Clear Sky Science · en
A spatiotemporal transcriptomic atlas of porcine (Sus scrofa) female early gonadal development
Why Early Egg Development Matters
Before any female mammal is born, her future eggs are already being set up inside tiny organs called gonads. Understanding how these earliest stages unfold is crucial for tackling infertility, improving livestock breeding, and safely developing lab-grown eggs. This study builds a detailed “map in space and time” of how early egg precursors form and organize in the ovaries of pig embryos—a powerful stand‑in for humans because pig development closely resembles our own.
Building a Cellular Map in Space and Time
The researchers used a high‑resolution method called spatial transcriptomics, which reads thousands of genes while also keeping track of where each cell sits in the tissue. They examined female pig gonads at five key embryonic days, from when germ cells first arrive to when they begin the special cell division that creates eggs. By analyzing over 50,000 tiny tissue pixels, they classified eight major cell types, including germ cells, several kinds of supporting cells, interstitial cells, blood vessel cells, and boundary‑forming mesenchymal cells. This allowed them to paint a fine‑grained atlas showing how each cell type is positioned and how those positions change as the ovary forms.
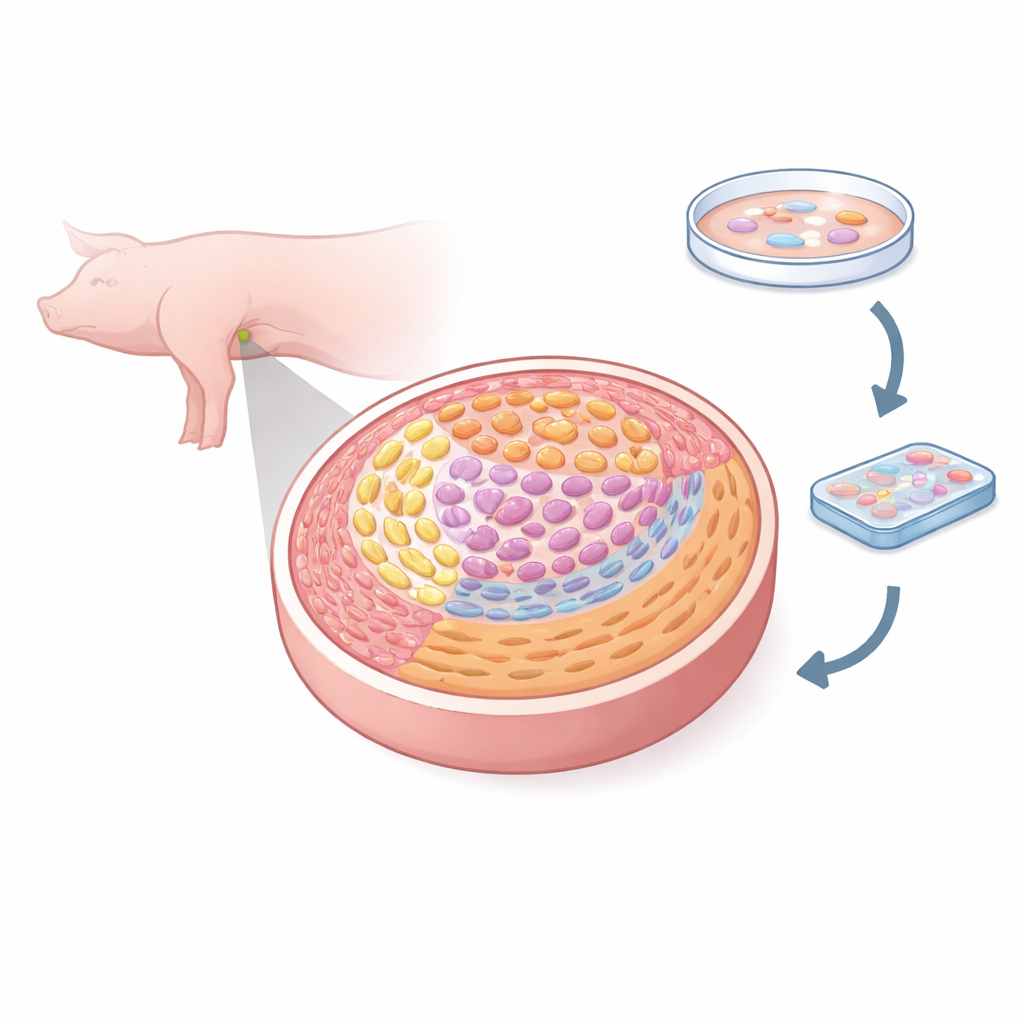
Tracking the Birth of Future Eggs
Within the germ cell population, the team identified three main stages: dividing primordial germ cells, egg‑like cells that respond to a vitamin A–related signal called retinoic acid, and cells that have entered meiosis, the special division that halves chromosome number. By following gene activity over developmental “pseudotime,” they showed that days E24–E50 in the pig embryo cover the full journey from newly arrived primordial germ cells to early egg precursors entering meiosis. At the same time, these cells undergo sweeping epigenetic changes—chemical marks on DNA and histones are stripped away or remodeled, resetting the germ cells for their future role while a key repressive mark, H3K27me3, gradually disappears as cells commit to an egg fate.
