Clear Sky Science · en
The impact of graphene oxide on the magnetic and hyperthermia properties of CoFe2O4 and MnFe2O4 ferrites
Heating Tumors with Tiny Magnets
Cancer doctors have long known that gently warming a tumor can make other treatments like chemotherapy and radiation work better. This study explores a new way to create that heat from inside the body using tiny magnetic particles. By comparing two types of particles and mixing them with ultra-thin carbon sheets, the researchers look for materials that heat tumors efficiently while remaining easy to deliver and control.
Why Magnetic Heat Can Help Treat Cancer
Instead of shining microwaves or light from outside the body, magnetic hyperthermia relies on nanoparticles that warm up when placed in a changing magnetic field. If these particles are injected near a tumor, the field can be switched on from outside the body and the particles act like miniature heaters, raising the local temperature just a few degrees above normal. The challenge is to design particles that produce enough heat at field strengths and frequencies that are safe for patients, while staying well dispersed in body fluids and posing minimal toxicity.
Two Magnetic Materials Under the Microscope
The team focused on two iron-based compounds, manganese ferrite (MnFe2O4) and cobalt ferrite (CoFe2O4). Both are tiny magnets, but they behave very differently. Manganese ferrite is a "soft" magnet, which means its internal magnetization can flip relatively easily. Cobalt ferrite is a "hard" magnet, whose magnetization is strongly locked in place. The nanoparticles were made in water using a hydrothermal method and examined with X-ray diffraction and electron microscopes to confirm their structure, shape, and size. MnFe2O4 particles were mostly cubes and cuboids around 20 to 30 nanometers across, while CoFe2O4 particles were smaller and more spherical, around 14 nanometers.
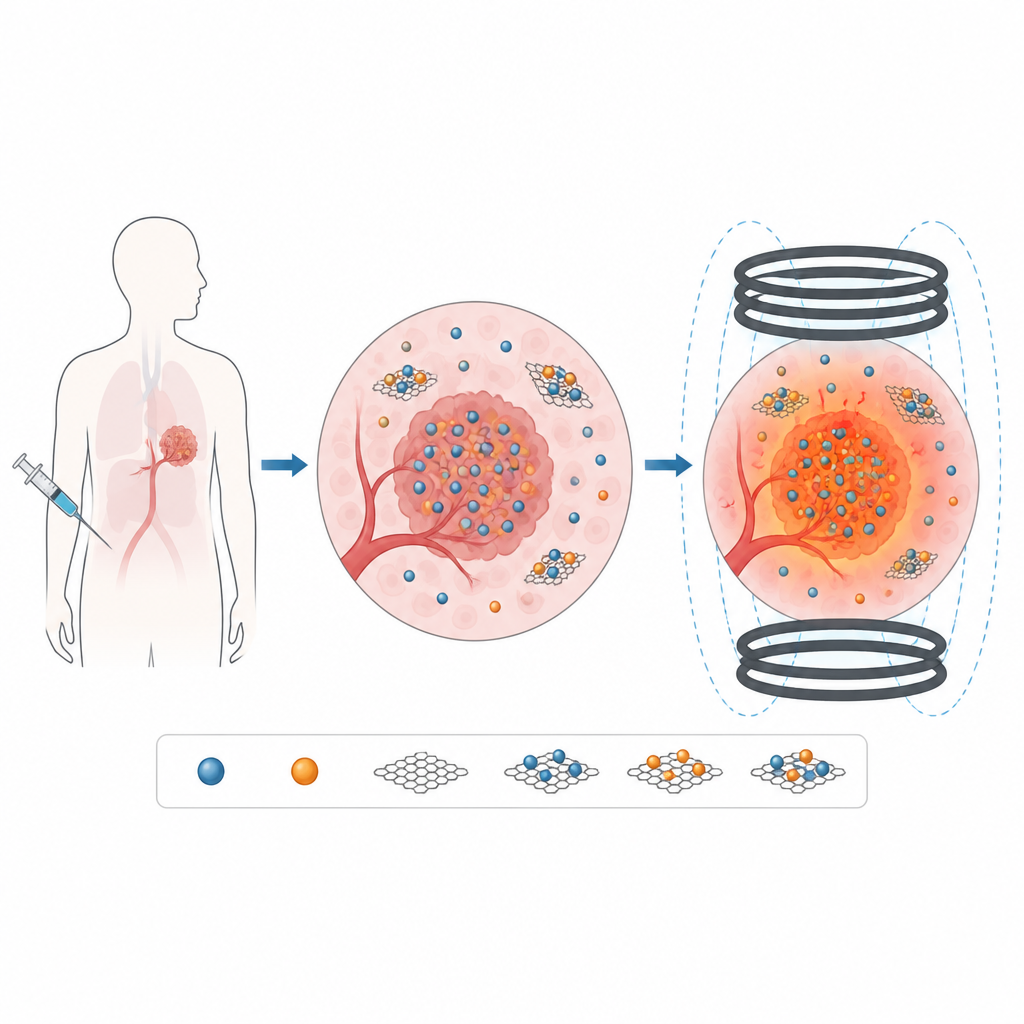
Adding Graphene Oxide: Help and Hindrance
To improve stability in liquid and create a surface that can later be decorated with drug or targeting molecules, the researchers anchored the ferrite nanoparticles onto sheets of graphene oxide, a flat carbon material rich in oxygen groups. Imaging showed MnFe2O4 and CoFe2O4 particles spread across the flexible sheets rather than clumping together. Spectroscopy confirmed that the chemical bonds in both the ferrites and the graphene oxide remained intact in the composites. However, magnetic tests revealed that adding graphene oxide consistently reduced the overall magnetization of the samples, because the non-magnetic carbon diluted the amount of active magnetic material and introduced extra defects at the interface.
