Clear Sky Science · en
Chromatin dynamics of the Klf4 locus in mouse pluripotent cells
How DNA Moves Inside Living Cells
Deep inside each of our cells, long strands of DNA fold and twist into a packed form called chromatin. How this chromatin wiggles and drifts through the nucleus matters, because distant pieces of DNA often need to meet to switch genes on or off. This study looks at whether turning a gene on actually changes how its stretch of DNA moves, using a key stem cell gene called Klf4 as a test case in living mouse cells.
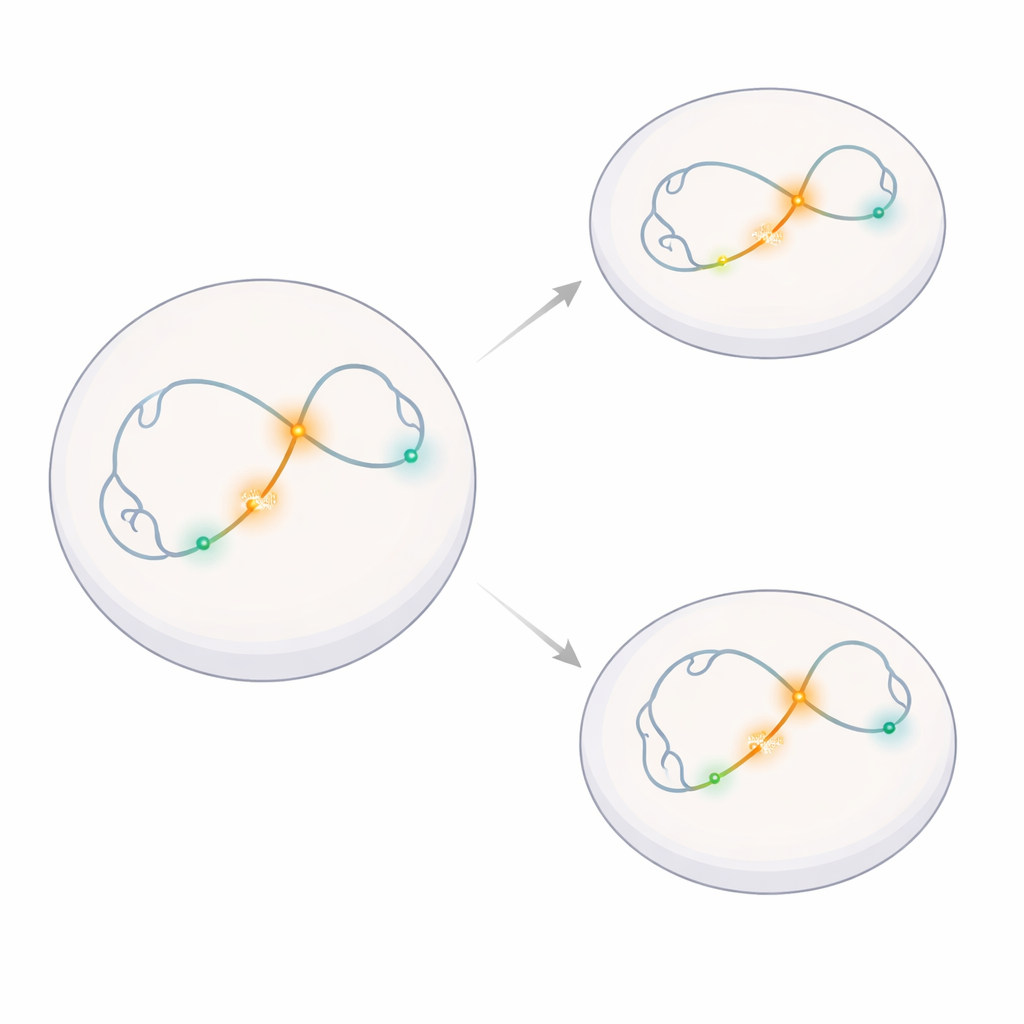
Watching a Single Gene Neighborhood
The researchers focused on a small region of the genome that contains Klf4, a gene important for keeping stem cells in a flexible, early state, and its nearby neighbor Rad23b, a routine “housekeeping” gene. They engineered mouse embryonic stem cells so that tiny fluorescent tags latched onto five chosen spots along this DNA neighborhood: the main Klf4 control hub (a superenhancer), the Klf4 gene start site, the Rad23b gene start site, and two nearby stretches of DNA that do not actively regulate genes. This allowed them to follow the motion of these exact points in living cells over time using high-speed microscopy.
Turning a Gene Off Without Changing Its Dance
To test whether gene activity affects motion, the team used a well-known transition in stem cells. In “naïve” embryonic stem cells, Klf4 is strongly active, helping maintain their ability to become many cell types. When these cells are nudged to become epiblast-like cells—a more advanced state—the main enhancer is chemically silenced and Klf4 expression drops sharply. The scientists confirmed that this genetic switch happened as expected, while Rad23b stayed active in both cell states, and that inserting their fluorescent labeling system did not measurably disturb normal gene activity.
