Clear Sky Science · en
The effect of switching from ribociclib to palbociclib due to toxicity in hormone receptor-positive, HER2-negative metastatic breast cancer: a real-world, multicenter, retrospective study
Why this matters to patients and families
Many women with advanced breast cancer now live longer thanks to targeted pills that work with hormone therapy. But these drugs can cause troubling side effects, raising a hard question for patients and doctors: if one medicine causes harm, is it safe and worthwhile to switch to a similar one, or does that risk losing control over the cancer? This study takes a careful look at what happens in real life when women change from one commonly used pill, ribociclib, to another, palbociclib, because of side effects.
Who was studied and what was done
Researchers in Turkey reviewed records from nine cancer centers of women with hormone-sensitive, HER2-negative metastatic breast cancer who first received ribociclib together with hormone therapy. Out of more than a thousand women treated with ribociclib, they focused on 44 who had to stop this drug because of side effects and then switched to palbociclib. All were adults in generally good enough condition to receive treatment. The team collected details on age, tumor features, other illnesses, which hormone partner drug was used, and where the cancer had spread, along with the types of side effects and how long each woman stayed on each drug.
What kinds of side effects led to the switch
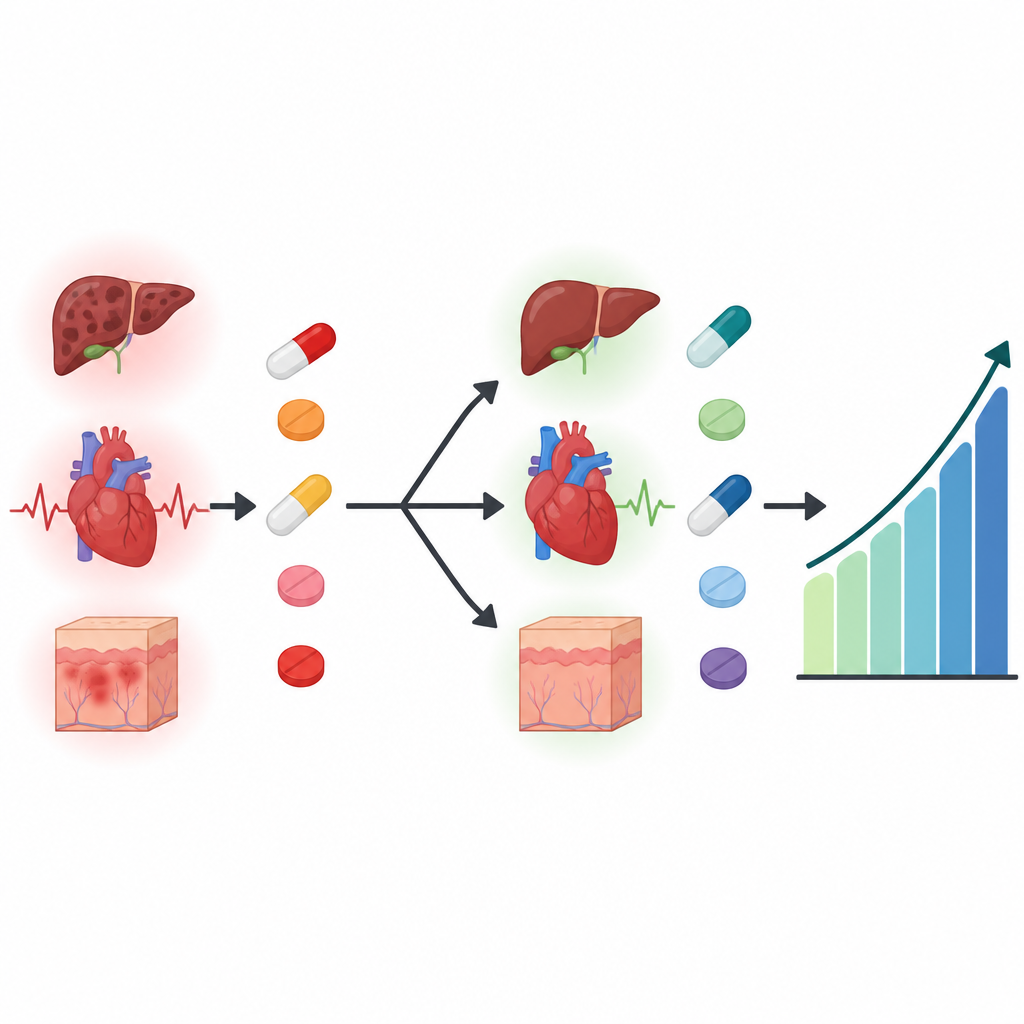
The most frequent problem driving the change was liver irritation, seen as a marked rise in a blood test called alanine aminotransferase in about one third of the patients. Some women developed abnormal heart rhythm findings on their electrocardiogram, including a longer electrical cycle and, in a few cases, a heart block. Others had skin problems such as rash, itching, or color changes, as well as blood count changes, kidney test changes, or hair loss. In most women, doctors first tried lowering the ribociclib dose, but for many this did not fully solve the issue, so the drug was stopped and palbociclib was started instead. After the switch, the original toxicities did not continue, although some women developed expected new side effects of palbociclib such as low white blood cell counts and fatigue, which could be managed.
How well the cancer stayed under control
The study tracked how long treatment could continue without stopping because of cancer growth or side effects, combining the time on ribociclib and the time on palbociclib into a single measure. On average, women remained on these two drugs together for about two and a half years before the cancer clearly worsened or treatment had to end. Roughly seven out of ten women saw their tumors shrink or at least improve measurably at some point, a rate that stayed similar before and after the switch. Importantly, women who changed early, within about four months, did just as well overall as those who switched later. A more detailed time-based analysis showed that the moment of switching did not measurably change the chance of the cancer progressing.
Role of other medicines and study limits
The researchers also asked whether interactions with other drugs could explain the side effects from ribociclib. Only about one in five patients were taking medicines expected to strongly affect how these cancer pills are broken down in the body, and the timing of toxicity was not clearly linked to the level of interaction. This suggests that many side effects likely came from ribociclib itself rather than from other prescriptions. Still, the study has limits: it was retrospective, included only 44 patients, and followed them for just under two years on average, so rare problems or very late effects might have been missed. The results also come from a setting where a third similar drug, abemaciclib, was not widely used or covered by insurance, which may limit how broadly the findings apply.
What this means for treatment choices
For women whose advanced hormone-sensitive breast cancer is well controlled on ribociclib but who develop serious side effects, the fear is that stopping or changing drugs might shorten their lives or allow the disease to flare. This study offers some reassurance. In this real-world group, moving from ribociclib to palbociclib because of side effects did not appear to worsen outcomes, and the troublesome toxicities usually eased after the switch. While larger and longer studies are needed, these findings support the idea that, when dose reduction and brief pauses are not enough, carefully switching to another related pill can keep treatment going without clearly sacrificing cancer control.
Citation: Oruç, A., Deliktaş Onur, İ., Uyar, G.C. et al. The effect of switching from ribociclib to palbociclib due to toxicity in hormone receptor-positive, HER2-negative metastatic breast cancer: a real-world, multicenter, retrospective study. Sci Rep 16, 15027 (2026). https://doi.org/10.1038/s41598-026-44980-w
Keywords: metastatic breast cancer, CDK4/6 inhibitors, ribociclib, palbociclib, drug toxicity