Clear Sky Science · en
Elucidation and functional characterization of the biosynthetic pathway of the natural sweetener phyllodulcin in Hydrangea macrophylla
A plant that makes an ultra-sweet leaf
Imagine a cup of tea made from leaves that are naturally hundreds of times sweeter than sugar, yet add almost no calories. The shrub Hydrangea macrophylla does exactly this by producing a compound called phyllodulcin, long used in traditional Japanese "sweet tea" and now attracting attention as a potential natural sweetener and medicinal ingredient. This study asks a deceptively simple question: how does the plant actually make this powerful sweet molecule inside its leaves?
From common building blocks to special sweetness
Plants manufacture an enormous variety of specialized chemicals from a small set of basic ingredients. In Hydrangea macrophylla, phyllodulcin belongs to a family of compounds that all start from the amino acid phenylalanine. The researchers collected 182 different hydrangea varieties and measured the amounts of phyllodulcin and a closely related compound called hydrangenol in their leaves. Some varieties produced a lot of these sweet molecules, others very little, and a related species, Hydrangea paniculata, produced none at all. This natural variation provided a living laboratory for tracing which internal chemical routes lead toward, or away from, phyllodulcin.
Following the chemical trails inside the leaf
The team next profiled 14 key intermediate compounds from the broader phenylpropanoid pathway, a central chemical route in many plants. Varieties rich in phyllodulcin tended to have high levels of phenylalanine, p-coumaric acid, naringenin, resveratrol, umbelliferone and a derivative called thunberginol C. In contrast, plants with little or no phyllodulcin accumulated more caffeic and ferulic acids and several related coumarins, suggesting that, in those plants, the chemical flow is diverted into side routes that do not end in the sweetener. Statistical analyses showed strong positive correlations between phyllodulcin and the "high" set of intermediates, and strong negative correlations with the "low" set, indicating preferred and alternative branches within the plant’s internal chemistry.
Reading the plant’s genetic recipe
Chemical clues alone cannot reveal which enzymes actually perform each step, so the scientists also examined gene activity in leaves from selected high- and low-sweetness varieties, plus the non-sweet species. Using RNA sequencing, they compared which genes were switched on or off and mapped them onto known metabolic pathways. In plants that made a lot of phyllodulcin or hydrangenol, genes associated with phenylpropanoid, flavonoid and stilbene production were strongly enriched. A network analysis that groups co-active genes into modules showed that certain gene clusters correlated tightly with the levels of phyllodulcin, hydrangenol and key intermediates like p-coumaric acid and resveratrol, hinting at shared control circuits.
Key steps on the road to the sweet compound
Among the many genes, several stood out as plausible drivers of phyllodulcin formation. Varieties high in the sweet compound strongly expressed genes for enzymes such as p-coumaroyltriacetic acid synthase (CTAS), type III polyketide synthases, ketoreductases, polyketide cyclases and a resveratrol-modifying enzyme (ROMT). These enzymes can build or reshape ring-like molecules that resemble known intermediates on the path to phyllodulcin and hydrangenol. In low-sweetness plants, a different set of genes that funnel p-coumaric acid toward caffeic and ferulic acids was more active, reinforcing the idea of a metabolic fork where one branch leads to sweetness and the other to unrelated compounds. The non-sweet Hydrangea paniculata largely lacked or underused the critical enzymes seen in the sweet-leaf plants.