Measuring the DNA’s Subtle Drift
Using automated tracking software and custom analysis, the researchers converted the shifting positions of each fluorescent spot into quantitative measures of motion. All five labeled regions moved in a “subdiffusive” way: instead of wandering freely like smoke in air, their paths were constrained, as if they were moving through a crowded, elastic environment. Crucially, the Klf4 superenhancer and promoter did not move differently from the two nearby control regions, and their motion looked essentially the same whether Klf4 was on in naïve cells or largely off in epiblast-like cells. This challenges the idea that transcription—the act of reading a gene into RNA—automatically tightens or loosens how that stretch of DNA moves.
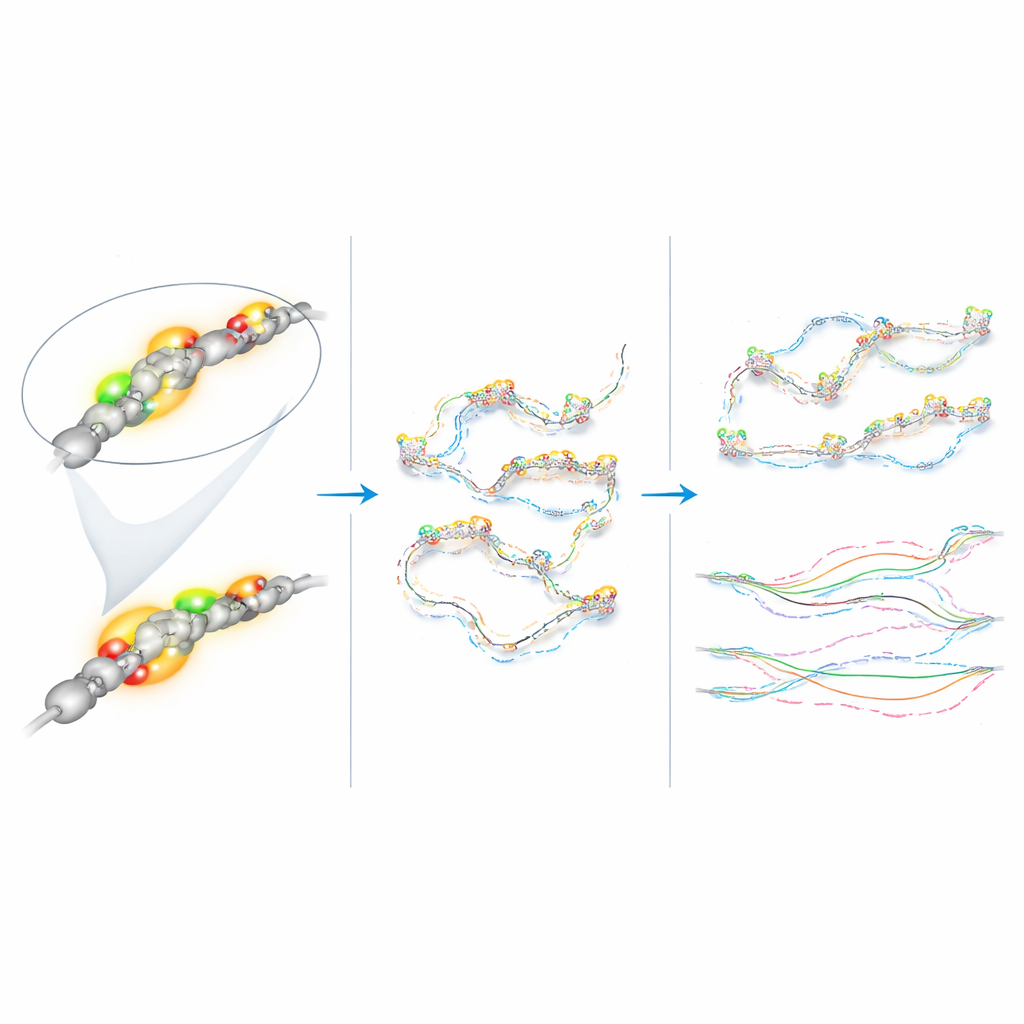
A Fast-Moving Neighbor
One exception stood out. The promoter of the neighboring housekeeping gene Rad23b consistently showed faster motion than all of the Klf4 viewpoints in both cell states. Even within a relatively small segment of the genome, DNA pieces that sit only hundreds of thousands of bases apart could move quite differently. The authors also detected subtle changes in motion for one of the control regions as cells matured, suggesting that broader shifts in the overall chromatin landscape—not just the on–off status of single genes—can alter how DNA drifts within the nucleus.
What This Means for Gene Control
Putting these observations together, the study suggests that simply turning a gene on or off does not necessarily reshape the local movements of its DNA in a major way. For the Klf4 region, active transcription did not cause its key control elements to become distinctly more confined or more mobile than nearby stretches of chromatin. Instead, chromatin motion seems to depend on a mix of local context, broader chromosome organization, and the special properties of individual genes like Rad23b. For readers, the take-home message is that gene regulation is not just about flipping switches; it is also about how pieces of DNA wander and encounter each other—and that wandering can be surprisingly steady, even as genes themselves change activity.
Citation: van Staalduinen, J., Kabbech, H., Yavuz, S. et al. Chromatin dynamics of the Klf4 locus in mouse pluripotent cells. Sci Rep 16, 10941 (2026). https://doi.org/10.1038/s41598-026-45230-9
Keywords: chromatin dynamics, gene regulation, stem cells, Klf4, live-cell imaging