Clear Sky Science · en
Green mycosynthesis of a CuO/ZnO heterojunction nanocomposite using Aspergillus terreus and its antibacterial and anti-virulence activity against multidrug-resistant Escherichia coli
Why stubborn infections matter to all of us
Many common infections that once cleared up with a short course of antibiotics are becoming harder and sometimes impossible to treat. A major culprit is multidrug-resistant Escherichia coli, a type of gut bacterium that can cause painful wound, urinary tract, and bloodstream infections. This study explores an unusual ally in the fight against such superbugs: a harmless soil fungus that helps build tiny metal particles able not only to kill resistant E. coli but also to weaken its ability to cause disease.
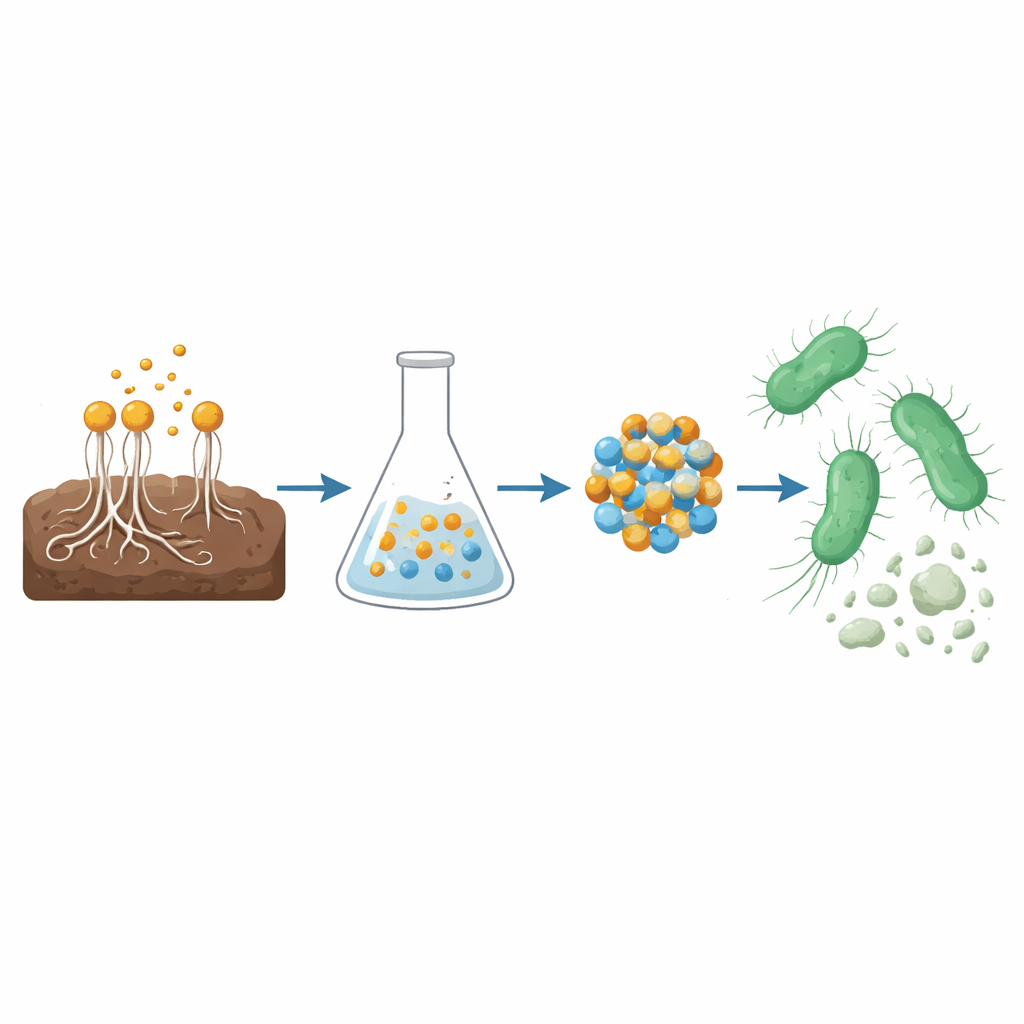
A fungus that works like a tiny factory
The researchers began in farm soil, looking for a fungus that could act as a natural nanotechnology workshop. They isolated and identified a strain of Aspergillus terreus, a filamentous fungus known to secrete a cocktail of organic molecules such as phenolic acids and flavonoids. Using advanced chemical analyses, they confirmed that the fungal broth contained compounds like gallic acid, ferulic acid, and apigenin. These substances can donate electrons and stick to metal surfaces, making them ideal to turn dissolved metal salts into stable, nanosized particles without harsh chemicals or high-energy industrial processes.
Building a two-metal shield against bacteria
Instead of using a single metal, the team combined copper and zinc to form a CuO/ZnO nanocomposite. In practical terms, they mixed the fungal filtrate with copper and zinc acetate solutions. The natural molecules from the fungus grabbed onto copper and zinc ions, reduced them, and guided the formation of extremely small crystals of the two metal oxides. After heating to remove leftover organic material, the result was a heterojunction nanocomposite—interlocking copper oxide and zinc oxide particles about 45 nanometers across, thousands of times thinner than a human hair. Microscopy and spectroscopy confirmed that both oxides were present, well-formed, and tightly connected, a structure known to enhance the generation of highly reactive oxygen-containing species.
