Clear Sky Science · en
Tailoring the emission color in Sm3+/Li+-doped calcium hydroxyapatite nanocrystals: a path toward cryogenic luminescent thermosensors
Glowing Thermometers for Extreme Cold
Keeping cells, tissues, and even organs alive at very low temperatures is crucial for modern medicine, from fertility treatments to organ banking. But inside a freezing vial, knowing the exact temperature right where the cells sit is surprisingly hard. This study presents a tiny, light-emitting thermometer made from a bone-like material whose glow changes color with temperature, offering a promising way to monitor how cold things really are during deep-freeze procedures.
A Nanomaterial Inspired by Bone
The researchers built their thermometer from hydroxyapatite, a mineral that closely resembles the main inorganic component of human bone and teeth. Because this material is already known to be biocompatible, it is an attractive starting point for medical uses. They created nanocrystals of hydroxyapatite and then substituted a small fraction of the calcium ions with two other ions: samarium, which shines in orange-red when excited by light, and lithium, which helps balance electric charge and fine-tune the crystal structure. These substitutions do not destroy the structure of the mineral but subtly distort it, creating tiny imperfections that turn out to be crucial for its light-emitting behavior.
How Light Reveals Temperature
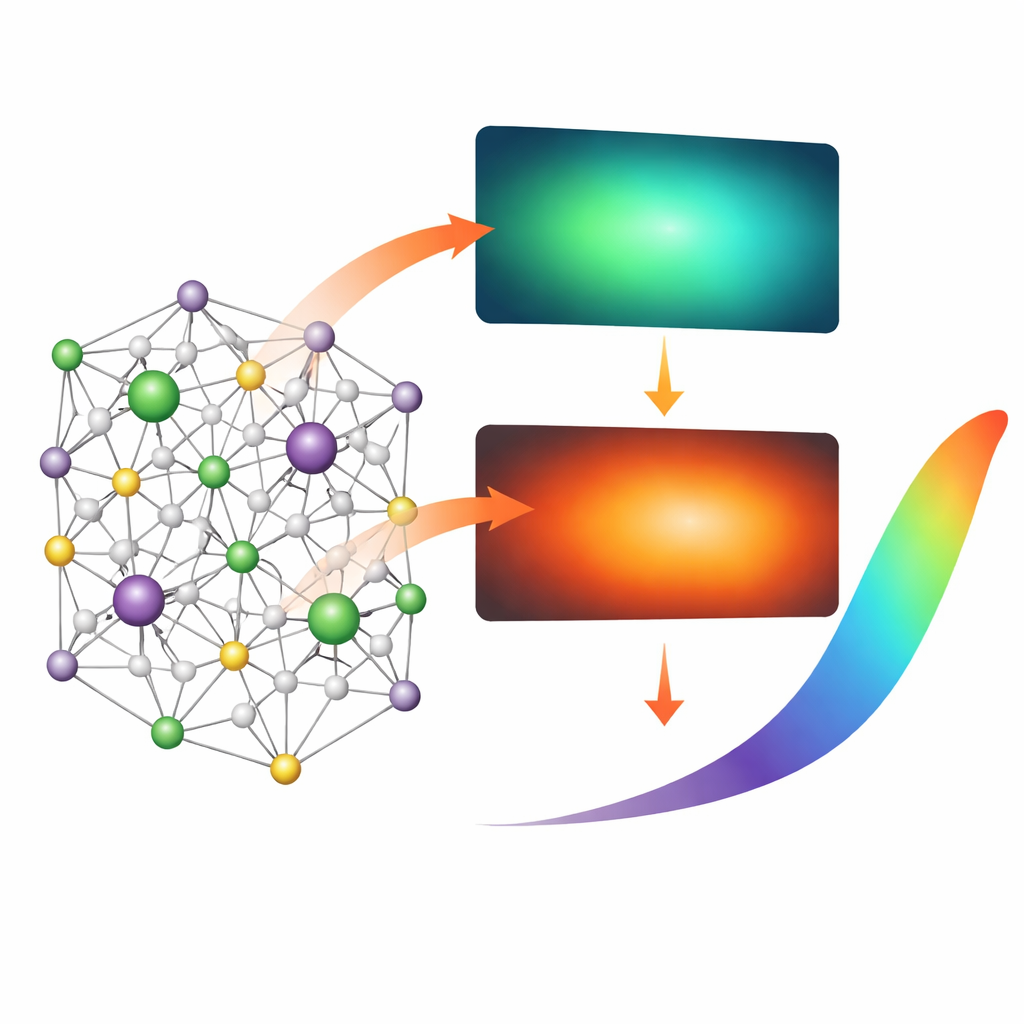
When the nanocrystals are illuminated with near-ultraviolet light, two kinds of glow appear. One is a broad blue-green emission coming from defects and irregularities in the hydroxyapatite lattice. The other consists of sharp orange-red lines produced by the samarium ions embedded in the structure. At room temperature, the orange-red light dominates, but as the material is cooled toward liquid-nitrogen temperatures, the blue-green glow grows stronger while the samarium emission remains relatively stable. As a result, the overall color shifts smoothly from blue-green at 77 K (about −196 °C) to orange-red at 300 K (about 27 °C). This predictable color change allows temperature to be read out simply by measuring how much blue-green and orange-red light the material emits.
Engineering Defects for Better Sensing