How the Particles Actually Produce Heat
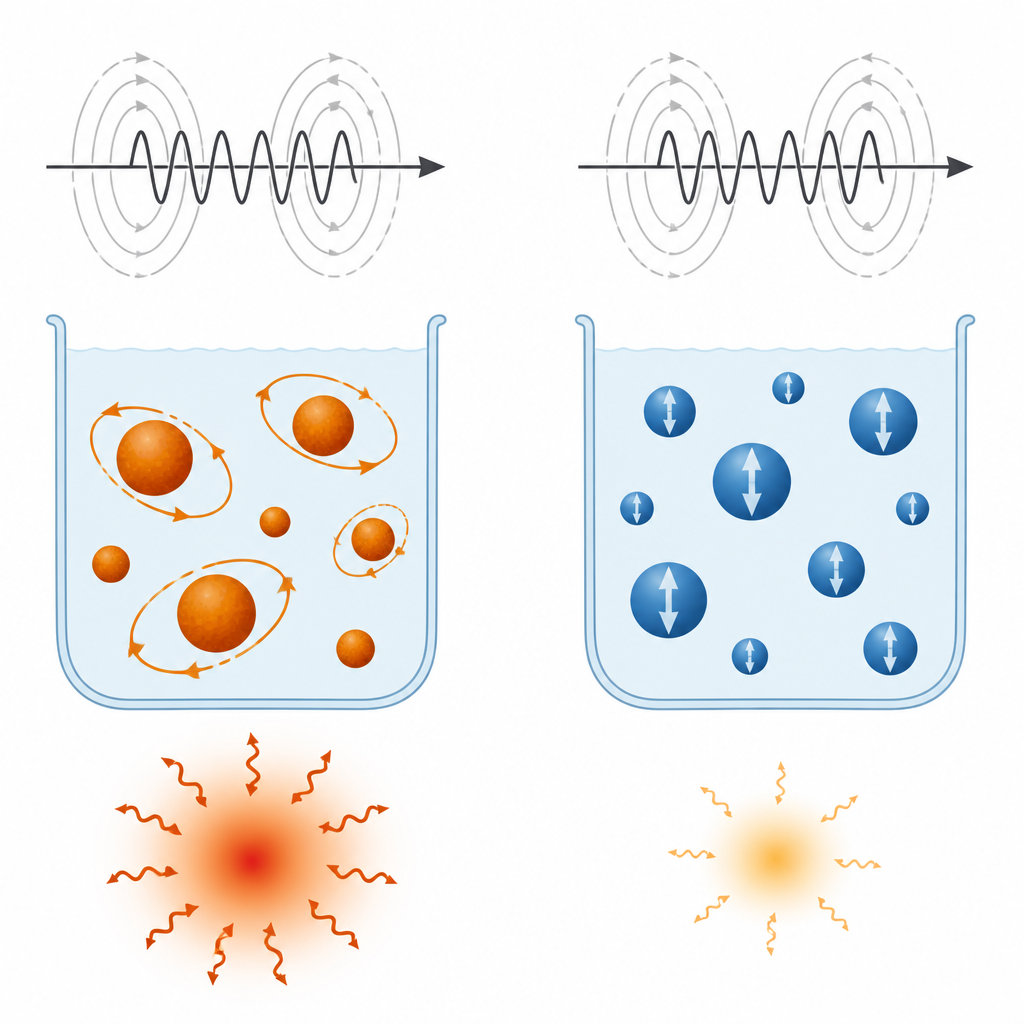
When the suspensions were placed in an alternating magnetic field similar to what might be used in treatment, all samples warmed up, but not equally. Manganese ferrite reached a specific absorption rate of about 110 watts per gram, while cobalt ferrite reached about 70 watts per gram. The key lies in how the particles respond to the changing field. For MnFe2O4, the magnetization inside each particle can both flip internally and allow the whole particle to rotate slightly in the liquid. These two kinds of motion work together and occur on a timescale that matches the speed of the applied field, making heating efficient. In CoFe2O4, the internal magnetization is effectively frozen by its strong magnetic locking, so only the slower physical rotation in the fluid contributes, which is less effective under the tested conditions. Adding graphene oxide lowered heating in both cases, more strongly for MnFe2O4, because it reduced magnetization and pinned some of the magnetic regions so they could not respond freely.
What This Means for Future Cancer Treatments
This work shows that simply choosing particles with high magnetization is not enough to get strong heating for cancer therapy. The ease with which the internal magnetization can move, known as magnetic anisotropy, must be tuned so that the particles respond on the same timescale as the applied field. Under the conditions tested here, manganese ferrite offered the best balance, making it the more promising candidate. Graphene oxide helps keep particles dispersed and offers handles for future drug targeting, but it also comes with a cost in heating strength. Future designs will need to balance these trade-offs, adjusting particle size, shape, surface coating, and the carbon support to create safe, efficient nano-heaters that can be guided precisely to tumors.
Citation: Ramadan, W., Gasser, A., Ramadan, A. et al. The impact of graphene oxide on the magnetic and hyperthermia properties of CoFe2O4 and MnFe2O4 ferrites. Sci Rep 16, 14736 (2026). https://doi.org/10.1038/s41598-026-51345-w
Keywords: magnetic hyperthermia, ferrite nanoparticles, graphene oxide, cancer therapy, nanomedicine