Clear Sky Science · en
Transcriptional and functional profiles of muscarinic receptor-expressing neurons in primate lateral prefrontal and anterior cingulate cortices
How a brain messenger shapes thought and feeling
Every moment, your brain juggles what you think with how you feel. Two key hubs for this are the lateral prefrontal cortex, which helps with planning and working memory, and the anterior cingulate cortex, which tracks motivation, conflict, and pain. This study asks a deceptively simple question: how does the same chemical signal, acetylcholine, tune these two regions in different ways to support learning, flexibility, and emotional control? 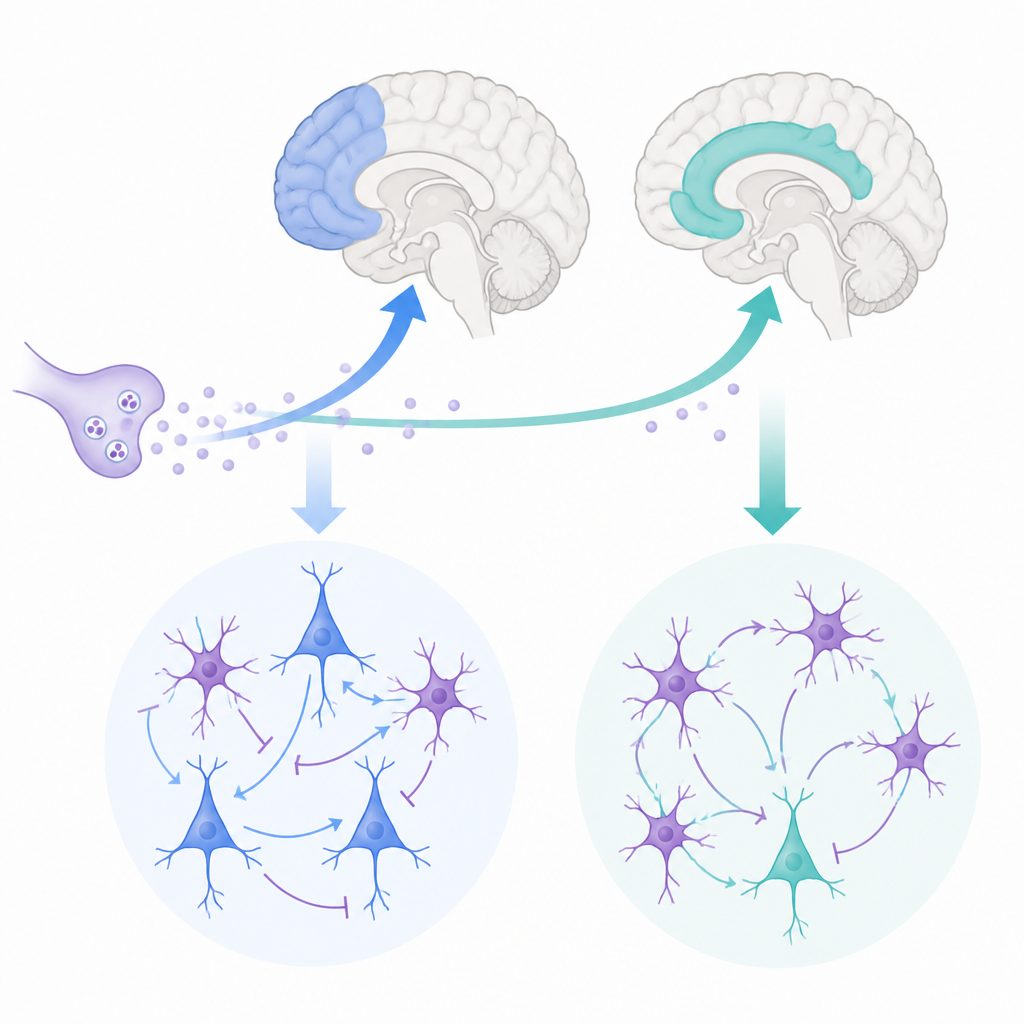
A closer look at two thinking hubs
The researchers focused on two areas in the frontal lobe of rhesus monkeys that closely resemble human brain regions. The lateral prefrontal cortex helps us hold information in mind and make decisions, while the anterior cingulate cortex links actions to rewards, errors, and emotions. Both regions receive input from acetylcholine, a chemical released by deep brain centers that is known to influence attention and memory. Yet the anterior cingulate receives denser acetylcholine input than the lateral prefrontal cortex, hinting that this messenger might sculpt their activity in distinct ways.
