Clear Sky Science · en
Interpretable, physics-informed learning reveals sulfur adsorption and poisoning mechanisms in 13-atom icosahedra nanoclusters
Why tiny metal clusters and sulfur matter
Many industrial catalysts are slowly “poisoned” by sulfur: atoms from fuels and exhaust gases cling to metal surfaces and shut down the reactions we want. This study zooms in to an extreme scale—metal particles made of just 13 atoms—to understand, atom by atom, how sulfur latches on, how it can damage these tiny catalysts, and how certain metals manage to stay both active and resilient. The insights provide design rules for future, sulfur-tolerant catalysts used in energy and environmental technologies.
Little metal cages at the nanoscale
The researchers focus on a simple but powerful model system: 13-atom metal clusters arranged in an icosahedron, a highly symmetric cage with one atom in the center and 12 at the corners. They systematically build such cages from 30 different transition metals, spanning three rows of the periodic table. Using quantum-mechanical simulations, they first examine the “bare” clusters: how tightly the atoms bind together, how they vibrate, how electrons are distributed, and how easily these nanoscale cages might react. Even at this tiny size, clear trends emerge from one metal to the next, reflecting how their outer electrons are filled.
How sulfur sticks and starts poisoning
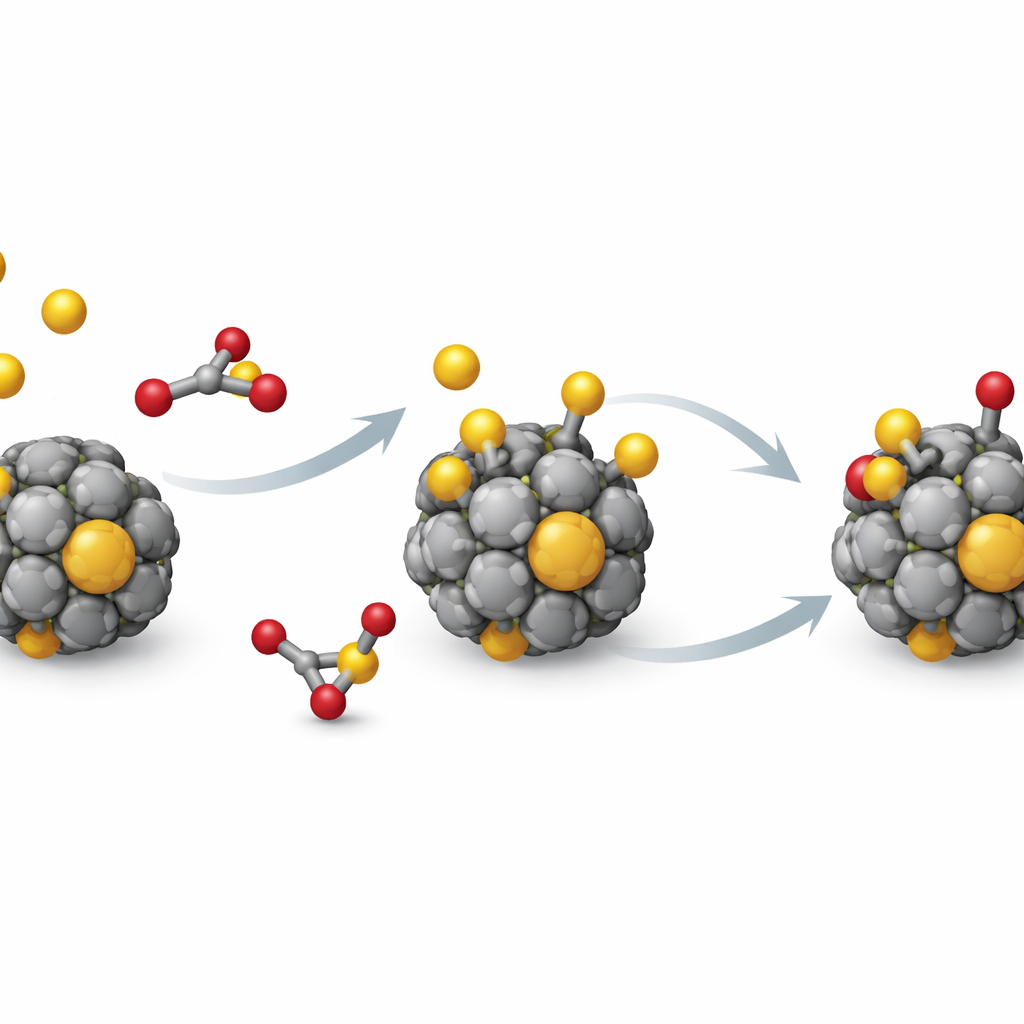
Next, the team adds single sulfur atoms to the clusters and lets the structures relax to their preferred shapes. Sulfur can land on three types of spots on the cage—directly above one atom, bridging two atoms, or sitting in a small hollow surrounded by three atoms. For most metals, sulfur prefers the hollow site, where it can bond to several neighbors at once. The simulations show that sulfur binding is always energetically favorable and often very strong, which explains why sulfur poisoning is such a stubborn problem. In many cases, the metal–sulfur attraction dominates, while the metal cage itself distorts only moderately—though for some metals, sulfur can trigger large reshaping of the cluster.
