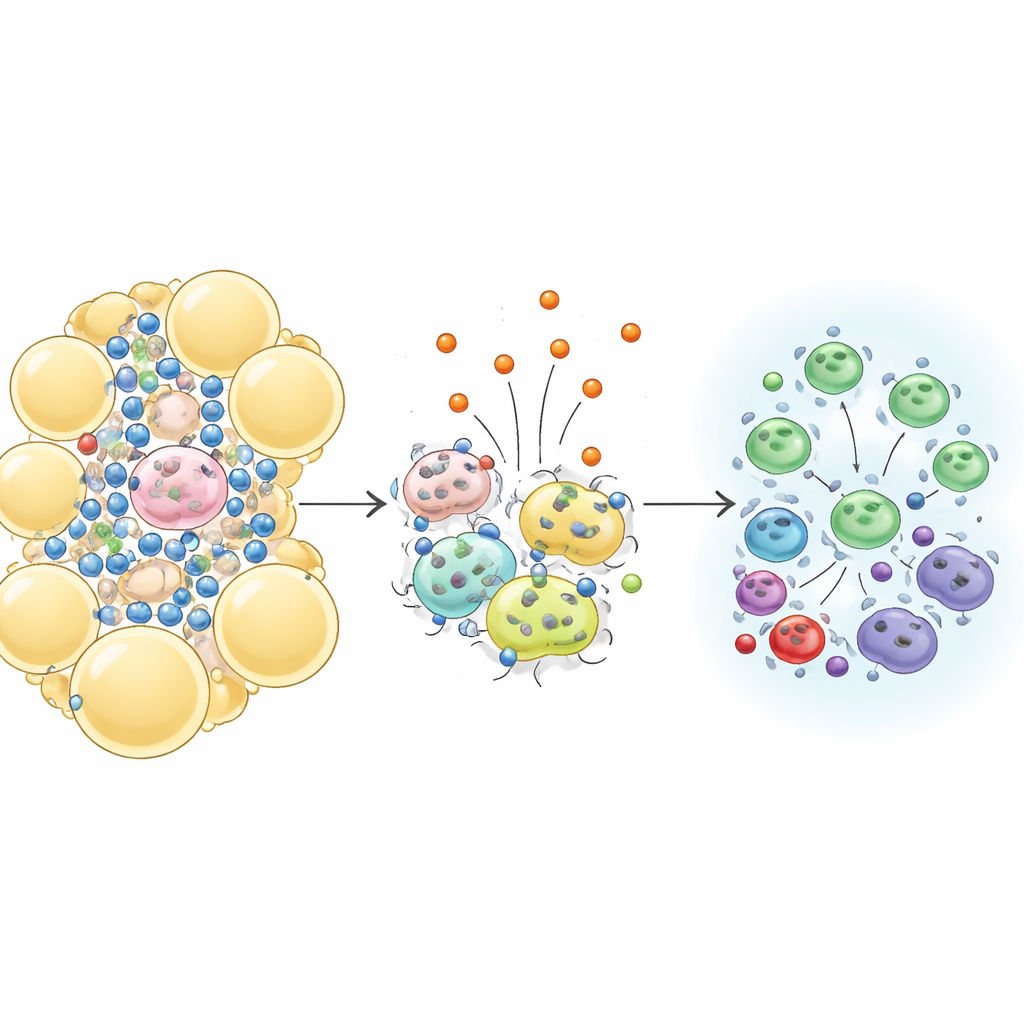
A Three-Way Conversation: Irisin, Stromal Cells and Immune Guardians
To understand how irisin preserved these guardian cells, the researchers turned to another key player in fat tissue: structural support cells known as mesenchymal stromal cells. In visceral fat, a subset of these stromal cells normally produces the signaling protein IL-33, which is vital for the survival of ST2-carrying regulatory T cells. The study found that chronic irisin treatment increased both the number and activity of IL-33–producing stromal cells in visceral fat, and raised IL-33 levels locally and in the bloodstream. In cell culture, irisin directly pushed stromal cells to switch on IL-33 and to dial down genes linked to turning into new fat cells, suggesting a shift toward an immune-supporting role. Blocking the specific integrin receptor that irisin uses on these cells prevented this IL-33 boost, tying the effect to a defined surface pathway.
Proving the IL-33 Link and Spreading the Signal to Other Fat
The team then asked whether IL-33 was truly required for irisin’s metabolic benefits. In mice lacking the irisin source protein (FNDC5), IL-33 levels in visceral fat and blood were lower, protective regulatory T cells were scarcer and obesity-related blood sugar problems were worse; supplying extra irisin restored IL-33 and improved metabolism. Conversely, when the researchers soaked up IL-33 with a designer “trap” protein in irisin-treated obese mice, the advantages largely vanished: fat mass climbed back, energy expenditure dropped, and glucose and insulin responses deteriorated. In another genetic test, removing the ST2 receptor only from regulatory T cells blunted irisin’s ability to quiet inflammation and improve blood sugar. At the same time, irisin increased activity of heat-producing genes in subcutaneous fat, and this required IL-33 as well. Together, these findings outline a multi-tissue relay in which irisin drives stromal cells to make IL-33, IL-33 nurtures regulatory T cells in belly fat and boosts thermogenic programs in under-the-skin fat, and the whole system shifts the body toward burning more energy with better metabolic control.
What This Could Mean for Future Treatments
This work shows that a hormone our bodies already make during exercise can reprogram fat tissue and local immune cells in ways that counteract obesity and insulin resistance—without suppressing appetite. By uncovering a specific irisin–IL-33–regulatory T cell axis, the study suggests new strategies for drugs that mimic the benefits of exercise on metabolism. While these experiments were done in male mice and much remains to be tested in humans, the findings hint that future therapies might combine appetite-focused drugs with agents that activate this fat–immune communication circuit to more safely and powerfully improve metabolic health.
Citation: A, M., Wang, G., Zammit, N.W. et al. Irisin ameliorates obesity and insulin resistance via adipose tissue IL-33 and regulatory T cells. Nat Metab 8, 885–901 (2026). https://doi.org/10.1038/s42255-026-01491-2
Keywords: irisin, adipose tissue, regulatory T cells, IL-33, thermogenesis