Reading the genetic fingerprints of brain cells
To see how individual cells respond to acetylcholine, the team used single-nucleus RNA sequencing, which reads out which genes are active in thousands of cells at once. They focused on muscarinic receptors, the main acetylcholine-sensitive switches on cortical neurons, encoded by four genes called CHRM1 through CHRM4. Surprisingly, they found that the CHRM3 gene was the most widely expressed in both regions, present in over half of all cells, even though earlier protein studies had suggested that the m1 receptor, encoded by CHRM1, was more abundant. Most excitatory and inhibitory neurons carried CHRM3, often together with CHRM1, while CHRM2 showed a more selective pattern, especially in deep-layer excitatory cells and a key group of fast-spiking inhibitory neurons.
When gene messages and proteins do not match
To resolve the mismatch between RNA and protein, the scientists combined fluorescent labeling of receptor proteins with in situ detection of their RNA in brain slices. They confirmed that m1 protein was overall stronger than m3 protein, but discovered that CHRM3 RNA tended to sit in both the nucleus and the surrounding cytoplasm, whereas CHRM1 RNA was mostly in the cytoplasm. This suggests that CHRM3 messages may be held back in the nucleus or turned into protein more slowly, helping explain why its RNA appears plentiful while its protein is less dominant. At the same time, cells that expressed CHRM1 and CHRM3 shared very similar gene signatures related to synaptic signaling and plasticity, while CHRM2-positive cells formed a clearly distinct group linked to pathways that dampen neurotransmitter release.
Two regions, two styles of synaptic tuning
The team then moved from molecules to function, recording tiny electrical currents from individual layer 3 pyramidal neurons in brain slices. They bathed the tissue in carbachol, a drug that activates acetylcholine receptors, and measured spontaneous excitatory and inhibitory synaptic events. In the anterior cingulate, this stimulation tended to reduce excitatory input while boosting inhibitory