To understand and optimize this behavior, the team carefully analyzed the crystal structure and optical properties of the nanocrystals. X-ray diffraction showed that samarium slightly expands the lattice, while the smaller lithium ions cause a gentle contraction, together generating a controlled pattern of distortions and vacancies. These features create extra energy levels within the material’s band gap, which are responsible for the blue-green defect emission. At low temperatures, these defect states release their energy as light; at higher temperatures, vibrations in the lattice open up non-radiative pathways that quench the blue-green glow. By comparing samples with different lithium contents, the authors found that lithium not only compensates charge but also suppresses unwanted, non-radiative defects and lengthens the light-emission lifetimes of samarium. The best-performing composition used 1 mol% samarium and 5 mol% lithium, which produced a stable reference orange-red signal and a strongly temperature-sensitive blue-green signal.
Reading Temperature in the Deep Freeze
The key to using this material as a thermometer is the ratio between the blue-green and orange-red emissions. Because the orange-red samarium light hardly changes with temperature while the blue-green band fades as it warms, their intensity ratio tracks temperature in a simple, nearly exponential way. Over the range from 77 K to 300 K, the authors measured how this ratio varies and calculated both absolute and relative sensitivity. They found that the material is particularly responsive at cryogenic temperatures, with the highest relative sensitivity (0.025 K⁻¹) between 200 and 225 K and a strong absolute sensitivity at 77 K. These values compare favorably with other samarium-based optical thermometers, which typically work best at much higher temperatures and are not optimized for deep-freeze conditions.
Why This Matters for Frozen Cells
The study concludes that samarium and lithium co-doped hydroxyapatite nanocrystals act as efficient, biocompatible luminescent nanothermometers tailored for cryogenic use. Their color changes smoothly and predictably with temperature below room temperature, and the tiny particles can, in principle, be placed close to cells or tissues without causing harm. In practical terms, this means that during cryopreservation—when ice crystals and small temperature missteps can damage delicate biological material—these glowing nanothermometers could provide real-time, local temperature readouts. That capability could help refine freezing and thawing protocols, improve survival rates of stored samples, and bring more control and safety to a wide range of cryogenic biomedical technologies.
Citation: Sobierajska, P., Wiglusz, R.J. Tailoring the emission color in Sm3+/Li+-doped calcium hydroxyapatite nanocrystals: a path toward cryogenic luminescent thermosensors. Sci Rep 16, 10708 (2026). https://doi.org/10.1038/s41598-026-45561-7
Keywords: luminescent nanothermometers, cryopreservation, hydroxyapatite nanoparticles, samarium doping, temperature sensing