Taking on a real-world superbug
To test whether these green-made particles could handle genuine medical threats, the scientists isolated an E. coli strain from wound infections in Iraqi hospitals and showed it resisted every antibiotic tested. On its own, the fungal broth did not stop the bacterium from growing. In contrast, the CuO/ZnO nanocomposite produced clear zones of inhibited growth on culture plates and blocked bacterial multiplication at relatively low concentrations in broth tests. Over time, the particles cut viable bacterial counts by several orders of magnitude, especially at higher doses, indicating true killing rather than just slowed growth. These effects are thought to arise from several combined actions: the particles cling to the bacterial surface, disrupt its protective envelope, release copper and zinc ions that interfere with vital enzymes, and promote formation of reactive oxygen species that damage lipids, proteins, and DNA.
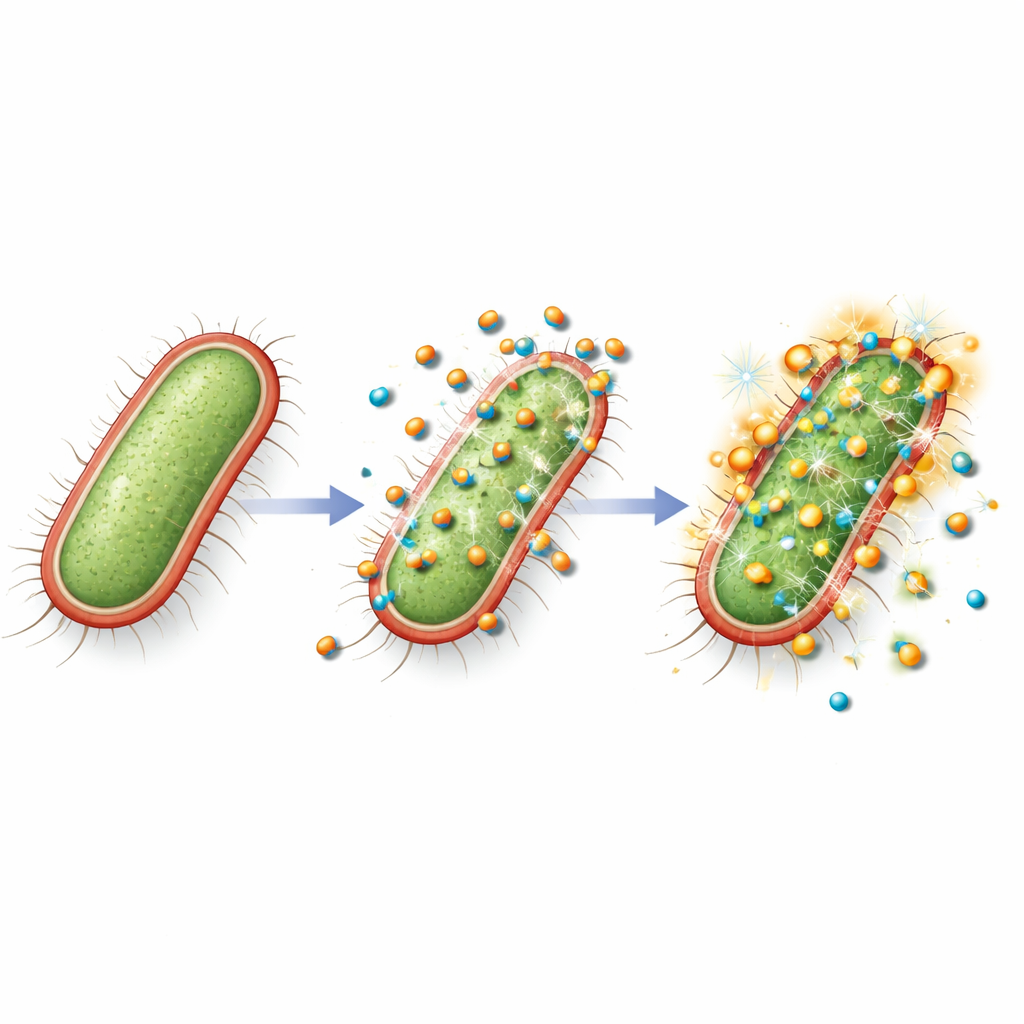
Silencing the tools of infection
Strikingly, the nanocomposite did more than kill bacteria. When the researchers exposed the multidrug-resistant E. coli to a dose too low to stop growth outright, they measured activity of key genes that help the bacterium cling to tissues, communicate with neighbors, and make toxins. Under this sublethal treatment, genes responsible for surface attachment structures and toxin production dropped several-fold in activity, and a central communication gene used in quorum sensing was also strongly suppressed. This means the bacteria that survived exposure were likely less able to form biofilms, coordinate attacks, and damage host cells, effectively disarming them even when they were not fully eliminated.
What this work means for future treatments
Overall, the study shows that a simple soil fungus can be harnessed to build a copper–zinc nanoparticle blend that acts on multidrug-resistant E. coli in two ways: it attacks the cells directly and at the same time turns down the genetic programs that make them dangerous. Because the process avoids toxic reagents and uses low-cost metals, it could be scaled up more sustainably than many current nanomaterials. Before it can be used in clinics, scientists must still confirm safety in animals and people and test how well it works in real wounds or on medical devices. But this fungus-built nanocomposite points toward a new class of treatments that not only kill superbugs, but also strip them of their most harmful tricks.
Citation: Obaid, A.N., Abdelghany, T.M., Soliman, A.M. et al. Green mycosynthesis of a CuO/ZnO heterojunction nanocomposite using Aspergillus terreus and its antibacterial and anti-virulence activity against multidrug-resistant Escherichia coli. Sci Rep 16, 12350 (2026). https://doi.org/10.1038/s41598-026-44775-z
Keywords: multidrug-resistant E. coli, green nanotechnology, metal oxide nanoparticles, antibacterial coatings, quorum sensing inhibition