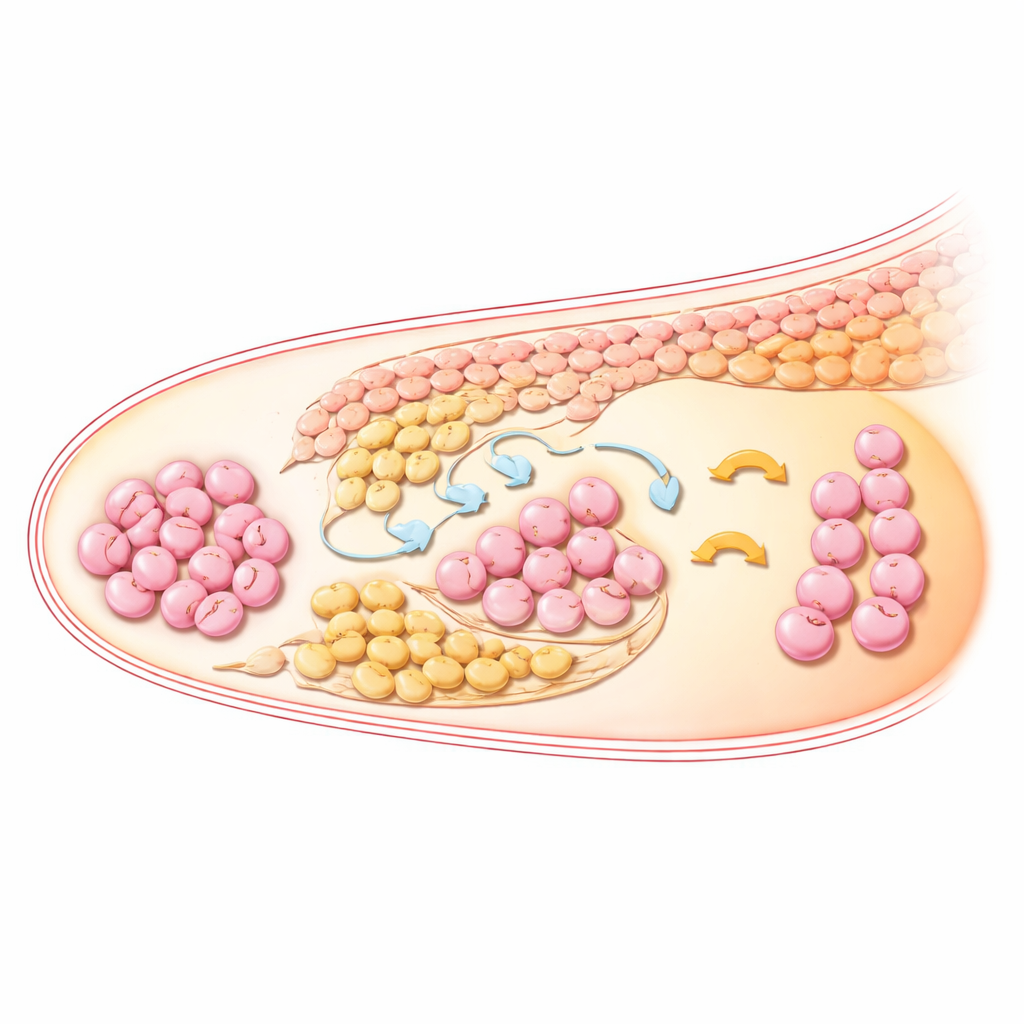
Supporting Cells Form Layered Neighborhoods
The ovary is not just a bag of germ cells; it is a layered community in which supporting cells create the neighborhoods that guide egg development. The study shows that three related supporting cell groups—surface epithelium cells and two waves of pre‑granulosa cells—are arranged in clear layers from the outer cortex inward. Over time, one wave settles mainly in the medulla (the central region), while the second wave stays closer to the germ cells in the cortex and inner cortex. These patterns match what is seen in human fetal ovaries, suggesting a deeply conserved blueprint. Other somatic cells also sort into distinct zones: interstitial cells fill much of the medulla and expand strongly by later stages, while endothelial cells organize into vessel‑like structures near germ cells, likely delivering nutrients and signals.
Signals That Tell Germ Cells to Become Eggs
To understand how the surrounding cells talk to germ cells, the authors modeled cell‑to‑cell signaling using known ligand–receptor pairs. They found that a well‑known developmental pathway driven by BMP molecules (especially BMP2, BMP4, and GDF5) is central to pushing primordial germ cells toward egg identity and into meiosis. These BMP signals are mainly produced by the second wave of pre‑granulosa cells and a subset of interstitial cells, which wrap around germ cells in the cortex. As germ cells progress from retinoic acid–responsive to meiotic stages, BMP signaling grows stronger and turns on downstream genes that mark pathway activation. Compared with mice and humans, pigs seem to maintain BMP activity for a longer window, hinting at species‑specific strategies for building the egg pool.
How This Work Points Toward Lab‑Grown Eggs
The authors conclude that early pig ovaries contain a carefully layered niche where two waves of pre‑granulosa cells, expanding interstitial cells, and nearby blood vessels cooperatively shape germ cell fate. Their atlas pinpoints BMP signaling, along with WNT, NOTCH, and matrix‑related pathways, as key ingredients of the recipe that turns primordial germ cells into egg precursors ready for meiosis. For non‑specialists, the takeaway is that we now have a much clearer, spatially precise picture of how early eggs are made in a species that closely parallels humans. This blueprint will help scientists design more faithful lab systems to grow human egg cells, improve reproductive treatments, and refine breeding strategies for large animals.
Citation: He, P., Xia, W., Chen, T. et al. A spatiotemporal transcriptomic atlas of porcine (Sus scrofa) female early gonadal development. Commun Biol 9, 487 (2026). https://doi.org/10.1038/s42003-026-09932-0
Keywords: ovary development, germ cells, spatial transcriptomics, BMP signaling, pig model