Clear Sky Science · en
Identification and validation of NETs-associated biomarkers in osteoporosis with diabetes
Why Bones and Blood Sugar Belong in the Same Conversation
As people live longer, more of us are facing two common but often separate diagnoses: osteoporosis, which weakens bones, and diabetes, which disrupts blood sugar. This study brings those worlds together by asking a simple but important question: is there a shared biological thread that helps explain why many people with diabetes also have fragile bones? By focusing on a little‑known defense mechanism of white blood cells, the researchers search for warning signals in the blood that could one day help doctors spot and perhaps even target this dangerous pairing earlier.
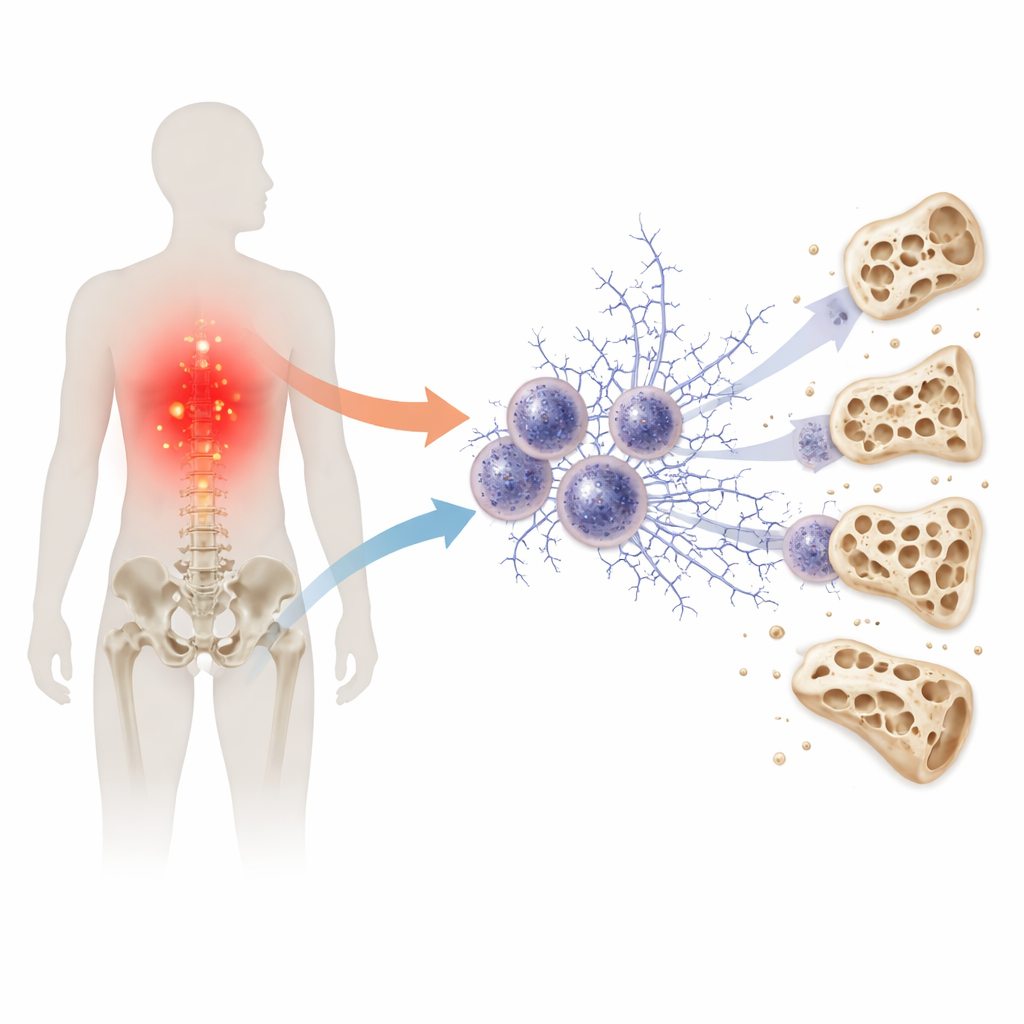
A Hidden Web in the Immune System
Our first line of defense against germs includes neutrophils, fast‑moving white blood cells that can cast out sticky webs made of DNA and proteins to trap invaders. These structures, called extracellular traps, are helpful in short bursts but can cause harm when they appear too often or linger too long. Earlier work has linked these immune webs to diabetic complications in the kidneys and blood vessels. The current study asks whether the same process might also connect diabetes to thinning bones, potentially turning an acute defense mechanism into a chronic source of damage.
Reading Molecular Clues in the Blood
To explore this idea, the team analyzed gene activity in blood samples from 15 people with both osteoporosis and diabetes and 15 healthy volunteers. They focused on 69 genes already known to be involved in forming neutrophil webs, then looked for which of these were switched on or off differently in patients. Using advanced computer methods, including two types of machine learning, they narrowed more than two thousand altered genes down to just two standout candidates whose activity patterns best separated patients from healthy controls.
Two Standout Signals: S100A12 and SLC25A37
The two highlighted genes, called S100A12 and SLC25A37, point to inflammation and energy metabolism as key players. S100A12 encodes a protein released by immune cells that can amplify inflammation and is already known to rise in several diabetic complications. SLC25A37 is involved in moving iron into the tiny power plants inside cells, the mitochondria, affecting how cells handle energy and oxidative stress. In a small follow‑up group, the researchers measured the proteins encoded by these genes in blood and found that both were clearly higher in people with diabetes‑related bone loss than in healthy participants, even when their gene activity levels were more variable.
