Clear Sky Science · en
Characterization, immune cell phenotyping and differential gene expression analysis of brain metastases and primary breast cancer samples
Why the Spread of Breast Cancer to the Brain Matters
As treatments for breast cancer improve, many women are living longer—but this extra time has revealed a serious problem. More patients are developing tumors in the brain, called brain metastases, which are hard to treat and often lead to severe symptoms. This study asks a simple but crucial question: how different are these brain tumors from the original breast tumors they came from, especially in how they interact with the immune system? The answers could shape future therapies for one of the most feared complications of breast cancer. 
Taking a Paired Look Inside the Body
The researchers examined tumor samples from ten women who had both a primary breast tumor and a brain metastasis removed. This paired approach allowed them to compare, for each patient, how the cancer had changed over time and after treatment. They used several laboratory techniques: traditional staining to see key proteins that guide treatment decisions; measurements of immune cells within and around the tumor; and modern RNA sequencing to read out which genes were turned on or off. Together, these methods offered a layered view of both the cancer cells and the surrounding immune “neighborhood.”
Shifting Identities of the Tumor Cells
One major finding was that the identity of the cancer, as doctors usually define it, can change when it reaches the brain. In four out of ten patients, the brain metastasis no longer matched the original breast tumor in important markers such as estrogen receptor and HER2. In some cases, tumors lost hormone sensitivity; in others, the brain lesions gained HER2, a change that might open or close doors to targeted drugs. When the team used gene-based classification, a more global way to categorize breast cancers, they again saw that some tumors shifted subtype between breast and brain. These changes highlight that relying only on the original tumor for treatment decisions may miss critical information once the disease has spread.
An Immune Landscape That Grows Colder
The most striking contrast emerged in the immune environment. Using RNA data, the team estimated what kinds of immune cells were present in each sample. Primary breast tumors tended to host more B cells, plasma cells, and CD8 “killer” T cells—cells that are generally associated with the body’s ability to recognize and attack cancer. In the brain metastases, these helpful cells were much scarcer. Instead, there was a relative rise in cells linked with a dampened or “peacekeeping” immune state, such as M2-type macrophages and certain helper T cells. This pattern suggests that by the time breast cancer cells settle in the brain, they inhabit or help create a setting that blunts immune attack. 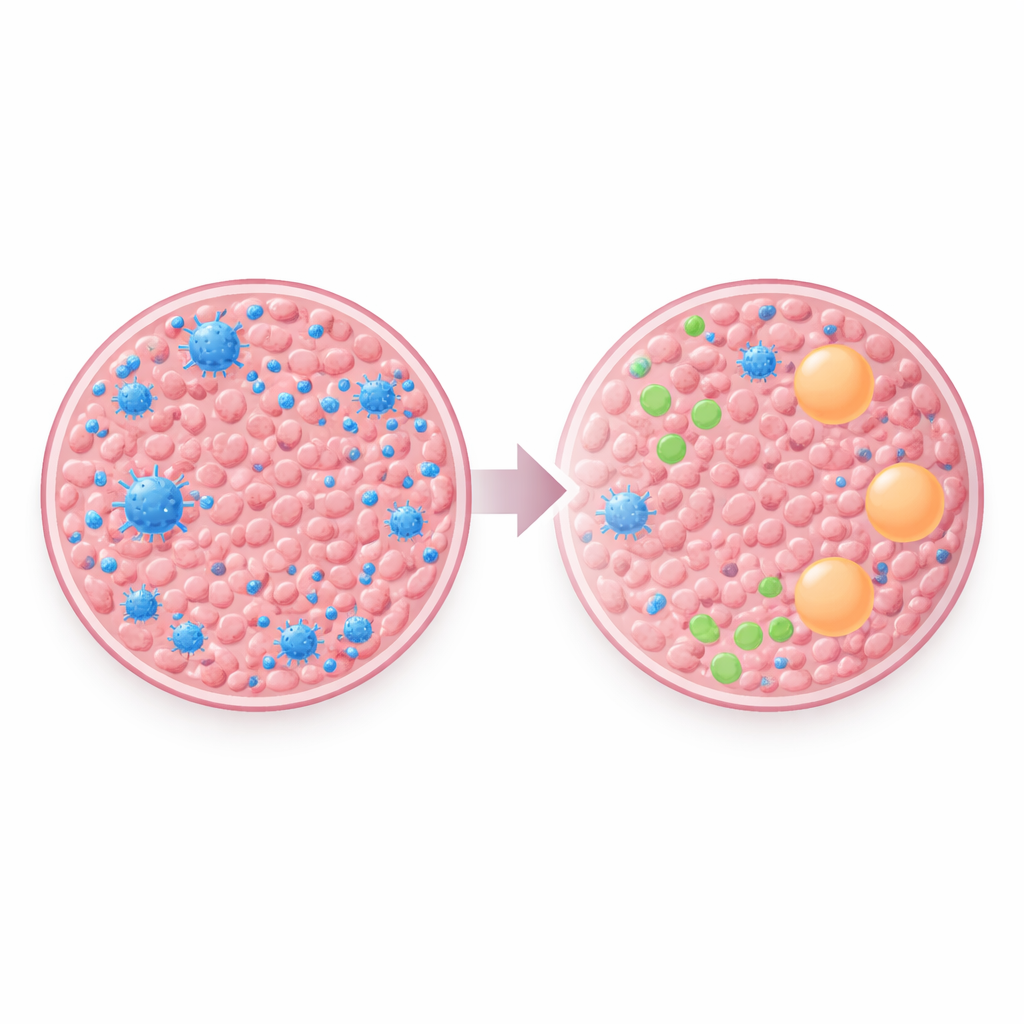
Energy Use and Signaling Also Change
Gene activity patterns told an additional story. In primary breast tumors, the most active gene groups were tied to immune and inflammatory signals, echoing the richer presence of fighting immune cells. In brain metastases, in contrast, genes involved in cellular power production—such as those guiding mitochondrial energy pathways—were more prominent. While the study did not directly test metabolism, this shift hints that brain metastases may rewire how they generate energy, perhaps to suit the brain’s unique environment or to support their growth under immune pressure.
What This Means for Future Treatments
To a lay observer, these findings help explain why current immunotherapies, such as drugs that release the brakes on immune cells, have not yet shown strong results in breast cancer that has reached the brain. The tumors found there appear to live in a more immune-silent, or even actively immunosuppressive, niche with fewer of the very cells these drugs depend on. Because the study involved only ten patients and mainly observational methods, its conclusions are preliminary. Still, it strongly suggests that brain metastases from breast cancer are not simple copies of the original tumors: they differ in key markers, in how they talk to the immune system, and possibly in how they fuel themselves. Understanding and targeting this altered landscape will be essential for designing future therapies that can truly protect the brain.
Citation: Gassner, F.J., Rinnerthaler, G., Castagnaviz, V. et al. Characterization, immune cell phenotyping and differential gene expression analysis of brain metastases and primary breast cancer samples. Sci Rep 16, 10721 (2026). https://doi.org/10.1038/s41598-026-45607-w
Keywords: breast cancer brain metastases, tumor microenvironment, immune evasion, immunotherapy resistance, RNA sequencing