Clear Sky Science · en
Distinct 72-hour fluid balance trajectories are associated with 28-day mortality in a multicenter cohort of sepsis patients
Why hospital fluids matter in deadly infections
When people develop sepsis, a life threatening reaction to infection, doctors urgently give fluids through a vein to keep blood pressure up and organs working. But how much fluid is enough, and how much becomes too much, is still hotly debated. This study followed thousands of intensive care patients with sepsis to see how their fluid balance over the first three days in the ICU related to their chances of survival.
Different paths of fluid gain and loss
Instead of taking a single snapshot of how much fluid patients had on board, the researchers traced each person’s net fluid balance over 72 hours after sepsis was diagnosed. Using data from three large US hospital databases, they grouped more than 16,000 patients by how their fluid status changed over time. Five clear patterns emerged, ranging from patients who moved toward a neutral or negative balance to those who remained heavily overloaded with fluid.
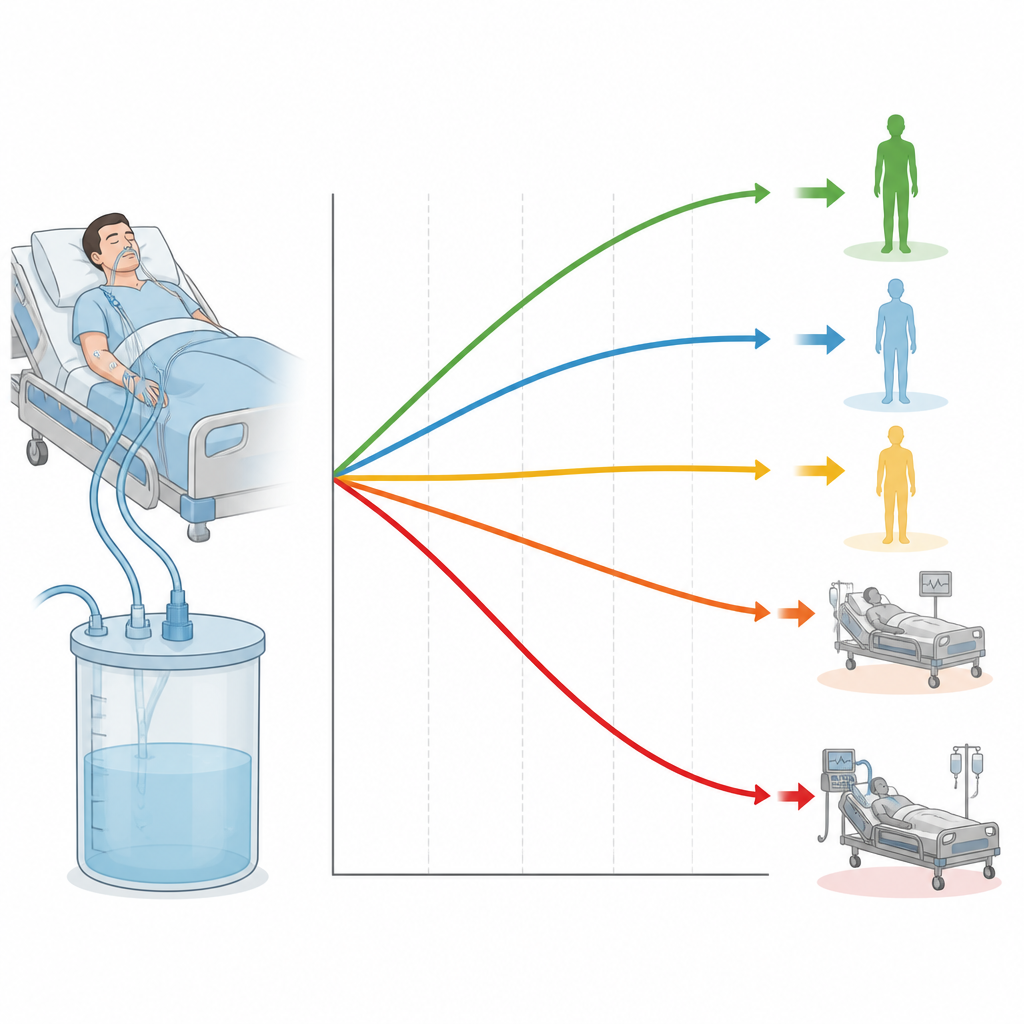
Five fluid patterns with very different risks
One group, called “Persistent Negative Balance,” started with only a small excess and gradually moved toward even or slightly negative fluid levels. Another group kept a low or medium, steady positive balance. A fourth, the largest group, began with a high positive balance but then showed a steep and steady drop as excess fluid was removed. The final group stayed with a very high positive balance for the full 72 hours, meaning they continued to carry a large fluid load. These patterns were not rare quirks of one hospital; they appeared again in two separate validation cohorts from different times and centers.
Who did better and who did worse
When the team linked these fluid paths to deaths within 28 days, the contrasts were striking. Patients who remained heavily overloaded with fluid had the highest death rate, along with more time on a ventilator and greater need for kidney support. In contrast, those who either stayed near or below even balance or started high and then rapidly shed fluid had the best survival. Crucially, these links held up even after accounting for how sick patients were at the start, their other illnesses, vital signs, lab results, and treatments like ventilators and blood pressure drugs.
