Clear Sky Science · en
Synthesis, antioxidant and antimicrobial activities, molecular docking study of new pyrimidine derivatives
New Weapons Against Tough Germs
Antibiotic resistance and hard‑to‑treat fungal infections are making once‑routine illnesses more dangerous again. This study describes a family of newly built small molecules designed to both disarm harmful microbes and mop up damaging “rust‑like” chemicals in our bodies. By blending classic organic chemistry with modern computer simulations, the researchers home in on one standout compound that hits a key bacterial enzyme while also showing strong antioxidant power, pointing toward future medicines that could fight infection on several fronts at once.
Building a Versatile Chemical Scaffold
The team focused on pyrimidines, a class of ring‑shaped molecules that already appear in many drugs and even in our own DNA. Starting from a simple building block called a chalcone, they stepped through a series of reactions to construct more complex, fused rings containing sulfur and nitrogen. This strategy generated a small library of related compounds, labeled 3 through 11, each carrying slightly different side groups. Those subtle changes in shape and chemical makeup were intended to tune how strongly the molecules could interact with biological targets such as bacterial enzymes or unstable oxygen‑based species linked to cell damage.
Testing Antioxidant Power
To see whether these compounds could neutralize harmful free radicals, the scientists used a common lab test based on a purple dye known as DPPH. When an antioxidant donates an electron or hydrogen atom to this dye, the solution fades, and the degree of fading reveals how potent the antioxidant is. Several of the new molecules showed noticeable activity, but three—called 5, 9a, and especially 11—stood out. At the tested doses, they reduced the dye’s signal nearly as well as, or better than, the widely used synthetic antioxidant BHT. Compound 11 required an even lower concentration than the control to cut the radical level in half, marking it as the strongest radical scavenger in the series.
Fighting Bacteria and Fungi
The same set of molecules was then challenged against a small panel of disease‑causing microbes: two common bacteria, Escherichia coli and Bacillus subtilis, and two troublesome fungi, Aspergillus niger and Candida albicans. In petri‑dish tests, solutions of the compounds were allowed to diffuse through agar containing these organisms. Clear halos around the wells showed where growth had been blocked. Most compounds produced moderate zones of inhibition, but again 5, 9a, and 11 were exceptional, carving out broad, strong halos against all four species. In some cases, compound 11’s performance closely matched that of standard drugs such as streptomycin for bacteria and cycloheximide for fungi, suggesting it could serve as a promising lead for a new broad‑spectrum antimicrobial.
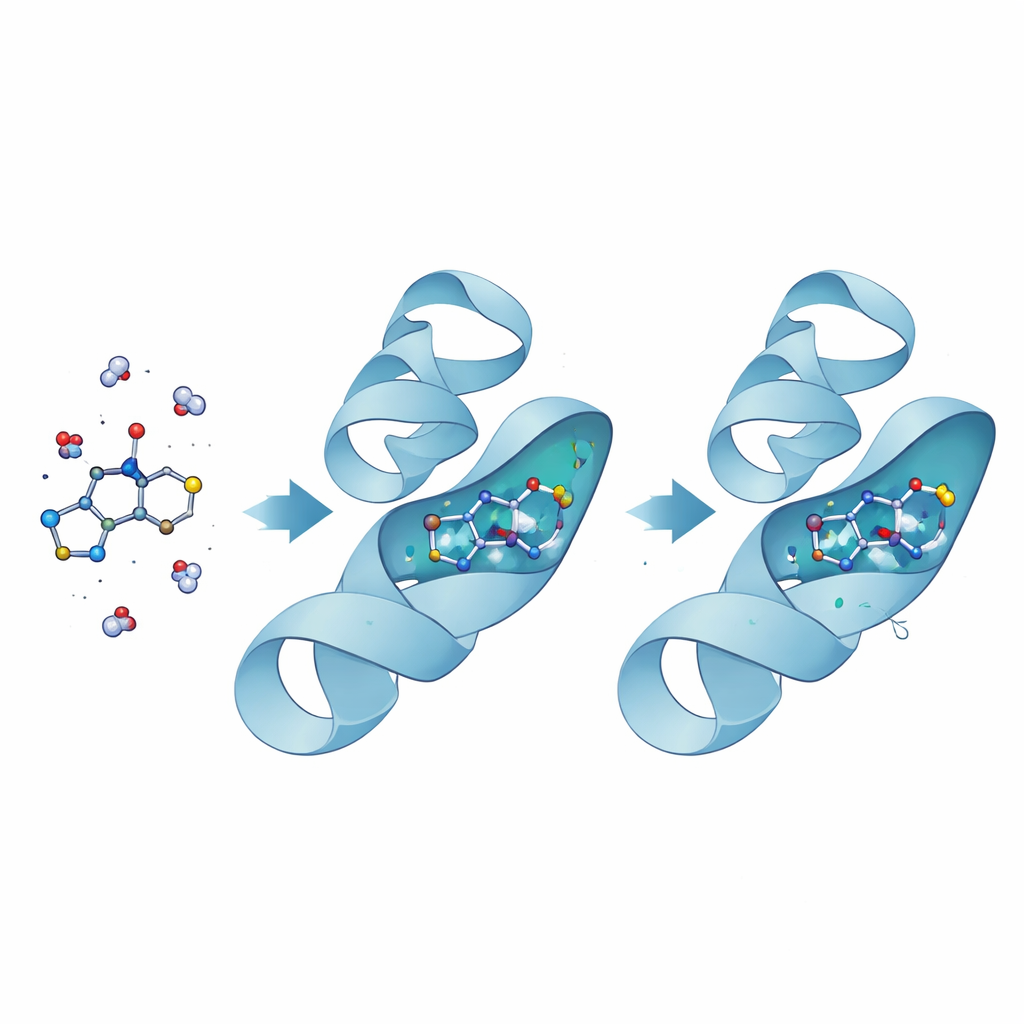
Watching a Molecule Grip Its Target
To understand why compound 11 was so effective against bacteria, the researchers turned to computer modeling of its interaction with DNA gyrase, a key enzyme that helps bacterial DNA coil and uncoil. Docking calculations first positioned the molecule inside the enzyme’s energy‑using pocket, where it appeared to fit snugly. Long, detailed molecular dynamics simulations then followed how the complex behaved over tens of nanoseconds of virtual time. With compound 11 bound, the enzyme’s structure became slightly more compact and less wobbly, especially around the active site, indicating a tight and stabilizing grip. Calculations of binding energy showed that attractive forces between the drug and the protein comfortably outweighed the energetic cost of pushing water molecules aside, confirming a strong, favorable interaction.
Why This Matters for Future Medicines
Taken together, the chemistry, lab tests, and computer simulations paint a coherent picture. By carefully shaping pyrimidine‑based rings, the team created molecules that not only soak up harmful radicals but also latch firmly onto a vital bacterial enzyme. Compound 11, in particular, combines potent antioxidant behavior with antibacterial and antifungal strength on par with existing drugs, supported by a clear, atomic‑level explanation of how it works. While these molecules are still at an early, experimental stage, they showcase how blending synthesis, biological screening, and digital modeling can speed the search for new treatments that tackle infection and oxidative damage at the same time.
Citation: Khalaf, H.S., El-Rashedy, A.A., Abd El-Gwaad, A.A. et al. Synthesis, antioxidant and antimicrobial activities, molecular docking study of new pyrimidine derivatives. Sci Rep 16, 12354 (2026). https://doi.org/10.1038/s41598-026-45654-3
Keywords: pyrimidine derivatives, antimicrobial agents, antioxidants, DNA gyrase inhibitors, drug design