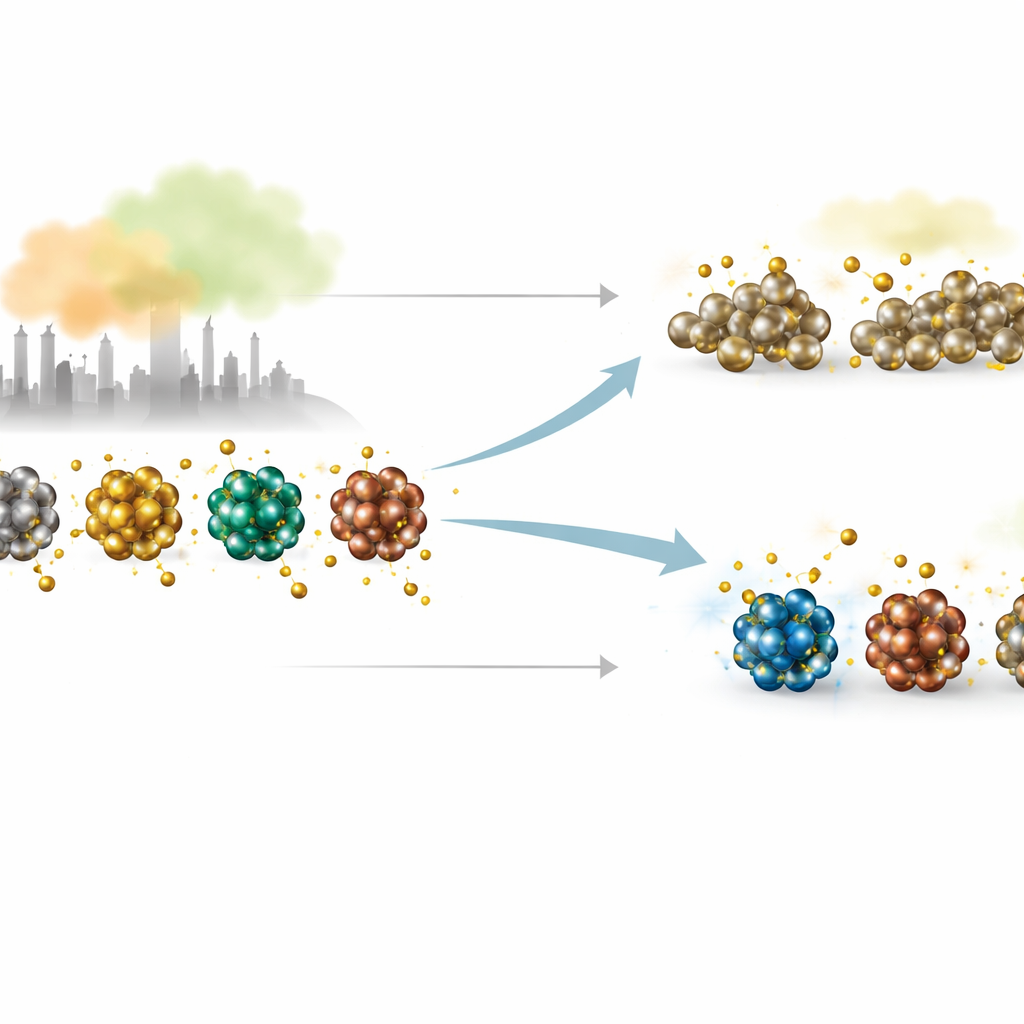
Patterns across the periodic table
Because these clusters are so small, simple one-number rules do not fully capture how strongly sulfur will bind. To make sense of the complex data, the authors combine their physics-based calculations with machine learning. They feed the models a rich set of descriptors: bond lengths and coordination, how stiff or soft the vibrations are, how the electronic states are arranged, and how much charge flows from metal to sulfur. Unsupervised learning groups metals that behave similarly when sulfur binds, while regression models test which underlying properties are most useful for predicting sulfur adsorption. Together, these analyses reveal periodic patterns and highlight where sulfur drives gentle surface changes versus major structural rearrangements that could degrade a catalyst.
A trio of tough, balanced candidates
From this data-driven map, one standout group appears: clusters made of titanium, zirconium, and hafnium. These three metals share the same number of outer electrons and show remarkably similar behavior across all descriptors. Sulfur binds to their 13-atom cages strongly enough to activate sulfur-containing molecules, but not so strongly that the cage collapses or reshapes dramatically. To test this further, the authors study a more realistic pollutant, sulfur dioxide (SO2), on these three clusters. Their simulations reveal that SO2 tends to break apart on contact, forming robust metal–sulfur and metal–oxygen bonds, yet the metal cage largely maintains its overall structure—a promising balance between activity and durability.
What this means for future catalysts
In plain terms, the study shows that not all tiny metal particles succumb to sulfur in the same way. By carefully linking structure, vibrations, and electronic behavior to sulfur binding, the authors identify which metals are more likely to survive in sulfur-rich environments. The titanium–zirconium–hafnium trio, in particular, hits a sweet spot: they grab sulfur strongly enough to help break down harmful sulfur gases, but they resist the severe damage that would shut a catalyst down. These insights, distilled from detailed quantum calculations and interpretable machine learning, offer practical guidelines for designing next-generation, sulfur-tolerant nanocatalysts.
Citation: Monteiro, R.F., Palheta, J.M.T., Grison, T.G. et al. Interpretable, physics-informed learning reveals sulfur adsorption and poisoning mechanisms in 13-atom icosahedra nanoclusters. Sci Rep 16, 14174 (2026). https://doi.org/10.1038/s41598-026-50998-x
Keywords: nanocatalysts, sulfur poisoning, transition metal clusters, density functional theory, machine learning in materials science