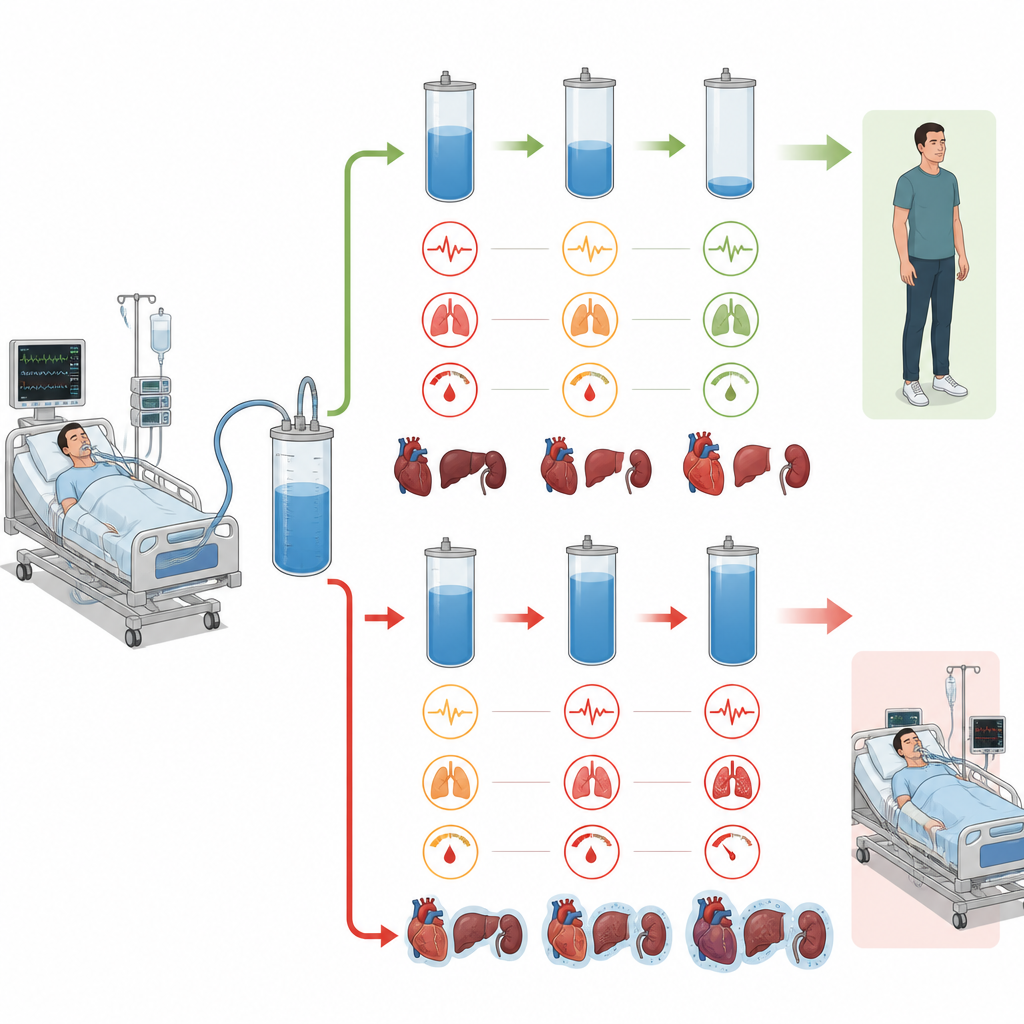
Clues from organs under strain
The sickest high fluid group also showed stubbornly low blood pressure, fast heart rates, and worsening kidney and heart function over the three days. This suggests that in some patients, extra fluid is not just a marker of severe illness but part of a vicious cycle: organs fail, the body cannot clear fluid, and added fluid may further stress the heart, lungs, and kidneys. By contrast, patients who could be safely “de-resuscitated” after the initial emergency phase seemed to escape this spiral and had outcomes similar to those who were never very overloaded.
Using smart tools to spot danger early
To make these insights more useful at the bedside, the researchers trained machine learning models to predict, within the first 24 hours, which patients would fall into the most dangerous persistent high fluid group. Their best model reached good accuracy in all three datasets and highlighted straightforward clinical features such as overall illness severity, blood lactate levels, and calcium levels as key signals of risk. This raises the possibility of an early warning system that alerts clinicians when a patient is likely to stay fluid overloaded unless care is adjusted.
What this means for patients and clinicians
For people with sepsis and their families, the study does not offer a simple fluid recipe. Instead, it shows that the pattern of fluid gain and loss over several days matters at least as much as the total amount given at one time. Patients who stay swollen with fluid for days fare far worse than those whose teams manage to turn the corner and gently remove the excess once circulation stabilizes. In other words, getting fluid early can be life saving, but failing to back off and let the body dry out again can be deadly. Watching fluid balance as a moving curve rather than a static number may help guide more personalized, safer care in sepsis.
Citation: Wang, K., Xiong, H., Zhu, Y. et al. Distinct 72-hour fluid balance trajectories are associated with 28-day mortality in a multicenter cohort of sepsis patients. Sci Rep 16, 15126 (2026). https://doi.org/10.1038/s41598-026-46063-2
Keywords: sepsis, fluid balance, intensive care, organ failure, machine learning