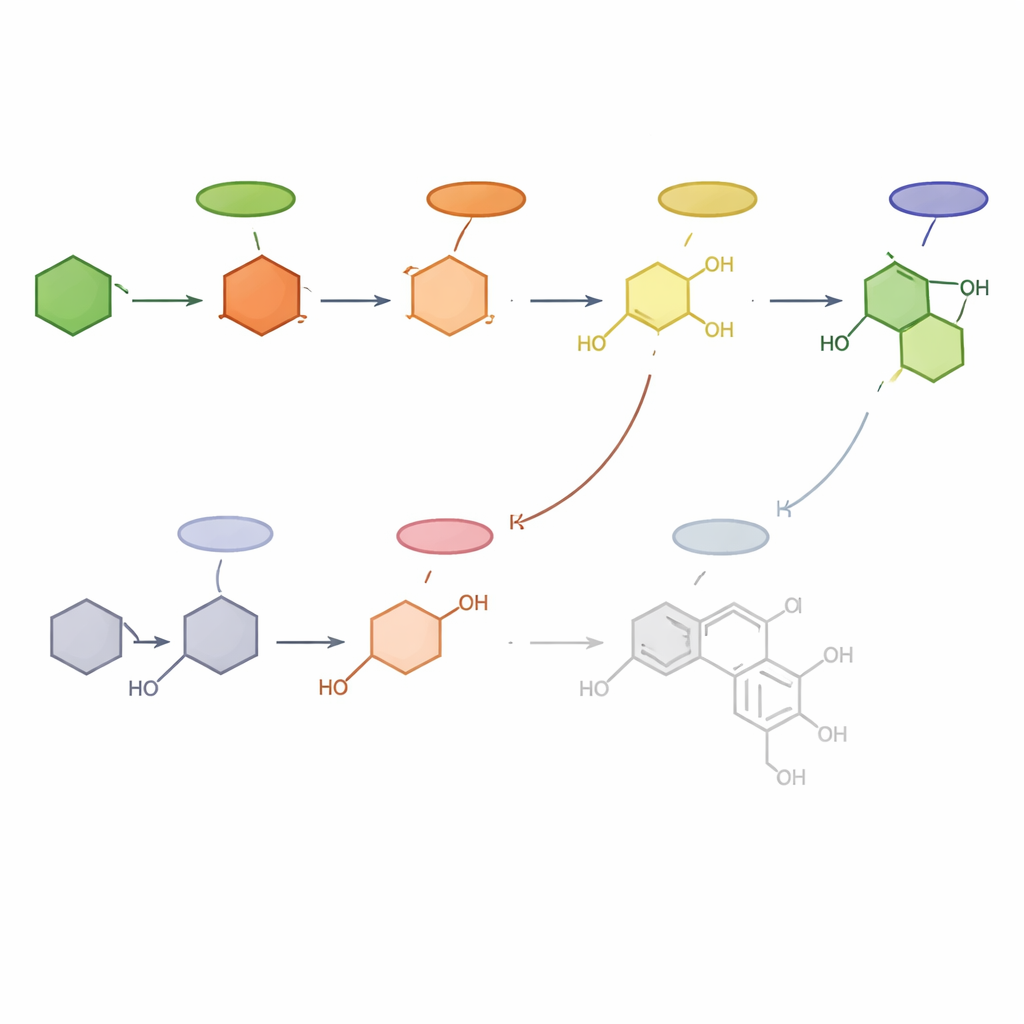
Drawing the new map of a sweet pathway
By combining the chemical fingerprints with gene activity patterns, the researchers propose a detailed working model for how Hydrangea macrophylla builds phyllodulcin. In their view, the pathway begins with phenylalanine and moves through p-coumaric acid, then branches into at least three routes involving hydrangenol, resveratrol and thunberginol C, with the latter likely serving as the final direct precursor. In varieties where side routes dominate, much less phyllodulcin is made. Although some steps remain hypothetical, this map transforms a once-mysterious traditional sweetener into a well-defined biochemical product. For non-specialists, the takeaway is that understanding this natural factory at the molecular level opens the door to breeding sweeter hydrangea varieties, improving sustainable production of phyllodulcin, and exploring its promising health-related properties in a more targeted way.
Citation: Padmakumar Sarala, G., Engel, F., Hartmann, A. et al. Elucidation and functional characterization of the biosynthetic pathway of the natural sweetener phyllodulcin in Hydrangea macrophylla. Sci Rep 16, 12044 (2026). https://doi.org/10.1038/s41598-026-47892-x
Keywords: natural sweeteners, Hydrangea, plant metabolism, phyllodulcin, biosynthetic pathways