currents, shifting the local balance toward stronger braking of activity. In the lateral prefrontal cortex, by contrast, cholinergic stimulation most often weakened inhibitory currents and left excitation relatively stronger, nudging the network toward a more active, excitable state. These changes were accompanied by shifts in the shape and density of dendritic spines, the tiny protrusions that host excitatory synapses: carbachol reduced large, stable “mushroom” spines and increased thinner, more flexible ones, with slightly different time courses in each region. 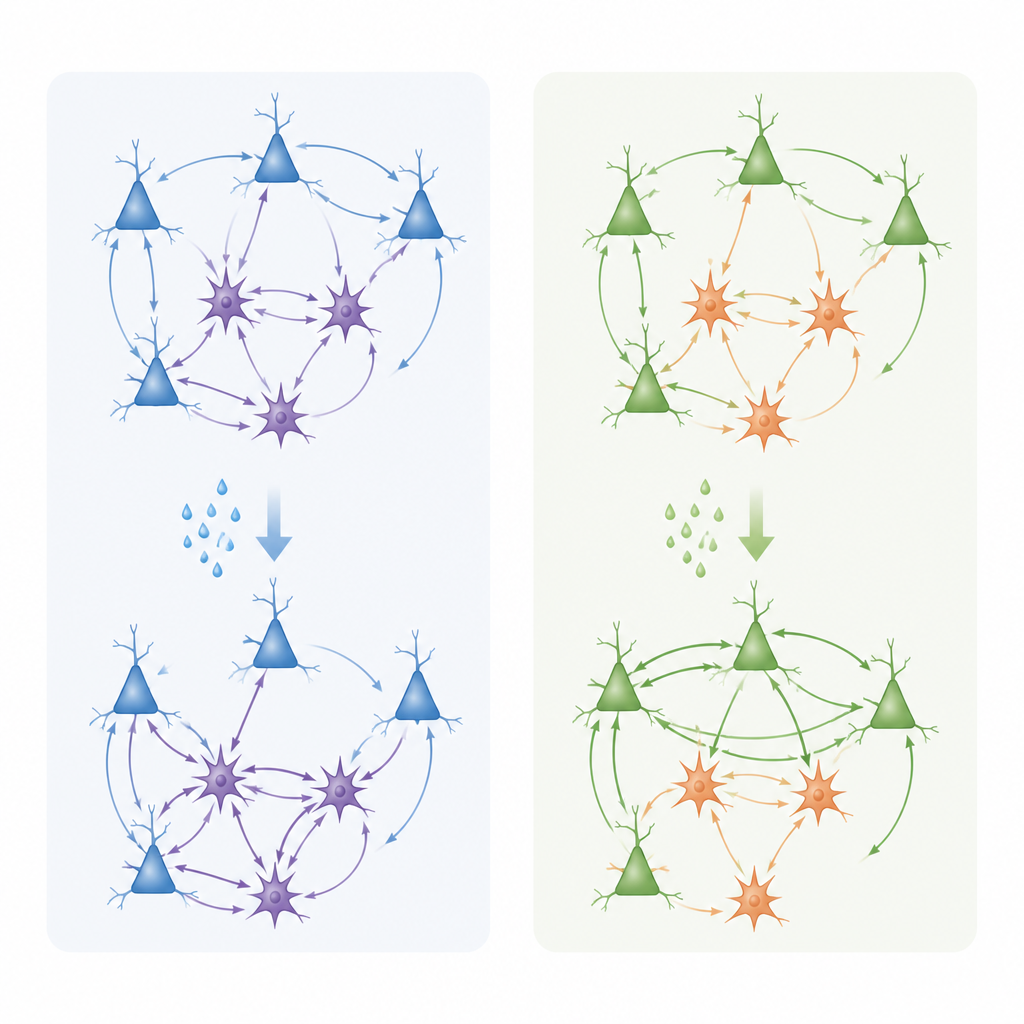
What this means for learning and mental health
Together, the results show that acetylcholine does not act as a simple on–off switch in the frontal cortex. Instead, it engages different mixes of muscarinic receptors in distinct cell types and layers to tune the balance between excitation and inhibition in a region-specific way. In the anterior cingulate, acetylcholine appears to sharpen signal-to-noise by dampening excitatory drive and strengthening inhibition, while supporting genes involved in synaptic remodeling important for emotional learning and pain processing. In the lateral prefrontal cortex, it tends to preserve or enhance excitatory tone and promote structural flexibility at synapses, which may help sustain working memory and adaptive decision-making. By linking detailed gene-expression maps to real-time changes in synaptic activity, this work offers a mechanistic framework for how cholinergic imbalances could contribute to cognitive and affective symptoms in neuropsychiatric disorders, and suggests that targeting specific muscarinic receptor subtypes in defined circuits may allow more precise therapies in the future.
Citation: Tsolias, A., Mojica, C.A., Yamani, R. et al. Transcriptional and functional profiles of muscarinic receptor-expressing neurons in primate lateral prefrontal and anterior cingulate cortices. Commun Biol 9, 620 (2026). https://doi.org/10.1038/s42003-026-09866-7
Keywords: acetylcholine, prefrontal cortex, anterior cingulate, muscarinic receptors, synaptic plasticity