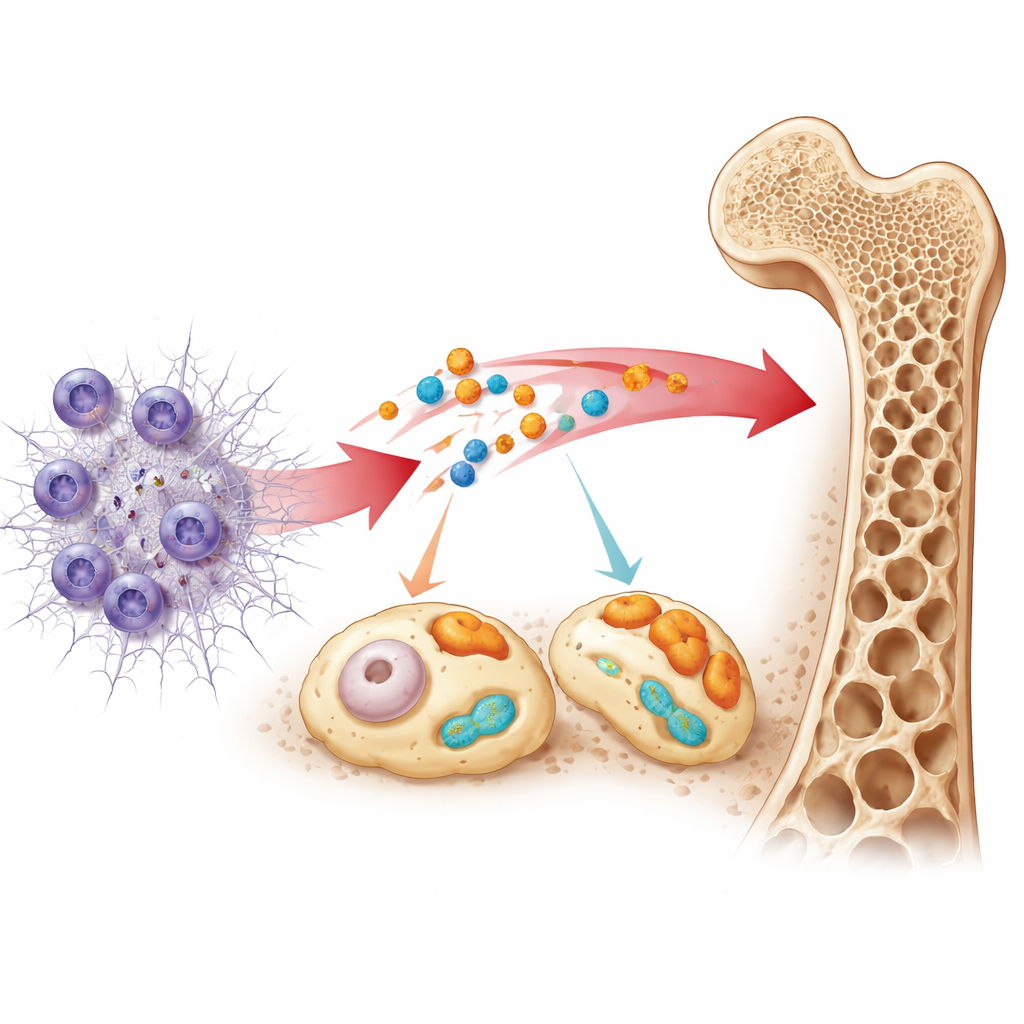
What These Signals Say About the Body’s Defenses
By mapping which biological pathways are linked to these two markers, the study suggests that they sit at crossroads between immune reactions, metabolism, and bone remodeling. S100A12 was tied to pathways that respond to sugar‑damaged proteins and to signaling systems that can spur bone breakdown. SLC25A37 was linked to pathways involving protein production and sensors that detect danger in the body. The team also found shifts in several types of immune cells, especially neutrophils, in patients compared with healthy volunteers. Finally, by mining drug databases, they identified existing medicines that act on related signaling systems, hinting at possible future treatment directions, though these leads remain speculative.
What This Could Mean for Patients
Taken together, the findings suggest that high blood sugar, overactive immune webs, and disturbed cell energy all converge to weaken bone in people with diabetes. S100A12 and SLC25A37 emerge as promising blood‑based indicators of this process and as starting points for thinking about new therapies, such as drugs that calm harmful inflammation or restore healthy signaling between nerves, blood vessels, and bone. Because this was a small, early‑stage study that relied heavily on computer analysis, its results need to be confirmed in larger and more diverse groups, and the roles of these markers must be tested directly in cells and animal models. Still, the work lays a foundation for turning a complex tangle of immune and metabolic changes into practical tools for predicting and eventually reducing fracture risk in people living with diabetes.
Citation: Zhang, L., Hao, L., Wang, Y. et al. Identification and validation of NETs-associated biomarkers in osteoporosis with diabetes. Sci Rep 16, 14074 (2026). https://doi.org/10.1038/s41598-026-44721-z
Keywords: diabetic osteoporosis, neutrophil extracellular traps, bone fragility, inflammation and metabolism, biomarkers