Clear Sky Science · en
Synthetically designed circRNA can be used to target cardiovascular relevant microRNAs to improve cellular function
Why this matters for heart health
As we age, our blood vessels stiffen and misbehaving cells in the vessel wall help drive heart attacks, strokes, and other cardiovascular diseases. This study explores a new kind of RNA-based molecule, built in the lab as a tiny loop, that can soak up harmful regulatory RNAs inside vascular cells. By taming these small but powerful molecules, the researchers hope to gently nudge aging blood vessels back toward healthier behavior.
Small genetic switches that go wrong with age
Inside our cells, short RNA snippets called microRNAs act as fine-tuning knobs for gene activity. Two of them, named miR-21-5p and miR-146a-5p, become overactive in aging blood vessels and are linked to inflammation, scarring, and excessive growth of vascular smooth muscle cells, the contractile cells that help vessels keep their shape. When these microRNAs are too abundant, they silence protective genes that normally restrain cell proliferation and dampen inflammation, contributing to the gradual damage seen in cardiovascular disease.
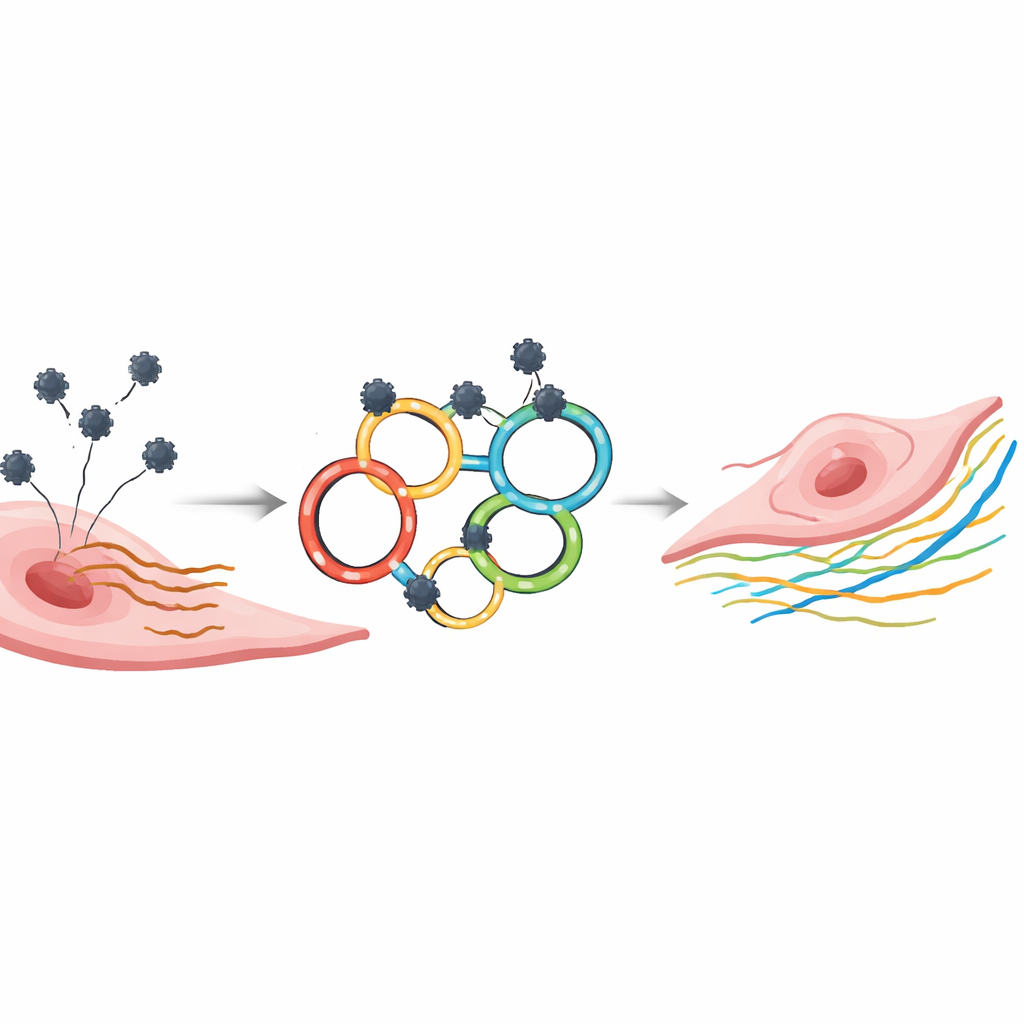
Turning natural RNA loops into therapeutic tools
Nature already uses circular RNA loops as sponges that bind microRNAs and keep them from interfering with other RNAs. Inspired by this, the team engineered synthetic circular RNAs, or circRNAs, each carrying several carefully tuned docking sites for either miR-21-5p or miR-146a-5p. Because these constructs are closed loops, they resist the cellular enzymes that quickly chew up conventional, straight RNA drugs. The researchers produced these circRNAs in vitro, purified them to remove leftover linear pieces, and confirmed their circular shape and stability using gel-based methods and northern blotting.
Testing the loops in aging vessel cells
To see whether these circRNAs could actually improve cell behavior, the scientists worked with human vascular smooth muscle cells and endothelial cells grown in culture and pushed into an early aging-like state. In these senescent cells, levels of miR-21-5p and miR-146a-5p were indeed higher than in younger cells. When the researchers introduced low doses of their circular constructs, they saw the circRNA levels peak within about a day and then slowly decline over several days. During this window, the targeted microRNAs fell, indicating that the loops were successfully tying up the excess microRNA molecules.
Stronger gene effects than standard anti-miR drugs
The team compared their circular designs to standard antisense “anti-miR” oligonucleotides, which are straight pieces of RNA that directly neutralize microRNAs but are less stable. Importantly, similar drops in miR-21-5p and miR-146a-5p were achieved using much lower concentrations of circRNAs. In aging smooth muscle cells, circRNAs aimed at miR-21-5p boosted the levels of its known protective targets, such as PTEN and PDCD4, more clearly than the anti-miR approach. CircRNAs against miR-146a-5p likewise increased several genes involved in controlling inflammation and cell growth. In functional tests, miR-21–targeting circRNAs modestly reduced the migration of smooth muscle cells, a behavior that, when excessive, contributes to vessel wall thickening. Endothelial cells showed more subtle gene changes but displayed improvements in migration and proliferation, consistent with better repair capacity.
Promise and challenges of RNA circles for therapy
Although this work was done entirely in dishes, it highlights several advantages of circular RNAs over traditional microRNA blockers: they are more stable, work at lower doses, and provide sustained, though time-limited, reshaping of gene activity. The authors also point out the remaining hurdles, such as ensuring high purity to avoid unwanted immune responses and finding safe, efficient carriers that can deliver these loops into blood vessel cells throughout the body. Nonetheless, their results suggest that custom-designed RNA circles could one day form the basis of tailored treatments to calm harmful microRNAs and help preserve healthier blood vessels as we age.
Citation: Böttcher, S., Kalies, K., Knöpp, K. et al. Synthetically designed circRNA can be used to target cardiovascular relevant microRNAs to improve cellular function. Sci Rep 16, 12023 (2026). https://doi.org/10.1038/s41598-026-46870-7
Keywords: circular RNA therapy, vascular aging, microRNA inhibition, cardiovascular disease, vascular smooth muscle cells